The First Cloned Monkeys Provoked More Shrugs Than Shocks

Zhong Zhong and Hua Hua, the two cloned macaques.
A few months ago, it was announced that not one, but two healthy long-tailed macaque monkeys were cloned—a first for primates of any kind. The cells were sourced from aborted monkey fetuses and the DNA transferred into eggs whose nuclei had been removed, the same method that was used in 1996 to clone "Dolly the Sheep." Two live births, females named Zhong Zhong and Hua Hua, resulted from 60 surrogate mothers. Inefficient, it's true. But over time, the methods are likely to be improved.
The scientist who supervised the project predicts that cloning, along with gene editing, will result in "ideal primate models" for studying disease mechanisms and drug screening.
Dr. Gerald Schatten, a famous would-be monkey cloner, authored a controversial paper in 2003 describing the formidable challenges to cloning monkeys and humans, speculating that the feat might never be accomplished. Now, some 15 years later, that prediction, insofar as it relates to monkeys, has blown away.
Zhong Zhong and Hua Hua were created at the Chinese Academy of Science's Institute of Neuroscience in Shanghai. The Institute founded in 1999 boasts 32 laboratories, expanding to 50 labs in 2020. It maintains two non-human primate research facilities.
The founder and director, Dr. Mu-ming Poo, supervised the project. Poo is an extremely accomplished senior researcher at the pinnacle of his field, a distinguished professor emeritus in Biology at UC Berkeley. In 2016, he was awarded the prestigious $500,000 Gruber Neuroscience Prize. At that time, Poo's experiments were described by a colleague as being "innovative and very often ingenious."
Poo maintains the reputation of studying some of the most important questions in cellular neuroscience.
But is society ready to accept cloned primates for medical research without the attendant hysteria about fears of cloned humans?
By Western standards, use of non-human primates in research focuses on the welfare of the animal subjects. As PETA reminds us, there is a dreadful and sad history of mistreatment. Dr. Poo assures us that his cloned monkeys are treated ethically and that the Institute is compliant with the highest regulatory standards, as promulgated by the U.S. National Institutes of Health.
He presents the noblest justifications for the research. He predicts that cloning, along with gene editing, will result in "ideal primate models" for studying disease mechanisms and drug screening. He declares that this will eventually help to solve Parkinson's, Huntington's and Alzheimer's disease.
But is society ready to accept cloned primates for medical research without the attendant hysteria about fears of cloned humans? It appears so.
While much of the news coverage expressed this predictable worry, my overall impression is that the societal response was muted. Where was the expected outrage? Then again, we've come a long way since Dolly the Sheep in terms of both the science and the cultural acceptance of cloning. Perhaps my unique vantage point can provide perspective on how much attitudes have evolved.
Perhaps my unique vantage point can provide perspective on how much attitudes have evolved.
I sometimes joke that I am the world's only human cloning lawyer—a great gig but there are still no clients.
I first crashed into the cloning scene in 2002 when I sued the so-called human cloning company "Clonaid" and asked in court to have a temporary guardian appointed for the alleged first human clone "Baby Eve." The claim needed to be tested, and mine was the first case ever aiming to protect the rights of a human clone. My legal basis was child welfare law, protecting minors from abuse, negligence, and exploitation.
The case had me on back-to-back global television broadcasts around the world; there was live news and "breathless" coverage at the courthouse emblazoned in headlines in every language on the planet. Cloning was, after all, perceived as a species-altering event: asexual reproduction. The controversy dominated world headlines for month until Clonaid's claim was busted as the "fakest" of fake news.
Fresh off the cloning case, the scientific community reached out to me, seeing me as the defender of legitimate science, an opponent of cloning human babies but a proponent of using cloning techniques to accelerate ethical regenerative medicine and embryonic stem cell research in general.
The years 2003 to 2006 were the era of the "stem cell wars" and a dominant issue was human cloning. Social conservative lawmakers around the world were seeking bans or criminalization not only of cloning babies but also the cloning of cells to match the donor's genetics. Scientists were being threatened with fines and imprisonment. Human cloning was being challenged in the United Nations with the United States backing a global treaty to ban and morally condemn all cloning -- including the technique that was crucial for research.
