The Dangers of Hype: How a Bold Claim and Sensational Media Unraveled a Company
Magnetic resonance imaging of the brain.
This past March, headlines suddenly flooded the Internet about a startup company called Nectome. Founded by two graduates of the Massachusetts Institute of Technology, the new company was charging people $10,000 to join a waiting list to have their brains embalmed, down to the last neuron, using an award-winning chemical compound.
While the lay public presumably burnt their wills and grew ever more excited about the end of humanity's quest for immortality, neurologists let out a collective sigh.
Essentially, participants' brains would turn to a substance like glass and remain in a state of near-perfect preservation indefinitely. "If memories can truly be preserved by a sufficiently good brain banking technique," Nectome's website explains, "we believe that within the century it could become feasible to digitize your preserved brain and use that information to recreate your mind." But as with most Faustian bargains, Nectome's proposition came with a serious caveat -- death.
That's right, in order for Nectome's process to properly preserve your connectome, the comprehensive map of the brain's neural connections, you must be alive (and under anesthesia) while the fluid is injected. This way, the company postulates, when the science advances enough to read and extract your memories someday, your vitrified brain will still contain your perfectly preserved essence--which can then be digitally recreated as a computer simulation.
Almost immediately this story gained buzz with punchy headlines: "Startup wants to upload your brain to the cloud, but has to kill you to do it," "San Junipero is real: Nectome wants to upload your brain," and "New tech firm promises eternal life, but you have to die."
While the lay public presumably burnt their wills and grew ever more excited about the end of humanity's quest for immortality, neurologists let out a collective sigh -- hype had struck the scientific community once again.
The truth about Nectome is that its claims are highly speculative and no hard science exists to suggest that our connectome is the key to our 'being,' nor that it can ever be digitally revived. "We haven't come even close to understanding even the most basic types of functioning in the brain," says neuroscientist Alex Fox, who was educated at the University of Queensland in Australia. "Memory storage in the brain is only a theoretical concept [and] there are some seriously huge gaps in our knowledge base that stand in the way of testing [the connectome] theory."
After the Nectome story broke, Harvard computational neuroscientist Sam Gershman tweeted out:
"Didn't anyone tell them that we've known the C Elegans (a microscopic worm) connectome for over a decade but haven't figured out how to reconstruct all of their memories? And that's only 7000 synapses compared to the trillions of synapses in the human brain!"
Hype can come from researchers themselves, who are under an enormous amount of pressure to publish original work and maintain funding.
How media coverage of Nectome went from an initial fastidiously researched article in the MIT Technology Review by veteran science journalist Antonio Regalado to the click-bait frenzy it became is a prime example of the 'science hype' phenomenon. According to Adam Auch, who holds a doctorate in philosophy from Dalhousie University in Nova Scotia, Canada, "Hype is a feature of all stages of the scientific dissemination process, from the initial circulation of preliminary findings within particular communities of scientists, to the process by which such findings come to be published in peer-reviewed journals, to the subsequent uptake these findings receive from the non-specialist press and the general public."
In the case of Nectome, hype was present from the word go. Riding the high of several major wins, including having raised over one million dollars in funding and partnering with well-known MIT neurologist Edward Boyden, Nectome founders Michael McCanna and Robert McIntyre launched their website on March 1, 2018. Just one month prior, they were able to purchase and preserve a newly deceased corpse in Portland, Oregon, showing that vitrifixation, their method of chemical preservation, could be used on a human specimen. It had previously won an award for preserving every synaptic structure on a rabbit brain.
The Nectome mission statement, found on its website, is laced with saccharine language that skirts the unproven nature of the procedure the company is peddling for big bucks: "Our mission is to preserve your brain well enough to keep all its memories intact: from that great chapter of your favorite book to the feeling of cold winter air, baking an apple pie, or having dinner with your friends and family."
This rhetoric is an example of hype that can come from researchers themselves, who are under an enormous amount of pressure to publish original work and maintain funding. As a result, there is a constant push to present science as "groundbreaking" when really, as is apparently the case with Nectome, it is only a small piece in a much larger effort.
Calling out the audacity of Nectome's posited future, neuroscientist Gershman commented to another publication, "The important question is whether the connectome is sufficient for memory: Can I reconstruct all memories knowing only the connections between neurons? The answer is almost certainly no, given our knowledge about how memories are stored (itself a controversial topic)."

The former home page of Nectome's website, which has now been replaced by a statement titled, "Response to recent press."
Furthermore, universities like MIT, who entered into a subcontract with Nectome, are under pressure to seek funding through partnerships with industry as a result of the Bayh-Dole Act of 1980. Also known as the Patent and Trademark Law Amendments Act, this piece of legislation allows universities to commercialize inventions developed under federally funded research programs, like Nectome's method of preserving brains, formally called Aldehyde-Stabilized Cryopreservation.
