Tiny, tough “water bears” may help bring new vaccines and medicines to sub-Saharan Africa
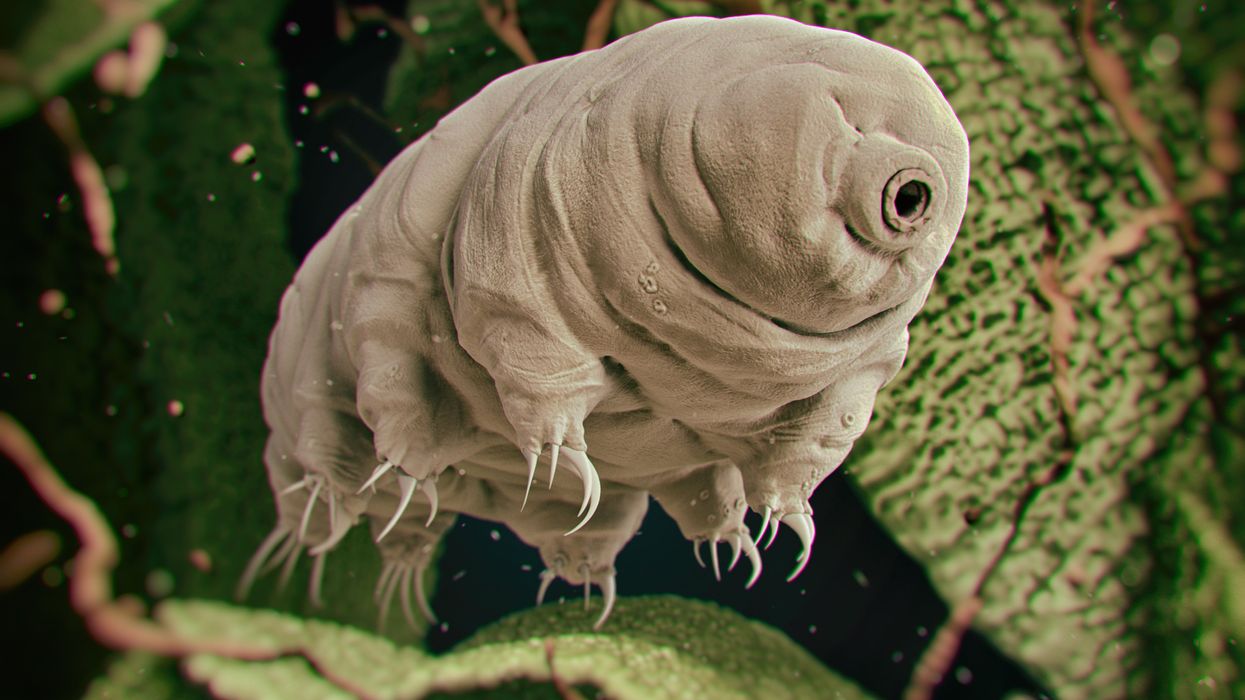
Tardigrades can completely dehydrate and later rehydrate themselves, a survival trick that scientists are harnessing to preserve medicines in hot temperatures.
Microscopic tardigrades, widely considered to be some of the toughest animals on earth, can survive for decades without oxygen or water and are thought to have lived through a crash-landing on the moon. Also known as water bears, they survive by fully dehydrating and later rehydrating themselves – a feat only a few animals can accomplish. Now scientists are harnessing tardigrades’ talents to make medicines that can be dried and stored at ambient temperatures and later rehydrated for use—instead of being kept refrigerated or frozen.
Many biologics—pharmaceutical products made by using living cells or synthesized from biological sources—require refrigeration, which isn’t always available in many remote locales or places with unreliable electricity. These products include mRNA and other vaccines, monoclonal antibodies and immuno-therapies for cancer, rheumatoid arthritis and other conditions. Cooling is also needed for medicines for blood clotting disorders like hemophilia and for trauma patients.
Formulating biologics to withstand drying and hot temperatures has been the holy grail for pharmaceutical researchers for decades. It’s a hard feat to manage. “Biologic pharmaceuticals are highly efficacious, but many are inherently unstable,” says Thomas Boothby, assistant professor of molecular biology at University of Wyoming. Therefore, during storage and shipping, they must be refrigerated at 2 to 8 degrees Celsius (35 to 46 degrees Fahrenheit). Some must be frozen, typically at -20 degrees Celsius, but sometimes as low -90 degrees Celsius as was the case with the Pfizer Covid vaccine.
For Covid, fewer than 73 percent of the global population received even one dose. The need for refrigerated or frozen handling was partially to blame.