Scientists and patients were touting the cloning technique as a major biomedical breakthrough because cells could be created as direct genetic matches from a specific donor.
At the same time, scientists and patients were touting the cloning technique as a major biomedical breakthrough because cells could be created as direct genetic matches from a specific donor.
So my organization organized a conference at UN headquarters to defend research cloning and all the big names in stem cell research were there. We organized petitions to the UN and faxed 35,000 signatures to the country mission. These ongoing public policy battles were exacerbated in part because of the growing fear that cloning babies was just around the corner.
Then in 2005, the first cloned dog stunned the world, an Afghan hound named Snuppy. I met him when I visited the laboratories of Professor Woo Suk Hwang in Korea. His minders let me hold his leash -- TIME magazine's scientific breakthrough of the year. He didn't lick me or even wag his tail; I figured he must not like lawyers.
Tragically, soon thereafter, I witnessed firsthand Dr. Hwang's fall from grace when his human stem cell cloning breakthroughs proved false. The massive scientific misconduct rocked the nation of Korea, stem cell science in general, and provoked terrible news coverage.
Nevertheless, by 2007, the proposed bans lost steam, overridden by the advent of a Japanese researcher's Nobel Prize winning formula for reprogramming human cells to create genetically matched cell lines, not requiring the destruction of human embryos.
After years of panic, none of the recent cloning headlines has caused much of a stir.
Five years later, when two American scientists accomplished therapeutic human cloned stem cell lines, their news was accepted without hysteria. Perhaps enough time had passed since Hwang and the drama was drained.
In the just past 30 days we have seen more cloning headlines. Another cultural icon, Barbara Streisand, revealed she owns two cloned Coton de Tulear puppies. The other weekend, the television news show "60 Minutes" devoted close to an hour on the cloned ponies used at the top level of professional polo. And in India, scientists just cloned the first Assamese buffalo.
And you know what? After years of panic, none of this has caused much of a stir. It's as if the future described by Alvin Toffler in "Future Shock" has arrived and we are just living with it. A couple of cloned monkeys barely move the needle.
Perhaps it is the advent of the Internet and the overall dilution of wonder and outrage. Or maybe the muted response is rooted in popular culture. From Orphan Black to the plotlines of dozens of shows and books, cloning is just old news. The hand-wringing discussions about "human dignity" and "slippery slopes" have taken a backseat to the AI apocalypse and Martian missions.
We humans are enduring plagues of dementia and Alzheimer's, and we will need more monkeys. I will take mine cloned, if it will speed progress.
Personally, I still believe that cloned children should not be an option. Child welfare laws might be the best deterrent.
The same does not hold for cloning monkey research subjects. Squeamishness aside, I think Zhong Zhong and Hua Hua will soon be joined by a legion of cloned macaques and probably marmosets.
We humans are enduring plagues of dementia and Alzheimer's, and we will need more monkeys. I will take mine cloned, if it will speed the mending of these consciousness-destroying afflictions.
Scientific revolutions once took centuries, then decades, and now seem to bombard us daily. The convergence of technologies has accelerated the future. To Zhong Zhong and Hua Hua, my best wishes with the hope that their sacrifices will contribute to the health of all primates -- not just humans.
60-Second Video: How Safe Are the COVID-19 Vaccines?
Comparing the Risks of COVID-19 vs. the Risks of the Vaccines:
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.