"[Universities use] every incentive now to talk about innovation," explains Dr. Ivan Oransky, president of the Association of Health Care Journalists and co-founder of retractionwatch.com, a blog that catalogues errors and fraud in published research. "Innovation to me is often a fancy word for hype. The role of journalists should not be to glorify what universities [say, but to] tell the closest version of the truth they can."
In this case, a combination of the hyperbolic press, combined with some impressively researched expose pieces, led MIT to cut its ties with Nectome on April 2nd, 2018, just two weeks after the news of their company broke.
The solution to the dangers of hype, experts say, is a more scientifically literate public—and less clickbait-driven journalism.
Because of its multi-layered nature, science hype carries several disturbing consequences. For one, exaggerated coverage of a discovery could mislead the public by giving them false hope or unfounded worry. And media hype can contribute to a general mistrust of science. In these instances, people might, as Auch puts it, "fall back on previously held beliefs, evocative narratives, or comforting biases instead of well-justified scientific evidence."
All of this is especially dangerous in today's 'fake news' era, when companies or political parties sow public confusion for their own benefit, such as with global warming. In the case of Nectome, the danger is that people might opt to end their lives based off a lacking scientific theory. In fact, the company is hoping to enlist terminal patients in California, where doctor-assisted suicide is legal. And 25 people have paid the $10,000 to join Nectome's waiting list, including Sam Altman, president of the famed startup accelerator Y Combinator. Nectome now has offered to refund the money.
Founders McCanna and McIntyre did not return repeated requests for comment for this article. A new statement on their website begins: "Vitrifixation today is a powerful research tool, but needs more research and development before anyone considers applying it in a context other than research."
The solution to the dangers of hype, experts say, is a more scientifically literate public—and less clickbait-driven journalism. Until then, it seems that companies like Nectome will continue to enjoy at least 15 minutes of fame.
Thanks to safety cautions from the COVID-19 pandemic, a strain of influenza has been completely eliminated.
If you were one of the millions who masked up, washed your hands thoroughly and socially distanced, pat yourself on the back—you may have helped change the course of human history.
Scientists say that thanks to these safety precautions, which were introduced in early 2020 as a way to stop transmission of the novel COVID-19 virus, a strain of influenza has been completely eliminated. This marks the first time in human history that a virus has been wiped out through non-pharmaceutical interventions, such as vaccines.
The flu shot, explained
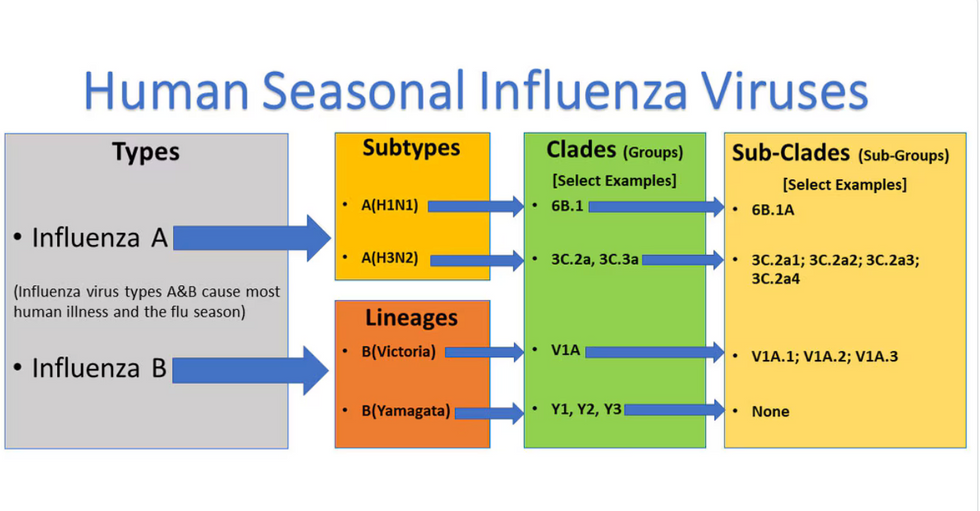
Influenza viruses type A and B are responsible for the majority of human illnesses and the flu season.
Centers for Disease Control
For more than a decade, flu shots have protected against two types of the influenza virus–type A and type B. While there are four different strains of influenza in existence (A, B, C, and D), only strains A, B, and C are capable of infecting humans, and only A and B cause pandemics. In other words, if you catch the flu during flu season, you’re most likely sick with flu type A or B.