The costly cold chain
The logistics network that ensures those temperature requirements are met from production to administration is called the cold chain. This cold chain network is often unreliable or entirely lacking in remote, rural areas in developing nations that have malfunctioning electrical grids. “Almost all routine vaccines require a cold chain,” says Christopher Fox, senior vice president of formulations at the Access to Advanced Health Institute. But when the power goes out, so does refrigeration, putting refrigerated or frozen medical products at risk. Consequently, the mRNA vaccines developed for Covid-19 and other conditions, as well as more traditional vaccines for cholera, tetanus and other diseases, often can’t be delivered to the most remote parts of the world.
To understand the scope of the challenge, consider this: In the U.S., more than 984 million doses of Covid-19 vaccine have been distributed so far. Each one needed refrigeration that, even in the U.S., proved challenging. Now extrapolate to all vaccines and the entire world. For Covid, fewer than 73 percent of the global population received even one dose. The need for refrigerated or frozen handling was partially to blame.
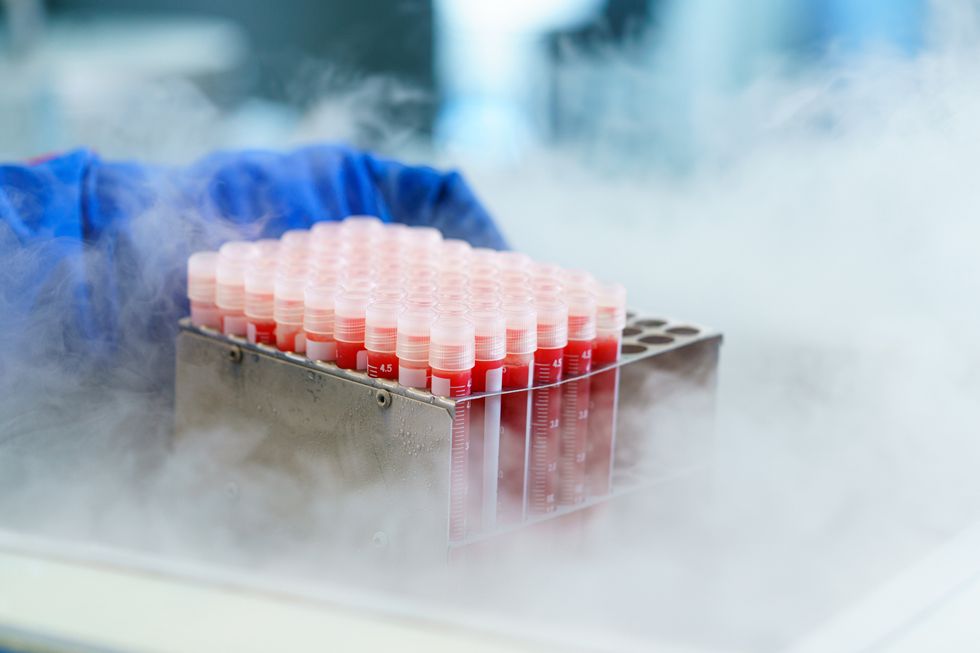
Globally, the cold chain packaging market is valued at over $15 billion and is expected to exceed $60 billion by 2033.
Adobe Stock
Freeze-drying, also called lyophilization, which is common for many vaccines, isn’t always an option. Many freeze-dried vaccines still need refrigeration, and even medicines approved for storage at ambient temperatures break down in the heat of sub-Saharan Africa. “Even in a freeze-dried state, biologics often will undergo partial rehydration and dehydration, which can be extremely damaging,” Boothby explains.
The cold chain is also very expensive to maintain. The global pharmaceutical cold chain packaging market is valued at more than $15 billion, and is expected to exceed $60 billion by 2033, according to a report by Future Market Insights. This cost is only expected to grow. According to the consulting company Accenture, the number of medicines that require the cold chain are expected to grow by 48 percent, compared to only 21 percent for non-cold-chain therapies.
Tardigrades to the rescue
Tardigrades are only about a millimeter long – with four legs and claws, and they lumber around like bears, thus their nickname – but could provide a big solution. “Tardigrades are unique in the animal kingdom, in that they’re able to survive a vast array of environmental insults,” says Boothby, the Wyoming professor. “They can be dried out, frozen, heated past the boiling point of water and irradiated at levels that are thousands of times more than you or I could survive.” So, his team is gradually unlocking tardigrades’ survival secrets and applying them to biologic pharmaceuticals to make them withstand both extreme heat and desiccation without losing efficacy.
