Genome Reading and Editing Tools for All

An open book representing the ability to read the human genome.
In 2006, the cover of Scientific American was "Know Your DNA" and the inside story was "Genomes for All." Today, we are closer to that goal than ever. Making it affordable for everyone to understand and change their DNA will fundamentally alter how we manage diseases, how we conduct clinical research, and even how we select a mate.
A frequent line of questions on the topic of making genome reading affordable is: Do we need to read the whole genome in order to accurately predict disease risk?
Since 2006, we have driven the cost of reading a human genome down from $3 billion to $600. To aid interpretation and research to produce new diagnostics and therapeutics, my research team at Harvard initiated the Personal Genome Project and later, Openhumans.org. This has demonstrated international informed consent for human genomes, and diverse environmental and trait data can be distributed freely. This is done with no strings attached in a manner analogous to Wikipedia. Cell lines from that project are similarly freely available for experiments on synthetic biology, gene therapy and human developmental biology. DNA from those cells have been chosen by the US National Institute of Standards and Technology and the Food and Drug Administration to be the key federal standards for the human genome.
A frequent line of questions on the topic of making genome reading affordable is: Do we need to read the whole genome in order to accurately predict disease risk? Can we just do most commonly varying parts of the genome, which constitute only a tiny fraction of a percent? Or just the most important parts encoding the proteins or 'exome,' which constitute about one percent of the genome? The commonly varying parts of the genome are poor predictors of serious genetic diseases and the exomes don't detect DNA rearrangements which often wipe out gene function when they occur in non-coding regions within genes. Since the cost of the exome is not one percent of the whole genome cost, but nearly identical ($600), missing an impactful category of mutants is really not worth it. So the answer is yes, we should read the whole genome to glean comprehensively meaningful information.
In parallel to the reading revolution, we have dropped the price of DNA synthesis by a similar million-fold and made genome editing tools close to free.
WRITING
In parallel to the reading revolution, we have dropped the price of DNA synthesis by a similar million-fold and made genome editing tools like CRISPR, TALE and MAGE close to free by distributing them through the non-profit Addgene.org. Gene therapies are already curing blindness in children and cancer in adults, and hopefully soon infectious diseases and hemoglobin diseases like sickle cell anemia. Nevertheless, gene therapies are (so far) the most expensive class of drugs in history (about $1 million dollars per dose).
This is in large part because the costs of proving safety and efficacy in a randomized clinical trial are high and that cost is spread out only over the people that benefit (aka the denominator). Striking growth is evident in such expensive hyper-personalized therapies ever since the "Orphan Drug Act of 1983." For the most common disease, aging (which kills 90 percent of people in wealthy regions of the world), the denominator is maximal and the cost of the drugs should be low as genetic interventions to combat aging become available in the next ten years. But what can we do about rarer diseases with cheap access to genome reading and editing tools? Try to prevent them in the first place.
A huge fraction of these births is preventable if unaffected carriers of such diseases do not mate.
ARITHMETIC
While the cost of reading has plummeted, the value of knowing your genome is higher than ever. About 5 percent of births result in extreme medical trauma over a person's lifetime due to rare genetic diseases. Even without gene therapy, these cost the family and society more than a million dollars in drugs, diagnostics and instruments, extra general care, loss of income for the affected individual and other family members, plus pain and anxiety of the "medical odyssey" often via dozens of mystified physicians. A huge fraction of these births is preventable if unaffected carriers of such diseases do not mate.
The non-profit genetic screening organization, Dor Yeshorim (established in 1983), has shown that this is feasible by testing for Tay–Sachs disease, Familial dysautonomia, Cystic fibrosis, Canavan disease, Glycogen storage disease (type 1), Fanconi anemia (type C), Bloom syndrome, Niemann–Pick disease, Mucolipidosis type IV. This is often done at the pre-marital, matchmaking phase, which can reduce the frequency of natural or induced abortions. Such matchmaking can be done in such a way that no one knows the carrier status of any individual in the system. In addition to those nine tests, many additional diseases can be picked up by whole genome sequencing. No person can know in advance that they are exempt from these risks.
Furthermore, concerns about rare "false positives" is far less at the stage of matchmaking than at the stage of prenatal testing, since the latter could involve termination of a healthy fetus, while the former just means that you restrict your dating to 90 percent of the population. In order to scale this up from 13 million Ashkenazim and Sephardim to billions in diverse cultures, we will likely see new computer security, encryption, blockchain and matchmaking tools.
Once the diseases are eradicated from our population, the interventions can be said to impact not only the current population, but all subsequent generations.
