Blood Money: Paying for Convalescent Plasma to Treat COVID-19
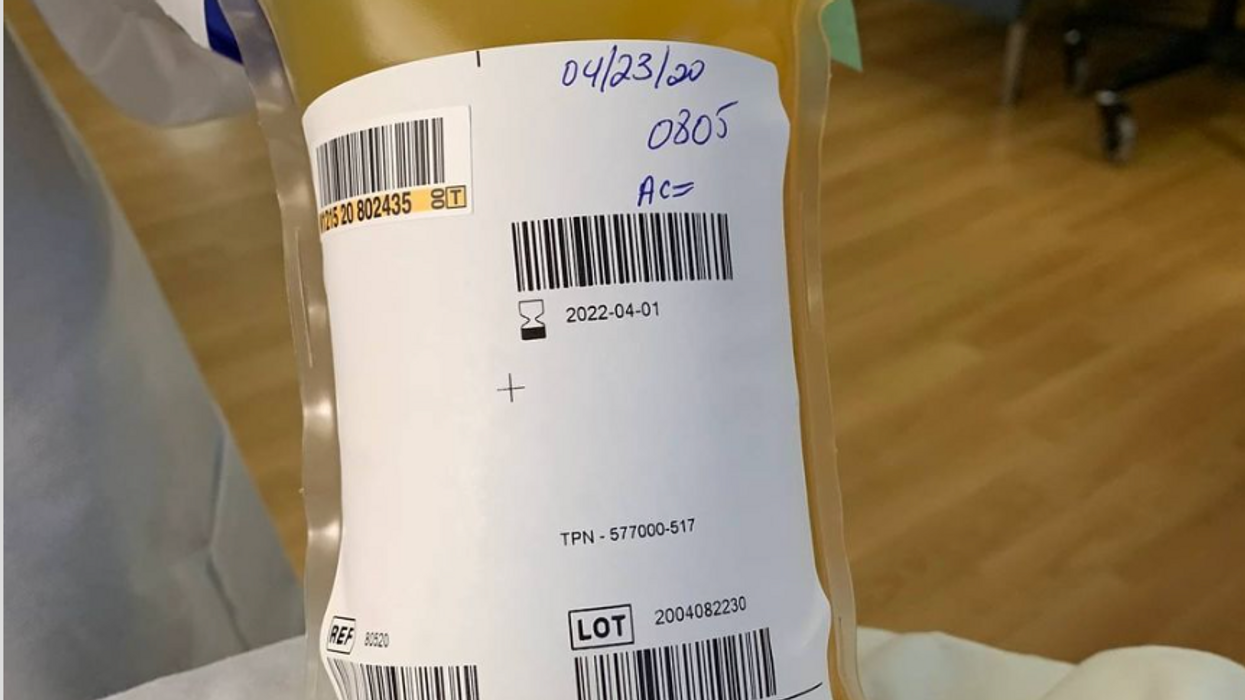
A bag of plasma that Tom Hanks donated back in April 2020 after his coronavirus infection. (He was not paid to donate.)
Convalescent plasma – first used to treat diphtheria in 1890 – has been dusted off the shelf to treat COVID-19. Does it work? Should we rely strictly on the altruism of donors or should people be paid for it?
The biologic theory is that a person who has recovered from a disease has chemicals in their blood, most likely antibodies, that contributed to their recovery, and transferring those to a person who is sick might aid their recovery. Whole blood won't work because there are too few antibodies in a single unit of blood and the body can hold only so much of it.
Plasma comprises about 55 percent of whole blood and is what's left once you take out the red blood cells that carry oxygen and the white blood cells of the immune system. Most of it is water but the rest is a complex mix of fats, salts, signaling molecules and proteins produced by the immune system, including antibodies.
A process called apheresis circulates the donors' blood through a machine that separates out the desired parts of blood and returns the rest to the donor. It takes several times the length of a regular whole blood donation to cycle through enough blood for the process. The end product is a yellowish concentration called convalescent plasma.
Recent History
It was used extensively during the great influenza epidemic off 1918 but fell out of favor with the development of antibiotics. Still, whenever a new disease emerges – SARS, MERS, Ebola, even antibiotic-resistant bacteria – doctors turn to convalescent plasma, often as a stopgap until more effective antibiotic and antiviral drugs are developed. The process is certainly safe when standard procedures for handling blood products are followed, and historically it does seem to be beneficial in at least some patients if administered early enough in the disease.
With few good treatment options for COVID-19, doctors have given convalescent plasma to more than a hundred thousand Americans and tens of thousand of people elsewhere, to mixed results. Placebo-controlled trials could give a clearer picture of plasma's value but it is difficult to enroll patients facing possible death when the flip of a coin will determine who will receive a saline solution or plasma.