Boothby’s team is focusing on blood clotting factor VIII, which, as the name implies, causes blood to clot. Currently, Boothby is concentrating on the so-called cytoplasmic abundant heat soluble (CAHS) protein family, which is found only in tardigrades, protecting them when they dry out. “We showed we can desiccate a biologic (blood clotting factor VIII, a key clotting component) in the presence of tardigrade proteins,” he says—without losing any of its effectiveness.
The researchers mixed the tardigrade protein with the blood clotting factor and then dried and rehydrated that substance six times without damaging the latter. This suggests that biologics protected with tardigrade proteins can withstand real-world fluctuations in humidity.
Furthermore, Boothby’s team found that when the blood clotting factor was dried and stabilized with tardigrade proteins, it retained its efficacy at temperatures as high as 95 degrees Celsius. That’s over 200 degrees Fahrenheit, much hotter than the 58 degrees Celsius that the World Meteorological Organization lists as the hottest recorded air temperature on earth. In contrast, without the protein, the blood clotting factor degraded significantly. The team published their findings in the journal Nature in March.
Although tardigrades rarely live more than 2.5 years, they have survived in a desiccated state for up to two decades, according to Animal Diversity Web. This suggests that tardigrades’ CAHS protein can protect biologic pharmaceuticals nearly indefinitely without refrigeration or freezing, which makes it significantly easier to deliver them in locations where refrigeration is unreliable or doesn’t exist.
The tricks of the tardigrades
Besides the CAHS proteins, tardigrades rely on a type of sugar called trehalose and some other protectants. So, rather than drying up, their cells solidify into rigid, glass-like structures. As that happens, viscosity between cells increases, thereby slowing their biological functions so much that they all but stop.
Now Boothby is combining CAHS D, one of the proteins in the CAHS family, with trehalose. He found that CAHS D and trehalose each protected proteins through repeated drying and rehydrating cycles. They also work synergistically, which means that together they might stabilize biologics under a variety of dry storage conditions.
“We’re finding the protective effect is not just additive but actually is synergistic,” he says. “We’re keen to see if something like that also holds true with different protein combinations.” If so, combinations could possibly protect against a variety of conditions.
Commercialization outlook
Before any stabilization technology for biologics can be commercialized, it first must be approved by the appropriate regulators. In the U.S., that’s the U.S. Food and Drug Administration. Developing a new formulation would require clinical testing and vast numbers of participants. So existing vaccines and biologics likely won’t be re-formulated for dry storage. “Many were developed decades ago,” says Fox. “They‘re not going to be reformulated into thermo-stable vaccines overnight,” if ever, he predicts.
Extending stability outside the cold chain, even for a few days, can have profound health, environmental and economic benefits.
Instead, this technology is most likely to be used for the new products and formulations that are just being created. New and improved vaccines will be the first to benefit. Good candidates include the plethora of mRNA vaccines, as well as biologic pharmaceuticals for neglected diseases that affect parts of the world where reliable cold chain is difficult to maintain, Boothby says. Some examples include new, more effective vaccines for malaria and for pathogenic Escherichia coli, which causes diarrhea.
Tallying up the benefits
Extending stability outside the cold chain, even for a few days, can have profound health, environmental and economic benefits. For instance, MenAfriVac, a meningitis vaccine (without tardigrade proteins) developed for sub-Saharan Africa, can be stored at up to 40 degrees Celsius for four days before administration. “If you have a few days where you don’t need to maintain the cold chain, it’s easier to transport vaccines to remote areas,” Fox says, where refrigeration does not exist or is not reliable.
Better health is an obvious benefit. MenAfriVac reduced suspected meningitis cases by 57 percent in the overall population and more than 99 percent among vaccinated individuals.
Lower healthcare costs are another benefit. One study done in Togo found that the cold chain-related costs increased the per dose vaccine price up to 11-fold. The ability to ship the vaccines using the usual cold chain, but transporting them at ambient temperatures for the final few days cut the cost in half.
There are environmental benefits, too, such as reducing fuel consumption and greenhouse gas emissions. Cold chain transports consume 20 percent more fuel than non-cold chain shipping, due to refrigeration equipment, according to the International Trade Administration.
A study by researchers at Johns Hopkins University compared the greenhouse gas emissions of the new, oral Vaxart COVID-19 vaccine (which doesn’t require refrigeration) with four intramuscular vaccines (which require refrigeration or freezing). While the Vaxart vaccine is still in clinical trials, the study found that “up to 82.25 million kilograms of CO2 could be averted by using oral vaccines in the U.S. alone.” That is akin to taking 17,700 vehicles out of service for one year.
