Why we need to get serious about ending aging

With the population of older people projected to grow dramatically, and the cost of healthcare with it, the future welfare of the country may depend on solving aging, writes philosopher Ingemar Patrick Linden.
It is widely acknowledged that even a small advance in anti-aging science could yield benefits in terms of healthy years that the traditional paradigm of targeting specific diseases is not likely to produce. A more youthful population would also be less vulnerable to epidemics. Approximately 93 percent of all COVID-19 deaths reported in the U.S. occurred among those aged 50 or older. The potential economic benefits would be tremendous. A more youthful population would consume less medical resources and be able to work longer. A recent study published in Nature estimates that a slowdown in aging that increases life expectancy by one year would save $38 trillion per year for the U.S. alone.
A societal effort to understand, slow down, arrest or even reverse aging of at least the size of our response to COVID-19 would therefore be a rational commitment. In fact, given that America’s older population is projected to grow dramatically, and the cost of healthcare with it, it is not an overstatement to say that the future welfare of the country may depend on solving aging.
This year, the kingdom of Saudi Arabia has announced that it will spend up to 1 billion dollars per year on science with the potential to slow down the aging process. We have also seen important investments from billionaires like Google co-founder Larry Page, Amazon founder Jeff Bezos, business magnate Larry Ellison, and PayPal co-founder Peter Thiel.
The U.S. government, however, is lagging: The National Institutes of Health spent less than one percent of its $43 billion budget for the fiscal year of 2021 on the National Institute on Aging’s Division of Aging Biology. When you visit the division’s webpage you find that their mission statement carefully omits any mention of the possibility of slowing down the aging process.
There is a lack of political will and leadership on the issue, and the idea that we should seek to do something about aging is generally met with a great deal of suspicion and trepidation. In a large representative study conducted by the Pew Research Center in 2013, only 38% of the respondents said that they would want a treatment that could slow the aging process and allow them to live at least 120 years. Apparently, most people prefer, or at least do not mind, to age and die within a natural lifespan. This result has been confirmed by smaller studies and it is, I think, surprising. Are we not supposed to live in a youth-culture? Are people not supposed to want to stay young and alive forever? Is self-preservation not the strong drive we have always assumed it to be?
We are inundated and saturated with an ideology of death-acceptance.
In my book, The Case against Death, I suggest that we have been culturally conditioned to think that it is virtuous to accept aging and death. We are taught to believe that although aging and death seem gruesome, they are what is best for us, all things considered. This is what we are supposed to think, and the majority accept it. I call this the Wise View because death acceptance has been the dominant view of philosophers since the beginning. Socrates compared our earthly life to an illness and a prison and described death as a healer and a liberator. The Buddha taught that life is suffering and that the way to escape suffering is to end the cycle of birth, death and rebirth. Stoic philosophers from Zeno to Marcus Aurelius believed that everything that happens in accordance with nature is good, and that therefore we should not only accept death but welcome it as an aspect of a perfect totality.
Epicureans agreed with these rival schools and famously argued that death cannot harm us because where we are, death is not, and where death is we are not. We cannot be harmed if we are not, so death is harmless. The simple view that death actually can harm us greatly is one of the least philosophical views one can hold.

In The Case Against Death, philosopher Ingemar Patrick Linden argues that we frown on using science to prolong healthy life only because we're culturally conditioned to think that way.
Many of the stories we tell promote the Wise View. One of the earliest known pieces of literature, the Epic of Gilgamesh, follows Gilgamesh on a quest for eternal life ending with the wisdom that death is the destiny of man. Today we learn about the tedium of immortality from the children’s book Tuck Everlasting by Natalie Babbitt, and we are warned about the vice of wanting to resist death in other books and films such as J.K Rowling’s Harry Potter, where Voldemort must kill Harry as a step towards his own immortality; C.S. Lewis’ The Chronicles of Narnia where the White Witch has gained immortal youth and madness in equal measures; J.R.R. Tolkien’s Lord of the Rings trilogy where the ring extends the wearer’s life but can also destroy them, as exemplified by the creep Gollum; and Doctor Strange where life extension is the one magical power that is taboo. In Star Wars, Yoda, a stereotype of the sage, teaches us the wisdom handed down by philosophers and prophets: “Death is a natural part of life. Rejoice for those around you who transform into the Force. Mourn them do not. Miss them do not.”
We are inundated and saturated with an ideology of death-acceptance. Can the dear reader name one single story where the hero is pursuing anti-aging, longevity or immortality and the villain tries to stop her?
