How Excessive Regulation Helped Ignite COVID-19's Rampant Spread
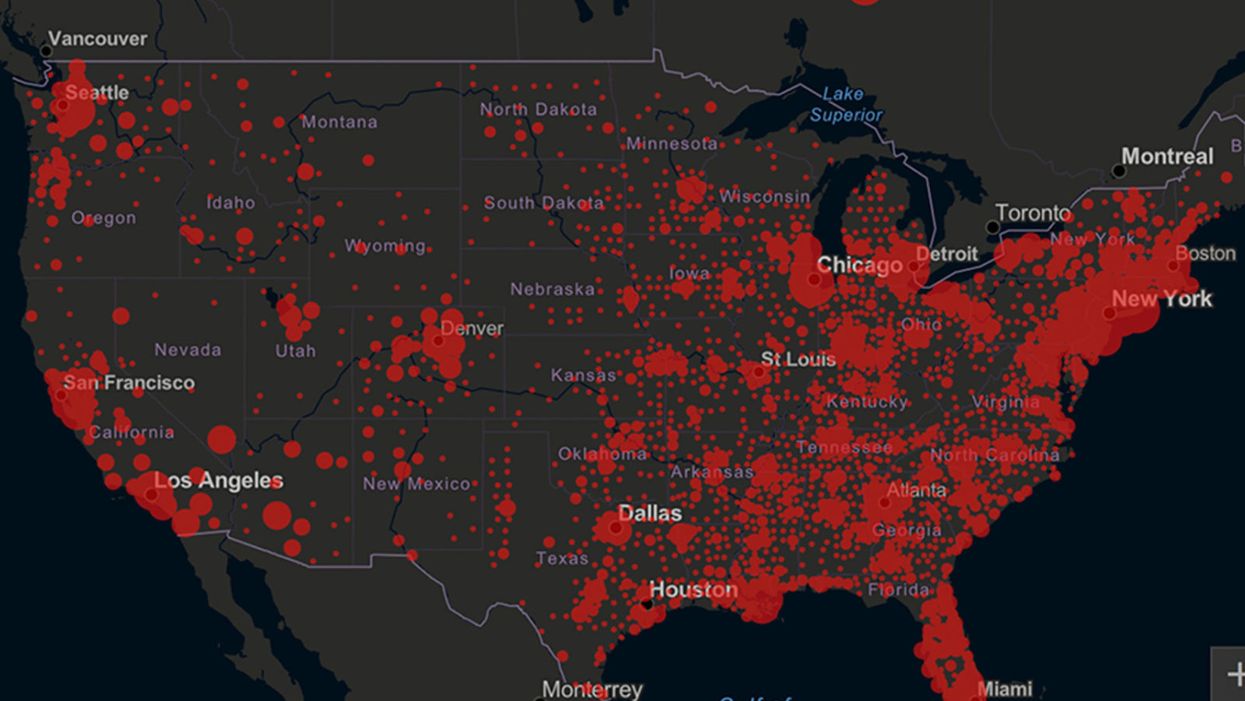
Screenshot of an interactive map of coronavirus cases across the United States, current as of 1:45 p.m. Pacific time on Tuesday, March 31st. Full map accessible at https://coronavirus.jhu.edu/map.html
When historians of the future look back at the 2020 pandemic, the heroic work of Helen Y. Chu, a flu researcher at the University of Washington, will be worthy of recognition.
Chu's team bravely defied the order and conducted the testing anyway.
In late January, Chu was testing nasal swabs for the Seattle Flu Study to monitor influenza spread when she learned of the first case of COVID-19 in Washington state. She deemed it a pressing public health matter to document if and how the illness was spreading locally, so that early containment efforts could succeed. So she sought regulatory approval to adapt the Flu Study to test for the coronavirus, but the federal government denied the request because the original project was funded to study only influenza.
Aware of the urgency, Chu's team bravely defied the order and conducted the testing anyway. Soon they identified a local case in a teenager without any travel history, followed by others. Still, the government tried to shutter their efforts until the outbreak grew dangerous enough to command attention.