And the plasma itself isn't some uniform pill stamped out in a factory, it's a natural product that is shaped by the immune history of the donor's body and its encounter not just with SARS-CoV-2 but a lifetime of exposure to different pathogens.
Researchers believe antibodies in plasma are a key factor in directly fighting the virus. But the variety and quantity of antibodies vary from donor to donor, and even over time from the same donor because once the immune system has cleared the virus from the body, it stops putting out antibodies to fight the virus. Often the quality and quantity of antibodies being given to a patient are not measured, making it somewhat hit or miss, which is why several companies have recently developed monoclonal antibodies, a single type of antibody found in blood that is effective against SARS-CoV-2 and that is multiplied in the lab for use as therapy.
Plasma may also contain other unknown factors that contribute to fighting disease, say perhaps signaling molecules that affect gene expression, which might affect the movement of immune cells, their production of antiviral molecules, or the regulation of inflammation. The complexity and lack of standardization makes it difficult to evaluate what might be working or not with a convalescent plasma treatment. Thus researchers are left with few clues about how to make it more effective.
Industrializing Plasma
Many Americans living along the border with Mexico regularly head south to purchase prescription drugs at a significant discount. Less known is the medical traffic the other way, Mexicans who regularly head north to be paid for plasma donations, which are prohibited in their country; the U.S. allows payment for plasma donations but not whole blood. A typical payment is about $35 for a donation but the sudden demand for convalescent plasma from people who have recovered from COVID-19 commands a premium price, sometimes as high as $200. These donors are part of a fast-growing plasma industry that surpassed $25 billion in 2018. The U.S. supplies about three-quarters of the world's needs for plasma.
Payment for whole blood donation in the U.S. is prohibited, and while payment for plasma is allowed, there is a stigma attached to payment and much plasma is donated for free.
The pharmaceutical industry has shied away from natural products they cannot patent but they have identified simpler components from plasma, such as clotting factors and immunoglobulins, that have been turned into useful drugs from this raw material of plasma. While some companies have retooled to provide convalescent plasma to treat COVID-19, often paying those donors who have recovered a premium of several times the normal rate, most convalescent plasma has come as donations through traditional blood centers.
In April the Mayo Clinic, in cooperation with the FDA, created an expanded access program for convalescent plasma to treat COVID-19. It was meant to reduce the paperwork associated with gaining access to a treatment not yet approved by the FDA for that disease. Initially it was supposed to be for 5000 units but it quickly grew to more than twenty times that size. Michael Joyner, the head of the program, discussed that experience in an extended interview in September.
The Centers for Medicare and Medicaid Services (CMS) also created associated reimbursement codes, which became permanent in August.
Mayo published an analysis of the first 35,000 patients as a preprint in August. It concluded, "The relationships between mortality and both time to plasma transfusion, and antibody levels provide a signature that is consistent with efficacy for the use of convalescent plasma in the treatment of hospitalized COVID-19 patients."
It seemed to work best when given early in infection and in larger doses; a similar pattern has been seen in studies of monoclonal antibodies. A revised version will soon be published in a major medical journal. Some criticized the findings as not being from a randomized clinical trial.
Convalescent plasma is not the only intervention that seems to work better when used earlier in the course of disease. Recently the pharmaceutical company Eli Lilly stopped a clinical trial of a monoclonal antibody in hospitalized COVID-19 patients when it became apparent it wasn't helping. It is continuing trials for patients who are less sick and begin treatment earlier, as well as in persons who have been exposed to the virus but not yet diagnosed as infected, to see if it might prevent infection. In November the FDA eased access to this drug outside of clinical trials, though it is not yet approved for sale.
Show Me the Money
The antibodies that seem to give plasma its curative powers are fragile proteins that the body produces to fight the virus. Production shuts down once the virus is cleared and the remaining antibodies survive only for a few weeks before the levels fade. [Vaccines are used to train immune cells to produce antibodies and other defenses to respond to exposure to future pathogens.] So they can be usefully harvested from a recovered patient for only a few short weeks or months before they decline precipitously. The question becomes, how does one mobilize this resource in that short window of opportunity?
The program run by the Mayo Clinic explains the process and criteria for donating convalescent plasma for COVID-19, as well as links to local blood centers equipped to handle those free donations. Commercial plasma centers also are advertising and paying for donations.
A majority of countries prohibit paying donors for blood or blood products, including India. But an investigation by India Today touted a black market of people willing to donate convalescent plasma for the equivalent of several hundred dollars. Officials vowed to prosecute, saying donations should be selfless.
But that enforcement threat seemed to be undercut when the health minister of the state of Assam declared "plasma donors will get preference in several government schemes including the government job interview." It appeared to be a form of compensation that far surpassed simple cash.