The Wise View resonates with us partly because we think that there is nothing we can do about aging and death, so we do not want to wish for what we cannot have. Youth and immortality are sour grapes to us. Believing that death is, all things considered, not such a bad thing, protects us from experiencing our aging and approaching death as a gruesome tragedy. This need to escape the thought that we are heading towards a personal catastrophe explains why many are so quick to accept arguments against radical life extension, despite their often glaring weaknesses.
One of the most common objections to radical life extension is that aging and death are natural. The problem with this argument is that many things that are natural are very bad, such as cancer, and other things that are not natural are very good, such as a cure for cancer. Why are we so sure that cancer is bad? Because we assume that it is bad to die. Indeed, nothing is more natural than wanting to live. We seem to need philosophers and story tellers to talk us out of it and, in the words of a distinguished bioethicist, “instruct and somewhat moderate our lust for life.”
Another standard objection is that we need a deadline, and that without death we could postpone every action forever. “Death brings urgency and seriousness to life,” say proponents of this view, but there are several problems with this argument. Even if our lives were endless, there would still be many things we would have to do at a certain time, and that could not be redone, for example, saving our planet from being destroyed, or becoming the first person on Venus. And if we prefer pleasant endless lives over unpleasant endless ones, we will have to exercise, eat right, keep our word, develop our talents, show up for time at work, pay our taxes by the due date, remember birthdays, and so on.
The Wise View provides us with a feel-good bromide for the anxiety created by the foreknowledge of our decay and death by telling us that these are not evils, but blessings in disguise. Once perhaps an innocuous delusion, today the view stands in the way of a necessary societal commitment to research that can prolong our healthy life.
Besides, even if we succeeded in ending aging, we would still die from other causes. Given the rate of accidental deaths we would be fortunate to live to age 2000 all things equal. So even if, contrary to what I have argued, we do need a deadline, we can still argue that the natural lifespan that we now labor under is inhuman and that it forces each human to limit her ambitions and to become only a fragment of all that she that could have been. Our tight time constraint imposes tragic choices and inflated opportunity costs. Death does not make life matter; it makes time matter.
The perhaps most awful argument against radical life extension is grounded in a pessimism that holds life in such little regard that it says that best of all is never to have born. This view was expressed by Ecclesiastes in the Hebrew Bible, by Sophocles and several other ancient Greeks, by the German philosopher Arthur Schopenhauer, and recently by, among others, the South African philosopher David Benatar who argues that it is wrong to bring children into the world and that we should euthanize all sentient life. Pessimism, one suspects, largely appeals to some for reasons having to do with personal temperament, but insofar as it is built on factual beliefs, they can be addressed by providing a less negatively biased understanding of the world, by pointing out that curing aging would decrease the badness that they are so hypersensitive to, and by reminding them that if life really becomes unbearable, they are free to quit at any time. Other means of persuasion could include recommending sleep, exercise and taking long brisk walks in nature.
The Wise View provides us with a feel-good bromide for the anxiety created by the foreknowledge of our decay and death by telling us that these are not evils, but blessings in disguise. Once perhaps an innocuous delusion, today the view stands in the way of a necessary societal commitment to research that can prolong our healthy life. We need abandon it and openly admit that aging is a scourge that deserves to be fought with the combined energies equaling those expended on fighting COVID-19, Alzheimer’s disease, cancer, stroke and all the other illnesses for which aging is the greatest risk factor. The fight to end aging transcends ordinary political boundaries and is therefore the kind of grand unifying enterprise that could re-energize a society suffering from divisiveness and the sense of a lack of a common purpose. It is hard to imagine a more worthwhile cause.
Jamie Rettinger with his now fiance Amie Purnel-Davis, who helped him through the clinical trial.
Jamie Rettinger was still in his thirties when he first noticed a tiny streak of brown running through the thumbnail of his right hand. It slowly grew wider and the skin underneath began to deteriorate before he went to a local dermatologist in 2013. The doctor thought it was a wart and tried scooping it out, treating the affected area for three years before finally removing the nail bed and sending it off to a pathology lab for analysis.
"I have some bad news for you; what we removed was a five-millimeter melanoma, a cancerous tumor that often spreads," Jamie recalls being told on his return visit. "I'd never heard of cancer coming through a thumbnail," he says. None of his doctors had ever mentioned it either. "I just thought I was being treated for a wart." But nothing was healing and it continued to bleed.