Needless testing delays, prompted by excessive regulatory interference, eliminated any chances of curbing the pandemic at its initial stages. Even after Chu went out on a limb to sound alarms, a heavy-handed bureaucracy crushed the nation's ability to roll out early and widespread testing across the country. The Centers for Disease Control and Prevention infamously blundered its own test, while also impeding state and private labs from coming on board, fueling a massive shortage.
The long holdup created "a backlog of testing that needed to be done," says Amesh Adalja, an infectious disease specialist who is a senior scholar at the Johns Hopkins University Center for Health Security.
In a public health crisis, "the ideal situation" would allow the government's test to be "supplanted by private laboratories" without such "a lag in that transition," Adalja says. Only after the eventual release of CDC's test could private industry "begin in earnest" to develop its own versions under the Food and Drug Administration's emergency use authorization.
In a statement, CDC acknowledged that "this process has not gone as smoothly as we would have liked, but there is currently no backlog for testing at CDC."
Now, universities and corporations are in a race against time, playing catch up as the virus continues its relentless spread, also afflicting many health care workers on the front lines.
"Home-testing accessibility is key to preventing further spread of the COVID-19 pandemic."
Hospitals are attempting to add the novel coronavirus to the testing panel of their existent diagnostic machines, which would reduce the results processing time from 48 hours to as little as four hours. Meanwhile, at least four companies announced plans to deliver at-home collection tests to help meet the demand – before a startling injunction by the FDA halted their plans.
Everlywell, an Austin, Texas-based digital health company, had been set to launch online sales of at-home collection kits directly to consumers last week. Scaling up in a matter of days to an initial supply of 30,000 tests, Everlywell collaborated with multiple laboratories where consumers could ship their nasal swab samples overnight, projecting capacity to screen a quarter-million individuals on a weekly basis, says Frank Ong, chief medical and scientific officer.
Secure digital results would have been available online within 48 hours of a sample's arrival at the lab, as well as a telehealth consultation with an independent, board-certified doctor if someone tested positive, for an inclusive $135 cost. The test has a less than 3 percent false-negative rate, Ong says, and in the event of an inadequate self-swab, the lab would not report a conclusive finding. "Home-testing accessibility," he says, "is key to preventing further spread of the COVID-19 pandemic."
But on March 20, the FDA announced restrictions on home collection tests due to concerns about accuracy. The agency did note "the public health value in expanding the availability of COVID-19 testing through safe and accurate tests that may include home collection," while adding that "we are actively working with test developers in this space."
After the restrictions were announced, Everlywell decided to allocate its initial supply of COVID-19 collection kits to hospitals, clinics, nursing homes, and other qualifying health care companies that can commit to no-cost screening of frontline workers and high-risk symptomatic patients. For now, no consumers can order a home-collection test.
"Losing two months is close to disastrous, and that's what we did."
Currently, the U.S. has ramped up to testing an estimated 100,000 people a day, according to Stat News. But 150,000 or more Americans should be tested every day, says Ashish Jha, professor and director of the Harvard Global Health Institute. Due to the dearth of tests, many sick people who suspect they are infected still cannot get confirmation unless they need to be hospitalized.
To give a concrete sense of how far behind we are in testing, consider Palm Beach County, Fla. The state's only drive-thru test center just opened there, requiring an appointment. The center aims to test 750 people per day, but more than 330,000 people have already called to try to book a slot.
"This is such a rapidly moving infection that losing a few days is bad, and losing a couple of weeks is terrible," says Jha, a practicing general internist. "Losing two months is close to disastrous, and that's what we did."
At this point, it will take a long time to fully ramp up. "We are blindfolded," he adds, "and I'd like to take the blindfolds off so we can fight this battle with our eyes wide open."
Better late than never: Yesterday, FDA Commissioner Stephen Hahn said in a statement that the agency has worked with more than 230 test developers and has approved 20 tests since January. An especially notable one was authorized last Friday – 67 days since the country's first known case in Washington state. It's a rapid point-of-care test from medical-device firm Abbott that provides positive results in five minutes and negative results in 13 minutes. Abbott will send 50,000 tests a day to urgent care settings. The first tests are expected to ship tomorrow.