For Kids with Progeria, New Therapies May Offer Revolutionary Hope for a Longer Life
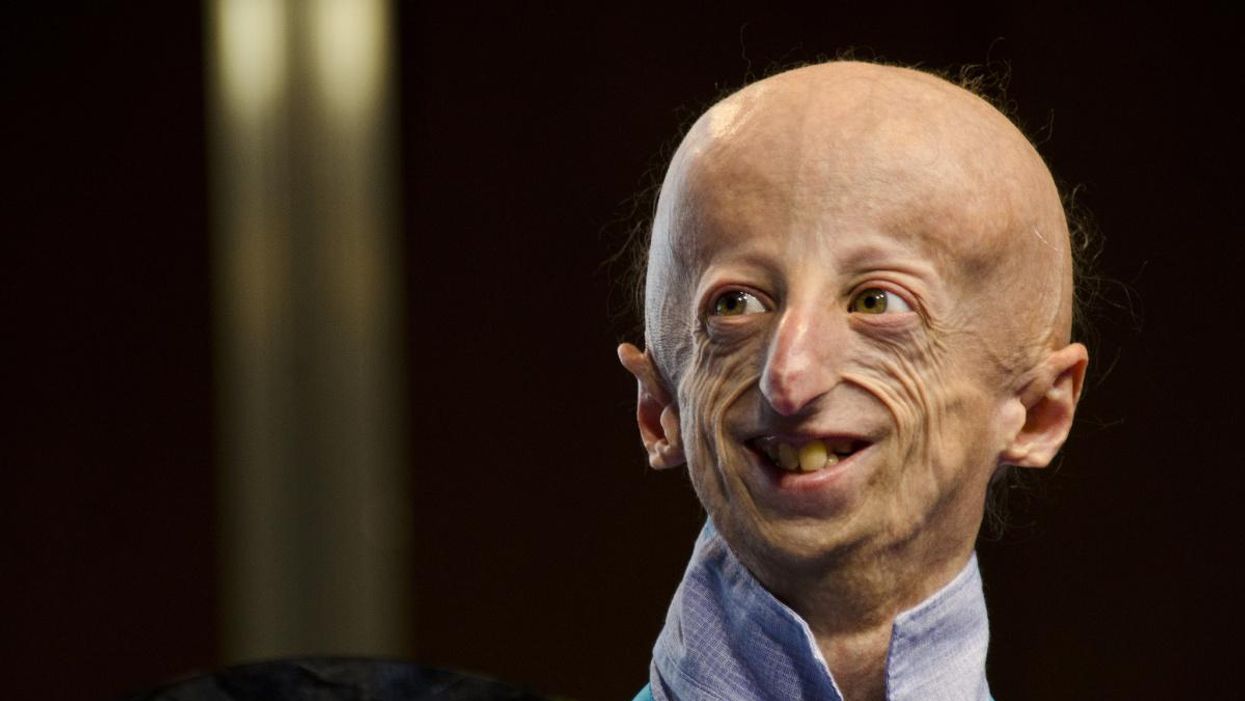
Sammy Basso is one of around 400 young people in the world with progeria.
Sammy Basso has some profound ideas about fate. As long as he has been alive, he has known he has minimal control over his own. His parents, however, had to transition from a world of unlimited possibility to one in which their son might not live to his 20s.
"I remember very clearly that day because Sammy was three years old," his mother says of the day a genetic counselor diagnosed Sammy with progeria. "It was a devastating day for me."
But to Sammy, he has always been himself: a smart kid, interested in science, a little smaller than his classmates, with one notable kink in his DNA. In one copy of the gene that codes for the protein Lamin A, Sammy has a T where there should be a C. The incorrect code creates a toxic protein called progerin, which destabilizes Sammy's cells and makes him age much faster than a person who doesn't have the mutation. The older he gets, the more he is in danger of strokes, heart failure, or a heart attack. "I am okay with my situation," he says from his home in Tezze sul Brenta, Italy. "But I think, yes, fate has a great role in my life."
Just 400 or so people in the world live with progeria: The mutation that causes it usually arises de novo, or "of new," meaning that it is not inherited but happens spontaneously during gestation. The challenge, as with all rare diseases, is that few cases means few treatments.
"When we first started, there was absolutely nothing out there," says Leslie Gordon, a physician-researcher who co-founded the Progeria Research Foundation in 1999 after her own son, also named Sam, was diagnosed with the disease. "We knew we had to jumpstart the entire field, so we collected money through road races and special events and writing grants and all sorts of donors… I think the first year we raised $75,000, most of it from one donor."
"We have not only the possibility but the responsibility to make the world a better world, and also to make a body a better body."
By 2003, the foundation had collaborated with Francis Collins, a geneticist who is now director of the National Institutes of Health, to work out the genetic basis for progeria—that single mutation Sammy has. The discovery led to interest in lonafarnib, a drug that was already being used in cancer patients but could potentially operate downstream of the mutation, preventing the buildup of the defective progerin in the body. "We funded cellular studies to look at a lonafarnib in cells, mouse studies to look at lonafarnib in mouse models of progeria… and then we initiated the clinical trials," Gordon says.