A few months later a surgeon amputated the top half of his thumb. Lymph node biopsy tested negative for spread of the cancer and when the bandages finally came off, Jamie thought his medical issues were resolved.
Melanoma is the deadliest form of skin cancer. About 85,000 people are diagnosed with it each year in the U.S. and more than 8,000 die of the cancer when it spreads to other parts of the body, according to the Centers for Disease Control and Prevention (CDC).
There are two peaks in diagnosis of melanoma; one is in younger women ages 30-40 and often is tied to past use of tanning beds; the second is older men 60+ and is related to outdoor activity from farming to sports. Light-skinned people have a twenty-times greater risk of melanoma than do people with dark skin.
"When I graduated from medical school, in 2005, melanoma was a death sentence" --Diwakar Davar.
Jamie had a follow up PET scan about six months after his surgery. A suspicious spot on his lung led to a biopsy that came back positive for melanoma. The cancer had spread. Treatment with a monoclonal antibody (nivolumab/Opdivo®) didn't prove effective and he was referred to the UPMC Hillman Cancer Center in Pittsburgh, a four-hour drive from his home in western Ohio.
An alternative monoclonal antibody treatment brought on such bad side effects, diarrhea as often as 15 times a day, that it took more than a week of hospitalization to stabilize his condition. The only options left were experimental approaches in clinical trials.
Early research
"When I graduated from medical school, in 2005, melanoma was a death sentence" with a cure rate in the single digits, says Diwakar Davar, 39, an oncologist at UPMC Hillman Cancer Center who specializes in skin cancer. That began to change in 2010 with introduction of the first immunotherapies, monoclonal antibodies, to treat cancer. The antibodies attach to PD-1, a receptor on the surface of T cells of the immune system and on cancer cells. Antibody treatment boosted the melanoma cure rate to about 30 percent. The search was on to understand why some people responded to these drugs and others did not.
At the same time, there was a growing understanding of the role that bacteria in the gut, the gut microbiome, plays in helping to train and maintain the function of the body's various immune cells. Perhaps the bacteria also plays a role in shaping the immune response to cancer therapy.
One clue came from genetically identical mice. Animals ordered from different suppliers sometimes responded differently to the experiments being performed. That difference was traced to different compositions of their gut microbiome; transferring the microbiome from one animal to another in a process known as fecal transplant (FMT) could change their responses to disease or treatment.
When researchers looked at humans, they found that the patients who responded well to immunotherapies had a gut microbiome that looked like healthy normal folks, but patients who didn't respond had missing or reduced strains of bacteria.
Davar and his team knew that FMT had a very successful cure rate in treating the gut dysbiosis of Clostridioides difficile, a persistant intestinal infection, and they wondered if a fecal transplant from a patient who had responded well to cancer immunotherapy treatment might improve the cure rate of patients who did not originally respond to immunotherapies for melanoma.
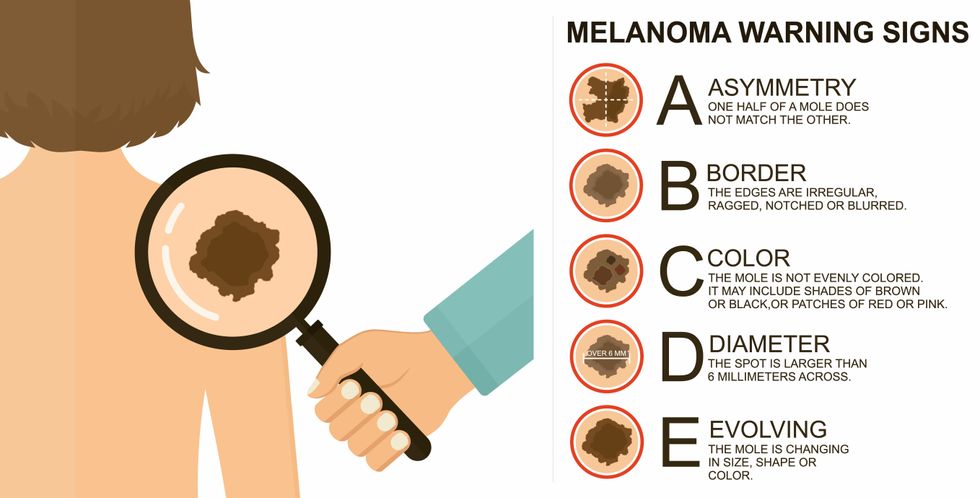
The ABCDE of melanoma detection
Adobe Stock
Clinical trial
"It was pretty weird, I was totally blasted away. Who had thought of this?" Jamie first thought when the hypothesis was explained to him. But Davar's explanation that the procedure might restore some of the beneficial bacterial his gut was lacking, convinced him to try. He quickly signed on in October 2018 to be the first person in the clinical trial.