THE FUTURE
As reading and writing become exponentially more affordable and reliable, we can tackle equitable distribution, but there remain issues of education and security. Society, broadly (insurers, health care providers, governments) should be able to see a roughly 12-fold return on their investment of $1800 per person ($600 each for raw data, interpretation and incentivizing the participant) by saving $1 million per diseased child per 20 families. Everyone will have free access to their genome information and software to guide their choices in precision medicines, mates and participation in biomedical research studies.
In terms of writing and editing, if delivery efficiency and accuracy keep improving, then pill or aerosol formulations of gene therapies -- even non-prescription, veterinary or home-made versions -- are not inconceivable. Preventions tends to be more affordable and more humane than cures. If gene therapies provide prevention of diseases of aging, cancer and cognitive decline, they might be considered "enhancement," but not necessarily more remarkable than past preventative strategies, like vaccines against HPV-cancer, smallpox and polio. Whether we're overcoming an internal genetic flaw or an external infectious disease, the purpose is the same: to minimize human suffering. Once the diseases are eradicated from our population, the interventions can be said to impact not only the current population, but all subsequent generations. This reminds us that we need to listen carefully, educate each other and proactively imagine and deflect likely, and even unlikely, unintended consequences, including stigmatization of the last few unprotected individuals.
Urinary tract infections account for more than 8 million trips to the doctor each year.
Few things are more painful than a urinary tract infection (UTI). Common in men and women, these infections account for more than 8 million trips to the doctor each year and can cause an array of uncomfortable symptoms, from a burning feeling during urination to fever, vomiting, and chills. For an unlucky few, UTIs can be chronic—meaning that, despite treatment, they just keep coming back.
But new research, presented at the European Association of Urology (EAU) Congress in Paris this week, brings some hope to people who suffer from UTIs.
Clinicians from the Royal Berkshire Hospital presented the results of a long-term, nine-year clinical trial where 89 men and women who suffered from recurrent UTIs were given an oral vaccine called MV140, designed to prevent the infections. Every day for three months, the participants were given two sprays of the vaccine (flavored to taste like pineapple) and then followed over the course of nine years. Clinicians analyzed medical records and asked the study participants about symptoms to check whether any experienced UTIs or had any adverse reactions from taking the vaccine.
The results showed that across nine years, 48 of the participants (about 54%) remained completely infection-free. On average, the study participants remained infection free for 54.7 months—four and a half years.
“While we need to be pragmatic, this vaccine is a potential breakthrough in preventing UTIs and could offer a safe and effective alternative to conventional treatments,” said Gernot Bonita, Professor of Urology at the Alta Bro Medical Centre for Urology in Switzerland, who is also the EAU Chairman of Guidelines on Urological Infections.
The news comes as a relief not only for people who suffer chronic UTIs, but also to doctors who have seen an uptick in antibiotic-resistant UTIs in the past several years. Because UTIs usually require antibiotics, patients run the risk of developing a resistance to the antibiotics, making infections more difficult to treat. A preventative vaccine could mean less infections, less antibiotics, and less drug resistance overall.
“Many of our participants told us that having the vaccine restored their quality of life,” said Dr. Bob Yang, Consultant Urologist at the Royal Berkshire NHS Foundation Trust, who helped lead the research. “While we’re yet to look at the effect of this vaccine in different patient groups, this follow-up data suggests it could be a game-changer for UTI prevention if it’s offered widely, reducing the need for antibiotic treatments.”