The small city of Rexburg, Idaho, with a population a bit over 50,000, overwhelmingly Mormon and home to a campus of Brigham Young University, at one point had one of the highest per capita rates of COVID-19 in the current wave of infection. Rumors circulated that some students were intentionally trying to become infected so they could later sell their plasma for top dollar, potentially as much as $200 a visit.
Troubled university officials investigated the allegations but could come up with nothing definitive; how does one prove intentionality with such an omnipresent yet elusive virus? They chalked it up to idle chatter, perhaps an urban legend, which might be associated with alcohol use on some other campus.
Doctors, hospitals, and drug companies are all rightly praised for their altruism in the fight against COVID-19, but they also get paid. Payment for whole blood donation in the U.S. is prohibited, and while payment for plasma is allowed, there is a stigma attached to payment and much plasma is donated for free. "Why do we expect the donors [of convalescent plasma] to be the only uncompensated people in the process? It really makes no sense," argues Mark Yarborough, an ethicist at the UC Davis School of Medicine in Sacramento.
"When I was in grad school, two of my closest friends, at least once a week they went and gave plasma. That was their weekend spending money," Yarborough recalls. He says upper and middle-income people may have the luxury of donating blood products but prohibiting people from selling their plasma is a bit paternalistic and doesn't do anything to improve the economic status of poor people.
"Asking people to dedicate two hours a week for an entire year in exchange for cookies and milk is demonstrably asking too much," says Peter Jaworski, an ethicist who teaches at Georgetown University.
He notes that companies that pay plasma donors have much lower total costs than do operations that rely solely on uncompensated donations. The companies have to spend less to recruit and retain donors because they increase payments to encourage regular repeat donations. They are able to more rationally schedule visits to maximize use of expensive apheresis equipment and medical personnel used for the collection.
It seems that COVID-19 has been with us forever, but in reality it is less than a year. We have learned much over that short time, can now better manage the disease, and have lower mortality rates to prove it. Just how much convalescent plasma may have contributed to that remains an open question. Access to vaccines is months away for many people, and even then some people will continue to get sick. Given the lack of proven treatments, it makes sense to keep plasma as part of the mix, and not close the door to any legitimate means to obtain it.
This man spent over 70 years in an iron lung. What he was able to accomplish is amazing.
Paul Alexander spent more than 70 years confined to an iron lung after a polio infection left him paralyzed at age 6. Here, Alexander uses a mirror attached to the top of his iron lung to view his surroundings.
It’s a sight we don’t normally see these days: A man lying prone in a big, metal tube with his head sticking out of one end. But it wasn’t so long ago that this sight was unfortunately much more common.
In the first half of the 20th century, tens of thousands of people each year were infected by polio—a highly contagious virus that attacks nerves in the spinal cord and brainstem. Many people survived polio, but a small percentage of people who did were left permanently paralyzed from the virus, requiring support to help them breathe. This support, known as an “iron lung,” manually pulled oxygen in and out of a person’s lungs by changing the pressure inside the machine.
Paul Alexander was one of several thousand who were infected and paralyzed by polio in 1952. That year, a polio epidemic swept the United States, forcing businesses to close and polio wards in hospitals all over the country to fill up with sick children. When Paul caught polio in the summer of 1952, doctors urged his parents to let him rest and recover at home, since the hospital in his home suburb of Dallas, Texas was already overrun with polio patients.
Paul rested in bed for a few days with aching limbs and a fever. But his condition quickly got worse. Within a week, Paul could no longer speak or swallow, and his parents rushed him to the local hospital where the doctors performed an emergency procedure to help him breathe. Paul woke from the surgery three days later, and found himself unable to move and lying inside an iron lung in the polio ward, surrounded by rows of other paralyzed children.

But against all odds, Paul lived. And with help from a physical therapist, Paul was able to thrive—sometimes for small periods outside the iron lung.
The way Paul did this was to practice glossopharyngeal breathing (or as Paul called it, “frog breathing”), where he would trap air in his mouth and force it down his throat and into his lungs by flattening his tongue. This breathing technique, taught to him by his physical therapist, would allow Paul to leave the iron lung for increasing periods of time.
With help from his iron lung (and for small periods of time without it), Paul managed to live a full, happy, and sometimes record-breaking life. At 21, Paul became the first person in Dallas, Texas to graduate high school without attending class in person, owing his success to memorization rather than taking notes. After high school, Paul received a scholarship to Southern Methodist University and pursued his dream of becoming a trial lawyer and successfully represented clients in court.

Throughout his long life, Paul was also able to fly on a plane, visit the beach, adopt a dog, fall in love, and write a memoir using a plastic stick to tap out a draft on a keyboard. In recent years, Paul joined TikTok and became a viral sensation with more than 330,000 followers. In one of his first videos, Paul advocated for vaccination and warned against another polio epidemic.