Although tardigrades’ protective proteins won’t be a component of biologic pharmaceutics for several years, scientists are proving that this approach is viable. They are hopeful that a day will come when vaccines and biologics can be delivered anywhere in the world without needing refrigerators or freezers en route.
A New Test Aims to Objectively Measure Pain. It Could Help Legitimate Sufferers Access the Meds They Need.
Sickle cell patient Bridgett Willkie found herself being labeled an addict when she sought an opioid prescription to control her pain.
"That throbbing you feel for the first minute after a door slams on your finger."
This is how Central Florida resident Bridgett Willkie describes the attacks of pain caused by her sickle cell anemia – a genetic blood disorder in which a patient's red blood cells become shaped like sickles and get stuck in blood vessels, thereby obstructing the flow of blood and oxygen.
"I found myself being labeled as an addict and I never was."
Willkie's lifelong battle with the condition has led to avascular necrosis in both of her shoulders, hips, knees and ankles. This means that her bone tissue is dying due to insufficient blood supply (sickle cell anemia is among the medical conditions that can decrease blood flow to one's bones).
"That adds to the pain significantly," she says. "Every time my heart beats, it hurts. And the pain moves. It follows the path of circulation. I liken it to a traffic jam in my veins."
For more than a decade, she received prescriptions for Oxycontin. Then, four years ago, her hematologist – who had been her doctor for 18 years – suffered a fatal heart attack. She says her longtime doctor's replacement lacked experience treating sickle cell patients and was uncomfortable writing her a prescription for opioids. What's more, this new doctor wanted to place her in a drug rehab facility.
"Because I refused to go, he stopped writing my scripts," she says. The ensuing three months were spent at home, detoxing. She describes the pain as unbearable. "Sometimes I just wanted to die."
One of the effects of the opioid epidemic is that many legitimate pain patients have seen their opioids significantly reduced or downright discontinued because of their doctors' fears of over-prescribing addictive medications.
"I found myself being labeled as an addict and I never was...Being treated like a drug-seeking patient is degrading and humiliating," says Willkie, who adds that when she is at the hospital, "it's exhausting arguing with the doctors...You dread them making their rounds because every day they come in talking about weaning you off your meds."
Situations such as these are fraught with tension between patients and doctors, who must remain wary about the risk of over-prescribing powerful and addictive medications. Adding to the complexity is that it can be very difficult to reliably assess a patient's level of physical pain.
However, this difficulty may soon decline, as Indiana University School of Medicine researchers, led by Dr. Alexander B. Niculescu, have reportedly devised a way to objectively assess physical pain by analyzing biomarkers in a patient's blood sample. The results of a study involving more than 300 participants were published earlier this year in the journal Molecular Psychiatry.
Niculescu – who is both a professor of psychiatry and medical neuroscience at the IU School of Medicine – explains that, when someone is in severe physical pain, a blood sample will show biomarkers related to intracellular adhesion and cell-signaling mechanisms. He adds that some of these biomarkers "have prior convergent evidence from animal or human studies for involvement in pain."
Aside from reliably measuring pain severity, Niculescu says blood biomarkers can measure the degree of one's response to treatment and also assess the risk of future recurrences of pain. He believes this new method's greatest benefit, however, might be the ability to identify a number of non-opioid medications that a particular patient is likely to respond to, based on his or her biomarker profile.
Clearly, such a method could be a gamechanger for pain patients and the professionals who treat them. As of yet, health workers have been forced to make crucial decisions based on their clinical impressions of patients; such impressions are invariably subjective. A method that enables people to prove the extent of their pain could remove the stigma that many legitimate pain patients face when seeking to obtain their needed medicine. It would also improve their chances of receiving sufficient treatment.
Niculescu says it's "theoretically possible" that there are some conditions which, despite being severe, might not reveal themselves through his testing method. But he also says that, "even if the same molecular markers that are involved in the pain process are not reflected in the blood, there are other indirect markers that should reflect the distress."
Niculescu expects his testing method will be available to the medical community at large within one to three years.
Willkie says she would welcome a reliable pain assessment method. Well-aware that she is not alone in her plight, she has more than 500 Facebook friends with sickle cell disease, and she says that "all of their opioid meds have been restricted or cut" as a result of the opioid crisis. Some now feel compelled to find their opioids "on the streets." She says she personally has never obtained opioids this way. Instead, she relies on marijuana to mitigate her pain.
Niculescu expects his testing method will be available to the medical community at large within one to three years: "It takes a while for things to translate from a lab setting to a commercial testing arena."
In the meantime, for Willkie and other patients, "we have to convince doctors and nurses that we're in pain."
Some people can eat red meat without negative health consequences, which may be due to variability between people's gut microbes.