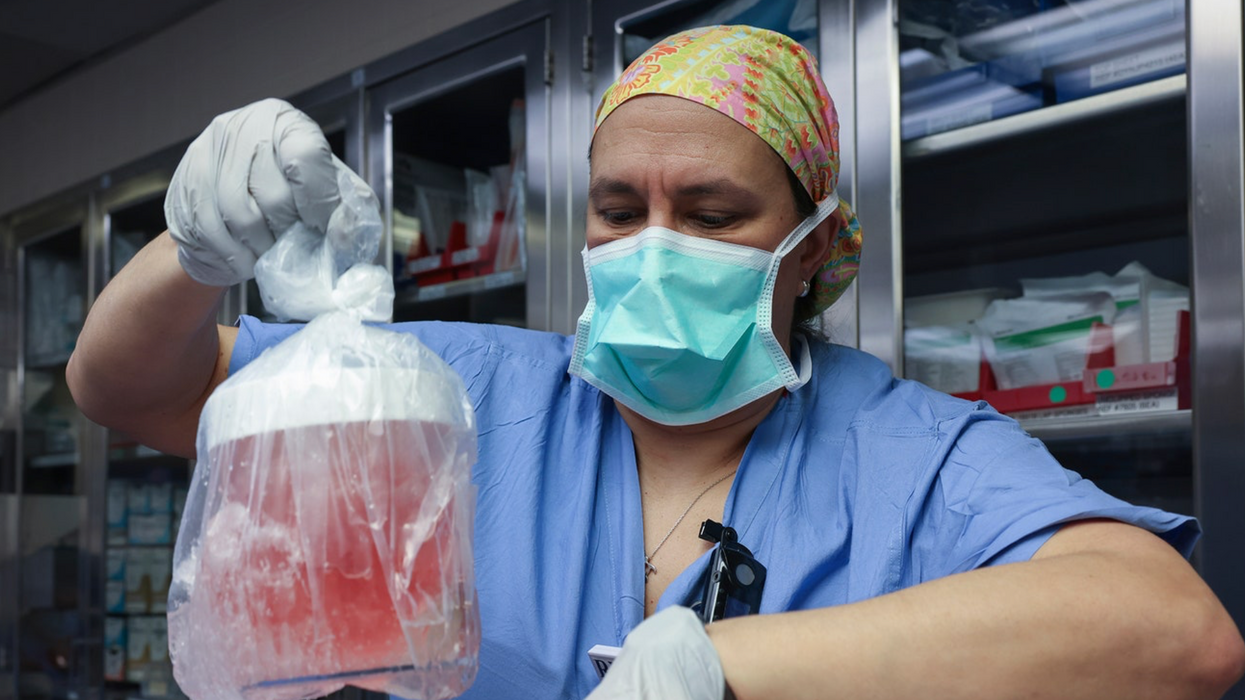
MILESTONE: Doctors have transplanted a pig organ into a human for the first time in history
A surgeon at Massachusetts General Hospital prepares a pig organ for transplant.
Surgeons at Massachusetts General Hospital made history last week when they successfully transplanted a pig kidney into a human patient for the first time ever.
The recipient was a 62-year-old man named Richard Slayman who had been living with end-stage kidney disease caused by diabetes. While Slayman had received a kidney transplant in 2018 from a human donor, his diabetes ultimately caused the kidney to fail less than five years after the transplant. Slayman had undergone dialysis ever since—a procedure that uses an artificial kidney to remove waste products from a person’s blood when the kidneys are unable to—but the dialysis frequently caused blood clots and other complications that landed him in the hospital multiple times.
As a last resort, Slayman’s kidney specialist suggested a transplant using a pig kidney provided by eGenesis, a pharmaceutical company based in Cambridge, Mass. The highly experimental surgery was made possible with the Food and Drug Administration’s “compassionate use” initiative, which allows patients with life-threatening medical conditions access to experimental treatments.
The new frontier of organ donation
Like Slayman, more than 100,000 people are currently on the national organ transplant waiting list, and roughly 17 people die every day waiting for an available organ. To make up for the shortage of human organs, scientists have been experimenting for the past several decades with using organs from animals such as pigs—a new field of medicine known as xenotransplantation. But putting an animal organ into a human body is much more complicated than it might appear, experts say.
“The human immune system reacts incredibly violently to a pig organ, much more so than a human organ,” said Dr. Joren Madsen, director of the Mass General Transplant Center. Even with immunosuppressant drugs that suppress the body’s ability to reject the transplant organ, Madsen said, a human body would reject an animal organ “within minutes.”
So scientists have had to use gene-editing technology to change the animal organs so that they would work inside a human body. The pig kidney in Slayman’s surgery, for instance, had been genetically altered using CRISPR-Cas9 technology to remove harmful pig genes and add human ones. The kidney was also edited to remove pig viruses that could potentially infect a human after transplant.
With CRISPR technology, scientists have been able to prove that interspecies organ transplants are not only possible, but may be able to successfully work long term, too. In the past several years, scientists were able to transplant a pig kidney into a monkey and have the monkey survive for more than two years. More recently, doctors have transplanted pig hearts into human beings—though each recipient of a pig heart only managed to live a couple of months after the transplant. In one of the patients, researchers noted evidence of a pig virus in the man’s heart that had not been identified before the surgery and could be a possible explanation for his heart failure.
So far, so good
Slayman and his medical team ultimately decided to pursue the surgery—and the risk paid off. When the pig organ started producing urine at the end of the four-hour surgery, the entire operating room erupted in applause.
Slayman is currently receiving an infusion of immunosuppressant drugs to prevent the kidney from being rejected, while his doctors monitor the kidney’s function with frequent ultrasounds. Slayman is reported to be “recovering well” at Massachusetts General Hospital and is expected to be discharged within the next several days.