Paul was reportedly hospitalized with COVID-19 at the end of February and died on March 11th, 2024. He currently holds the Guiness World Record for longest survival inside an iron lung—71 years.
Polio thankfully no longer circulates in the United States, or in most of the world, thanks to vaccines. But Paul continues to serve as a reminder of the importance of vaccination—and the power of the human spirit.
““I’ve got some big dreams. I’m not going to accept from anybody their limitations,” he said in a 2022 interview with CNN. “My life is incredible.”
When doctors couldn’t stop her daughter’s seizures, this mom earned a PhD and found a treatment herself.
Savannah Salazar (left) and her mother, Tracy Dixon-Salazaar, who earned a PhD in neurobiology in the quest for a treatment of her daughter's seizure disorder.
Twenty-eight years ago, Tracy Dixon-Salazaar woke to the sound of her daughter, two-year-old Savannah, in the midst of a medical emergency.
“I entered [Savannah’s room] to see her tiny little body jerking about violently in her bed,” Tracy said in an interview. “I thought she was choking.” When she and her husband frantically called 911, the paramedic told them it was likely that Savannah had had a seizure—a term neither Tracy nor her husband had ever heard before.
Over the next several years, Savannah’s seizures continued and worsened. By age five Savannah was having seizures dozens of times each day, and her parents noticed significant developmental delays. Savannah was unable to use the restroom and functioned more like a toddler than a five-year-old.
Doctors were mystified: Tracy and her husband had no family history of seizures, and there was no event—such as an injury or infection—that could have caused them. Doctors were also confused as to why Savannah’s seizures were happening so frequently despite trying different seizure medications.
Doctors eventually diagnosed Savannah with Lennox-Gaustaut Syndrome, or LGS, an epilepsy disorder with no cure and a poor prognosis. People with LGS are often resistant to several kinds of anti-seizure medications, and often suffer from developmental delays and behavioral problems. People with LGS also have a higher chance of injury as well as a higher chance of sudden unexpected death (SUDEP) due to the frequent seizures. In about 70 percent of cases, LGS has an identifiable cause such as a brain injury or genetic syndrome. In about 30 percent of cases, however, the cause is unknown.
Watching her daughter struggle through repeated seizures was devastating to Tracy and the rest of the family.
“This disease, it comes into your life. It’s uninvited. It’s unannounced and it takes over every aspect of your daily life,” said Tracy in an interview with Today.com. “Plus it’s attacking the thing that is most precious to you—your kid.”
Desperate to find some answers, Tracy began combing the medical literature for information about epilepsy and LGS. She enrolled in college courses to better understand the papers she was reading.
“Ironically, I thought I needed to go to college to take English classes to understand these papers—but soon learned it wasn’t English classes I needed, It was science,” Tracy said. When she took her first college science course, Tracy says, she “fell in love with the subject.”
Tracy was now a caregiver to Savannah, who continued to have hundreds of seizures a month, as well as a full-time student, studying late into the night and while her kids were at school, using classwork as “an outlet for the pain.”
“I couldn’t help my daughter,” Tracy said. “Studying was something I could do.”
Twelve years later, Tracy had earned a PhD in neurobiology.
After her post-doctoral training, Tracy started working at a lab that explored the genetics of epilepsy. Savannah’s doctors hadn’t found a genetic cause for her seizures, so Tracy decided to sequence her genome again to check for other abnormalities—and what she found was life-changing.
Tracy discovered that Savannah had a calcium channel mutation, meaning that too much calcium was passing through Savannah’s neural pathways, leading to seizures. The information made sense to Tracy: Anti-seizure medications often leech calcium from a person’s bones. When doctors had prescribed Savannah calcium supplements in the past to counteract these effects, her seizures had gotten worse every time she took the medication. Tracy took her discovery to Savannah’s doctor, who agreed to prescribe her a calcium blocker.
The change in Savannah was almost immediate.
Within two weeks, Savannah’s seizures had decreased by 95 percent. Once on a daily seven-drug regimen, she was soon weaned to just four, and then three. Amazingly, Tracy started to notice changes in Savannah’s personality and development, too.
“She just exploded in her personality and her talking and her walking and her potty training and oh my gosh she is just so sassy,” Tracy said in an interview.
Since starting the calcium blocker eleven years ago, Savannah has continued to make enormous strides. Though still unable to read or write, Savannah enjoys puzzles and social media. She’s “obsessed” with boys, says Tracy. And while Tracy suspects she’ll never be able to live independently, she and her daughter can now share more “normal” moments—something she never anticipated at the start of Savannah’s journey with LGS. While preparing for an event, Savannah helped Tracy get ready.
“We picked out a dress and it was the first time in our lives that we did something normal as a mother and a daughter,” she said. “It was pretty cool.”