Flu vaccines contain inactivated—or dead—influenza virus. These inactivated viruses can’t cause sickness in humans, but when administered as part of a vaccine, they teach a person’s immune system to recognize and kill those viruses when they’re encountered in the wild.
Each spring, a panel of experts gives a recommendation to the US Food and Drug Administration on which strains of each flu type to include in that year’s flu vaccine, depending on what surveillance data says is circulating and what they believe is likely to cause the most illness during the upcoming flu season. For the past decade, Americans have had access to vaccines that provide protection against two strains of influenza A and two lineages of influenza B, known as the Victoria lineage and the Yamagata lineage. But this year, the seasonal flu shot won’t include the Yamagata strain, because the Yamagata strain is no longer circulating among humans.
How Yamagata Disappeared

Flu surveillance data from the Global Initiative on Sharing All Influenza Data (GISAID) shows that the Yamagata lineage of flu type B has not been sequenced since April 2020.
Nature
Experts believe that the Yamagata lineage had already been in decline before the pandemic hit, likely because the strain was naturally less capable of infecting large numbers of people compared to the other strains. When the COVID-19 pandemic hit, the resulting safety precautions such as social distancing, isolating, hand-washing, and masking were enough to drive the virus into extinction completely.
Because the strain hasn’t been circulating since 2020, the FDA elected to remove the Yamagata strain from the seasonal flu vaccine. This will mark the first time since 2012 that the annual flu shot will be trivalent (three-component) rather than quadrivalent (four-component).
Should I still get the flu shot?
The flu shot will protect against fewer strains this year—but that doesn’t mean we should skip it. Influenza places a substantial health burden on the United States every year, responsible for hundreds of thousands of hospitalizations and tens of thousands of deaths. The flu shot has been shown to prevent millions of illnesses each year (more than six million during the 2022-2023 season). And while it’s still possible to catch the flu after getting the flu shot, studies show that people are far less likely to be hospitalized or die when they’re vaccinated.
Another unexpected benefit of dropping the Yamagata strain from the seasonal vaccine? This will possibly make production of the flu vaccine faster, and enable manufacturers to make more vaccines, helping countries who have a flu vaccine shortage and potentially saving millions more lives.
After his grandmother’s dementia diagnosis, one man invented a snack to keep her healthy and hydrated.
Founder Lewis Hornby and his grandmother Pat, sampling Jelly Drops—an edible gummy containing water and life-saving electrolytes.
On a visit to his grandmother’s nursing home in 2016, college student Lewis Hornby made a shocking discovery: Dehydration is a common (and dangerous) problem among seniors—especially those that are diagnosed with dementia.
Hornby’s grandmother, Pat, had always had difficulty keeping up her water intake as she got older, a common issue with seniors. As we age, our body composition changes, and we naturally hold less water than younger adults or children, so it’s easier to become dehydrated quickly if those fluids aren’t replenished. What’s more, our thirst signals diminish naturally as we age as well—meaning our body is not as good as it once was in letting us know that we need to rehydrate. This often creates a perfect storm that commonly leads to dehydration. In Pat’s case, her dehydration was so severe she nearly died.
When Lewis Hornby visited his grandmother at her nursing home afterward, he learned that dehydration especially affects people with dementia, as they often don’t feel thirst cues at all, or may not recognize how to use cups correctly. But while dementia patients often don’t remember to drink water, it seemed to Hornby that they had less problem remembering to eat, particularly candy.

Hornby wanted to create a solution for elderly people who struggled keeping their fluid intake up. He spent the next eighteen months researching and designing a solution and securing funding for his project. In 2019, Hornby won a sizable grant from the Alzheimer’s Society, a UK-based care and research charity for people with dementia and their caregivers. Together, through the charity’s Accelerator Program, they created a bite-sized, sugar-free, edible jelly drop that looked and tasted like candy. The candy, called Jelly Drops, contained 95% water and electrolytes—important minerals that are often lost during dehydration. The final product launched in 2020—and was an immediate success. The drops were able to provide extra hydration to the elderly, as well as help keep dementia patients safe, since dehydration commonly leads to confusion, hospitalization, and sometimes even death.
Not only did Jelly Drops quickly become a favorite snack among dementia patients in the UK, but they were able to provide an additional boost of hydration to hospital workers during the pandemic. In NHS coronavirus hospital wards, patients infected with the virus were regularly given Jelly Drops to keep their fluid levels normal—and staff members snacked on them as well, since long shifts and personal protective equipment (PPE) they were required to wear often left them feeling parched.
In April 2022, Jelly Drops launched in the United States. The company continues to donate 1% of its profits to help fund Alzheimer’s research.