In different countries' national dietary guidelines, red meats (beef, pork, and lamb) are often confined to a very small corner. Swedish officials, for example, advise the population to "eat less red and processed meat". Experts in Greece recommend consuming no more than four servings of red meat — not per week, but per month.
"Humans 100% rely on the microbes to digest this food."
Yet somehow, the matter is far from settled. Quibbles over the scientific evidence emerge on a regular basis — as in a recent BMJ article titled, "No need to cut red meat, say new guidelines." News headlines lately have declared that limiting red meat may be "bad advice," while carnivore diet enthusiasts boast about the weight loss and good health they've achieved on an all-meat diet. The wildly successful plant-based burgers? To them, a gimmick. The burger wars are on.
Nutrition science would seem the best place to look for answers on the health effects of specific foods. And on one hand, the science is rather clear: in large populations, people who eat more red meat tend to have more health problems, including cardiovascular disease, colorectal cancer, and other conditions. But this sort of correlational evidence fails to settle the matter once and for all; many who look closely at these studies cite methodological shortcomings and a low certainty of evidence.
Some scientists, meanwhile, are trying to cut through the noise by increasing their focus on the mechanisms: exactly how red meat is digested and the step-by-step of how this affects human health. And curiously, as these lines of evidence emerge, several of them center around gut microbes as active participants in red meat's ultimate effects on human health.
Dr. Stanley Hazen, researcher and medical director of preventive cardiology at Cleveland Clinic, was one of the first to zero in on gut microorganisms as possible contributors to the health effects of red meat. In looking for chemical compounds in the blood that could predict the future development of cardiovascular disease, his lab identified a molecule called trimethylamine-N-oxide (TMAO). Little by little, he and his colleagues began to gather both human and animal evidence that TMAO played a role in causing heart disease.
Naturally, they tried to figure out where the TMAO came from. Hazen says, "We found that animal products, and especially red meat, were a dietary source that, [along with] gut microbes, would generate this product that leads to heart disease development." They observed that the gut microbes were essential for making TMAO out of dietary compounds (like red meat) that contained its precursor, trimethylamine (TMA).
So in linking red meat to cardiovascular disease through TMAO, the surprising conclusion, says Hazen, was that, "Without a doubt, [the microbes] are the most important aspect of the whole pathway."
"I think it's just a matter of time [before] we will have therapeutic interventions that actually target our gut microbes, just like the way we take drugs that lower cholesterol levels."
Other researchers have taken an interest in different red-meat-associated health problems, like colorectal cancer and the inflammation that accompanies it. This was the mechanistic link tackled by the lab of professor Karsten Zengler of the UC San Diego Departments of Pediatrics and Bioengineering—and it also led straight back to the gut microbes.
Zengler and colleagues recently published a paper in Nature Microbiology that focused on the effects of a red meat carbohydrate (or sugar) called Neu5Gc.
He explains, "If you eat animal proteins in your diet… the bound sugars in your diet are cleaved off in your gut and they get recycled. Your own cells will not recognize between the foreign sugars and your own sugars, because they look almost identical." The unsuspecting human cells then take up these foreign sugars — spurring antibody production and creating inflammation.
Zengler showed, however, that gut bacteria use enzymes to cleave off the sugar during digestion, stopping the inflammation and rendering the sugar harmless. "There's no enzyme in the human body that can cleave this [sugar] off. Humans 100% rely on the microbes to digest this food," he says.
Both researchers are quick to caution that the health effects of diet are complex. Other work indicates, for example, that while intake of red meat can affect TMAO levels, so can intake of fish and seafood. But these new lines of evidence could help explain why some people, ironically, seem to be in perfect health despite eating a lot of red meat: their ideal frequency of meat consumption may depend on their existing community of gut microbes.
"It helps explain what accounts for inter-person variability," Hazen says.
These emerging mechanisms reinforce overall why it's prudent to limit red meat, just as the nutritional guidelines advised in the first place. But both Hazen and Zengler predict that interventions to buffer the effects of too many ribeyes may be just around the corner.
Zengler says, "Our idea is that you basically can help your own digestive system detoxify these inflammatory compounds in meat, if you continue eating red meat or you want to eat a high amount of red meat." A possibly strategy, he says, is to use specific pre- or probiotics to cultivate an inflammation-reducing gut microbial community.
Hazen foresees the emergence of drugs that act not on the human, but on the human's gut microorganisms. "I think it's just a matter of time [before] we will have therapeutic interventions that actually target our gut microbes, just like the way we take drugs that lower cholesterol levels."
He adds, "It's a matter of 'stay tuned', I think."