Fecal donations go through the same safety procedures of screening for and inactivating diseases that are used in processing blood donations to make them safe for transfusion. The procedure itself uses a standard hollow colonoscope designed to screen for colon cancer and remove polyps. The transplant is inserted through the center of the flexible tube.
Most patients are sedated for procedures that use a colonoscope but Jamie doesn't respond to those drugs: "You can't knock me out. I was watching them on the TV going up my own butt. It was kind of unreal at that point," he says. "There were about twelve people in there watching because no one had seen this done before."
A test two weeks after the procedure showed that the FMT had engrafted and the once-missing bacteria were thriving in his gut. More importantly, his body was responding to another monoclonal antibody (pembrolizumab/Keytruda®) and signs of melanoma began to shrink. Every three months he made the four-hour drive from home to Pittsburgh for six rounds of treatment with the antibody drug.
"We were very, very lucky that the first patient had a great response," says Davar. "It allowed us to believe that even though we failed with the next six, we were on the right track. We just needed to tweak the [fecal] cocktail a little better" and enroll patients in the study who had less aggressive tumor growth and were likely to live long enough to complete the extensive rounds of therapy. Six of 15 patients responded positively in the pilot clinical trial that was published in the journal Science.
Davar believes they are beginning to understand the biological mechanisms of why some patients initially do not respond to immunotherapy but later can with a FMT. It is tied to the background level of inflammation produced by the interaction between the microbiome and the immune system. That paper is not yet published.
Surviving cancer
It has been almost a year since the last in his series of cancer treatments and Jamie has no measurable disease. He is cautiously optimistic that his cancer is not simply in remission but is gone for good. "I'm still scared every time I get my scans, because you don't know whether it is going to come back or not. And to realize that it is something that is totally out of my control."
"It was hard for me to regain trust" after being misdiagnosed and mistreated by several doctors he says. But his experience at Hillman helped to restore that trust "because they were interested in me, not just fixing the problem."
He is grateful for the support provided by family and friends over the last eight years. After a pause and a sigh, the ruggedly built 47-year-old says, "If everyone else was dead in my family, I probably wouldn't have been able to do it."
"I never hesitated to ask a question and I never hesitated to get a second opinion." But Jamie acknowledges the experience has made him more aware of the need for regular preventive medical care and a primary care physician. That person might have caught his melanoma at an earlier stage when it was easier to treat.
Davar continues to work on clinical studies to optimize this treatment approach. Perhaps down the road, screening the microbiome will be standard for melanoma and other cancers prior to using immunotherapies, and the FMT will be as simple as swallowing a handful of freeze-dried capsules off the shelf rather than through a colonoscopy. Earlier this year, the Food and Drug Administration approved the first oral fecal microbiota product for C. difficile, hopefully paving the way for more.
An older version of this hit article was first published on May 18, 2021
All organisms can repair damaged tissue, but none do it better than salamanders and newts. A surprising area of science could tell us how they manage this feat - and perhaps even help us develop a similar ability.
All organisms have the capacity to repair or regenerate tissue damage. None can do it better than salamanders or newts, which can regenerate an entire severed limb.
That feat has amazed and delighted man from the dawn of time and led to endless attempts to understand how it happens – and whether we can control it for our own purposes. An exciting new clue toward that understanding has come from a surprising source: research on the decline of cells, called cellular senescence.
Senescence is the last stage in the life of a cell. Whereas some cells simply break up or wither and die off, others transition into a zombie-like state where they can no longer divide. In this liminal phase, the cell still pumps out many different molecules that can affect its neighbors and cause low grade inflammation. Senescence is associated with many of the declining biological functions that characterize aging, such as inflammation and genomic instability.
Oddly enough, newts are one of the few species that do not accumulate senescent cells as they age, according to research over several years by Maximina Yun. A research group leader at the Center for Regenerative Therapies Dresden and the Max Planck Institute of Molecular and Cell Biology and Genetics, in Dresden, Germany, Yun discovered that senescent cells were induced at some stages of regeneration of the salamander limb, “and then, as the regeneration progresses, they disappeared, they were eliminated by the immune system,” she says. “They were present at particular times and then they disappeared.”
Senescent cells added to the edges of the wound helped the healthy muscle cells to “dedifferentiate,” essentially turning back the developmental clock of those cells into more primitive states.