Sammy Basso's family had gotten involved with the Progeria Research Foundation through their international patient registry, which maintains relationships with families in 49 countries. "We started to hear about lonafarnib in 2006 from Leslie Gordon," says Sammy's father, Amerigo Basso, with his son translating. "She told us about the lonafarnib. And we were very happy because for the first time we understood that there was something that could help our son and our lives." Amerigo used the Italian word speranza, which means hope.
Still, Sammy wasn't sure if lonafarnib was right for him. "Since when I was very young I thought that everything happens for a reason. So, in my mind, if God made me with progeria, there was a reason, and to try to heal from progeria was something wrong," he says. Gradually, his parents and doctors, and Leslie Gordon, convinced him otherwise. Sammy began to believe that God was also the force behind doctors, science, and research. "And so we have not only the possibility but the responsibility to make the world a better world, and also to make a body a better body," he says.

Sammy Basso and his parents.
Courtesy of Basso
Sammy began taking lonafarnib, with the Progeria Research Foundation intermittently flying him, and other international trial participants, to Boston for tests. He was immediately beset by some of the drug's more unpleasant side effects: Stomach problems, nausea, and vomiting. "The first period was absolutely the worst period of my life," he says.
At first, doctors prescribed other medicines for the side effects, but to Sammy it had as much effect as drinking water. He visited doctor after doctor, with some calling him weekly or even daily to ask how he was doing. Eventually the specialists decided that he should lower his dose, balancing his pain with the benefit of the drug. Sammy can't actually feel any positive effect of the lonafarnib, but his health measurements have improved relative to people with progeria who don't take it.
While they never completely disappeared, Sammy's side effects decreased to the point that he could live. Inspired by the research that led to lonafarnib, he went to university to study molecular biology. For his thesis work, he travelled to Spain to perform experiments on cells and on mice with progeria, learning how to use the gene-editing technique CRISPR-Cas9 to cut out the mutated bit of DNA. "I was so excited to participate in this study," Sammy says. He felt like his work could make a difference.
In 2018, the Progeria Research Foundation was hosting one of their biennial workshops when Francis Collins, the researcher who had located the mutation behind progeria 15 years earlier, got in touch with Leslie Gordon. "Francis called me and said, Hey, I just saw a talk by David Liu from the Broad [Institute]. And it was pretty amazing. He has been looking at progeria and has very early, but very exciting data… Do you have any spaces, any slots you could make in your program for late breaking news?"
Gordon found a spot, and David Liu came to talk about what was going on in his lab, which was an even more advanced treatment that led to mice with the progeria mutation living into their senior mouse years—substantially closer to a normal lifespan. Liu's lab had built on the idea of CRISPR-Cas9 to create a more elegant genetic process called base editing: Instead of chopping out mutated DNA, a scientist could chemically convert an incorrect DNA letter to the correct one, like the search and replace function in word processing software. Mice who had their Lamin-A mutations corrected this way lived more than twice as long as untreated animals.
Sammy was in the audience at Dr. Liu's talk. "When I heard about this base editing as a younger scientist, I thought that I was living in the future," he says. "When my parents had my diagnosis of progeria, the science knew very little information about DNA. And now we are talking about healing the DNA… It is incredible."
Lonafarnib (also called Zokinvy) was approved by the US Food and Drug Administration this past November. Sammy, now 25, still takes it, and still manages his side effects. With luck, the gift of a few extra years will act as a bridge until he can try Liu's revolutionary new gene treatment, which has not yet begun testing in humans. While Leslie Gordon warns that she's always wrong about things like this, she hopes to see the new base editing techniques in clinical trials in the next year or two. Sammy won't need to be convinced to try it this time; his thinking on fate has evolved since his first encounter with lonafarnib.
"I would be very happy to try it," he says. "I know that for a non-scientist it can be difficult to understand. Some people think that we are the DNA. We are not. The DNA is a part of us, and to correct it is to do what we are already doing—just better." In short, a gene therapy, while it may seem like science fiction, is no different from a pill. For Sammy, both are a new way to think about fate: No longer something that simply happens to him.