Podcast: Wellness chatbots and meditation pods with Deepak Chopra
Leaps.org talked with Deepak Chopra about new technologies he's developing for mental health with Jonathan Marcoschamer, CEO of OpenSeed, and others.
Over the last few decades, perhaps no one has impacted healthy lifestyles more than Deepak Chopra. While several of his theories and recommendations have been criticized by prominent members of the scientific community, he has helped bring meditation, yoga and other practices for well-being into the mainstream in ways that benefit the health of vast numbers of people every day. His work has led many to accept new ways of thinking about alternative medicine, the power of mind over body, and the malleability of the aging process.
His impact is such that it's been observed our culture no longer recognizes him as a human being but as a pervasive symbol of new-agey personal health and spiritual growth. Last week, I had a chance to confirm that Chopra is, in fact, a human being – and deserving of his icon status – when I talked with him for the Leaps.org podcast. He relayed ideas that were wise and ancient, yet highly relevant to our world today, with the fluidity and ease of someone discussing the weather. Showing no signs of slowing down at age 76, he described his prolific work, including the publication of two books in the past year and a range of technologies he’s developing, including a meditation app, meditation pods for the workplace, and a chatbot for mental health called Piwi.
Take a listen and get inspired to do some meditation and deep thinking on the future of health. As Chopra told me, “If you don’t have time to meditate once per day, you probably need to meditate twice per day.”
Highlights:
2:10: Chopra talks about meditation broadly and meditation pods, including the ones made by OpenSeed for meditation in the workplace.
6:10: The drawbacks of quick fixes like drugs for mental health.
10:30: The benefits of group meditation versus individual meditation.
14:35: What is a "metahuman" and how to become one.
19:40: The difference between the conditioned mind and the mind that's infinitely creative.
22:48: How Chopra's views of free will differ from the views of many neuroscientists.
28:04: Thinking Fast and Slow, and the role of intuition.
31:20: Athletic and creative geniuses.
32:43: The nature of fundamental truth.
34:00: Meditation for kids.
37:12: Never alone.Love and how AI chatbots can support mental health.
42:30: Extending lifespan, gene editing and lifestyle.
46:05: Chopra's mentor in living a long good life (and my mentor).
47:45: The power of yoga.
Links:
- OpenSeed meditation pods for people to meditate at work (Chopra is an advisor to OpenSeed).
- Chopra's book from 2021, Metahuman: Unleash Your Infinite Potential
- Chopra's book from 2022, Abundance: The Inner Path to Wealth
- NeverAlone.Love, Chopra's collaboration of businesses, policy makers, mental health professionals and others to raise awareness about mental health, advance scientific research and "create a global technology platform to democratize access to resources."
- The Piwi chatbot for mental health
- The Chopra Meditation & Well-Being App for people of all ages
- Only 1.6 percent of U.S. children meditate, according to the National Center for Complementary and Integrative Health
Podcast: The Friday Five weekly roundup in health research
Scientists have designed a phone app that could alert consumers to high levels of cancer-causing chemicals, Yale researchers revive organs in dead pigs, and more in this week's Friday Five.
The Friday Five covers five stories in health research that you may have missed this week. There are plenty of controversies and troubling ethical issues in science – and we get into many of them in our online magazine – but this news roundup focuses on scientific creativity and progress to give you a therapeutic dose of inspiration headed into the weekend.
Listen to the Episode
Listen on Apple | Listen on Spotify | Listen on Stitcher | Listen on Amazon | Listen on Google
Covered in this week's Friday Five:
- A new blood test for cancer
- Patches of bacteria can use your sweat to power electronic devices
- Researchers revive organs of dead pigs
- Phone apps detects cancer-causing chemicals in foods
- Stem cells generate "synthetic placentas" in mice
Plus, an honorable mention for early research involving vitamin K and Alzheimer's