Previous research on senescence in aging had suggested, logically enough, that applying those cells to the stump of a newly severed salamander limb would slow or even stop its regeneration. But Yun stood that idea on its head. She theorized that senescent cells might also play a role in newt limb regeneration, and she tested it by both adding and removing senescent cells from her animals. It turned out she was right, as the newt limbs grew back faster than normal when more senescent cells were included.
Senescent cells added to the edges of the wound helped the healthy muscle cells to “dedifferentiate,” essentially turning back the developmental clock of those cells into more primitive states, which could then be turned into progenitors, a cell type in between stem cells and specialized cells, needed to regrow the muscle tissue of the missing limb. “We think that this ability to dedifferentiate is intrinsically a big part of why salamanders can regenerate all these very complex structures, which other organisms cannot,” she explains.
Yun sees regeneration as a two part problem. First, the cells must be able to sense that their neighbors from the lost limb are not there anymore. Second, they need to be able to produce the intermediary progenitors for regeneration, , to form what is missing. “Molecularly, that must be encoded like a 3D map,” she says, otherwise the new tissue might grow back as a blob, or liver, or fin instead of a limb.
Wound healing
Another recent study, this time at the Mayo Clinic, provides evidence supporting the role of senescent cells in regeneration. Looking closely at molecules that send information between cells in the wound of a mouse, the researchers found that senescent cells appeared near the start of the healing process and then disappeared as healing progressed. In contrast, persistent senescent cells were the hallmark of a chronic wound that did not heal properly. The function and significance of senescence cells depended on both the timing and the context of their environment.
The paper suggests that senescent cells are not all the same. That has become clearer as researchers have been able to identify protein markers on the surface of some senescent cells. The patterns of these proteins differ for some senescent cells compared to others. In biology, such physical differences suggest functional differences, so it is becoming increasingly likely there are subsets of senescent cells with differing functions that have not yet been identified.
There are disagreements within the research community as to whether newts have acquired their regenerative capacity through a unique evolutionary change, or if other animals, including humans, retain this capacity buried somewhere in their genes.
Scientists initially thought that senescent cells couldn’t play a role in regeneration because they could no longer reproduce, says Anthony Atala, a practicing surgeon and bioengineer who leads the Wake Forest Institute for Regenerative Medicine in North Carolina. But Yun’s study points in the other direction. “What this paper shows clearly is that these cells have the potential to be involved in tissue regeneration [in newts]. The question becomes, will these cells be able to do the same in humans.”
As our knowledge of senescent cells increases, Atala thinks we need to embrace a new analogy to help understand them: humans in retirement. They “have acquired a lot of wisdom throughout their whole life and they can help younger people and mentor them to grow to their full potential. We're seeing the same thing with these cells,” he says. They are no longer putting energy into their own reproduction, but the signaling molecules they secrete “can help other cells around them to regenerate.”
There are disagreements within the research community as to whether newts have acquired their regenerative capacity through a unique evolutionary change, or if other animals, including humans, retain this capacity buried somewhere in their genes. If so, it seems that our genes are unable to express this ability, perhaps as part of a tradeoff in acquiring other traits. It is a fertile area of research.
Dedifferentiation is likely to become an important process in the field of regenerative medicine. One extreme example: a lab has been able to turn back the clock and reprogram adult male skin cells into female eggs, a potential milestone in reproductive health. It will be more difficult to control just how far back one wishes to go in the cell's dedifferentiation – part way or all the way back into a stem cell – and then direct it down a different developmental pathway. Yun is optimistic we can learn these tricks from newts.
Senolytics
A growing field of research is using drugs called senolytics to remove senescent cells and slow or even reverse disease of aging.
“Senolytics are great, but senolytics target different types of senescence,” Yun says. “If senescent cells have positive effects in the context of regeneration, of wound healing, then maybe at the beginning of the regeneration process, you may not want to take them out for a little while.”
“If you look at pretty much all biological systems, too little or too much of something can be bad, you have to be in that central zone” and at the proper time, says Atala. “That's true for proteins, sugars, and the drugs that you take. I think the same thing is true for these cells. Why would they be different?”
Our growing understanding that senescence is not a single thing but a variety of things likely means that effective senolytic drugs will not resemble a single sledge hammer but more a carefully manipulated scalpel where some types of senescent cells are removed while others are added. Combinations and timing could be crucial, meaning the difference between regenerating healthy tissue, a scar, or worse.

