Vaccine Passports Are a Premature Solution to A Challenging Problem
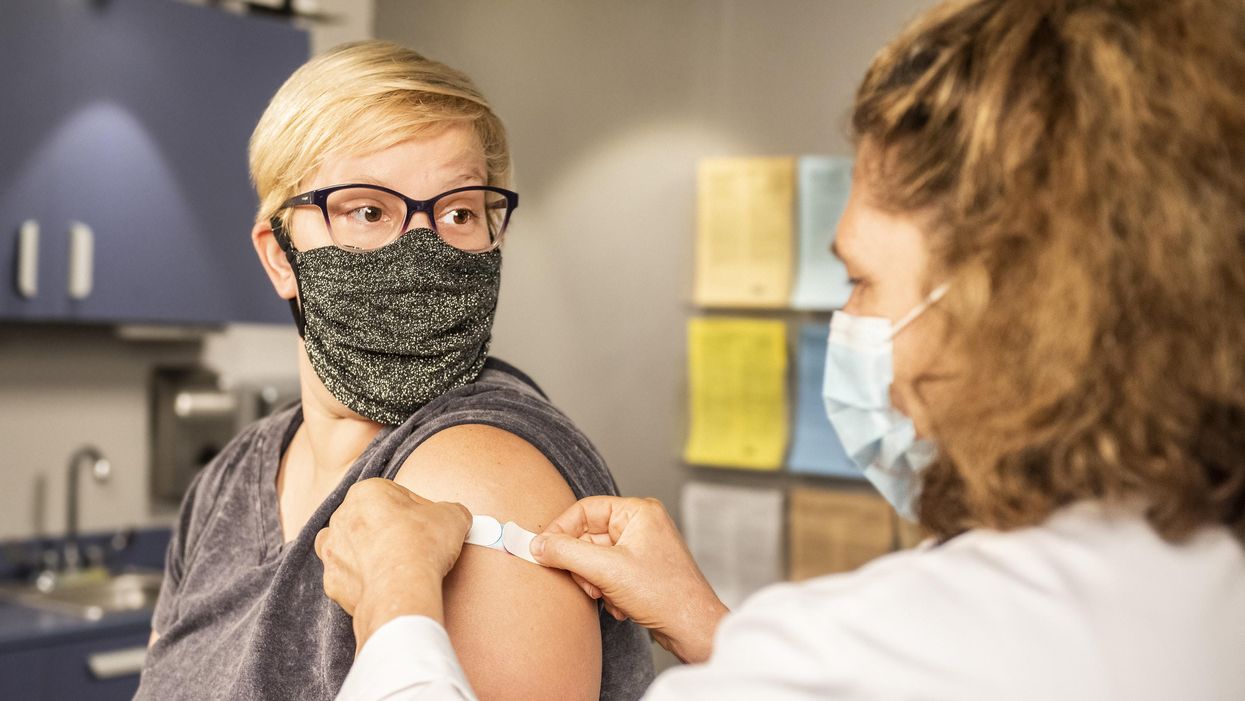
Vaccine passports, if required by businesses and universities, may give people a false sense of security, one expert cautions.
Vaccines are one of the greatest public health accomplishments of all time. For centuries, public health has relied on vaccinations to prevent and control disease outbreaks for a plethora of infectious scourges, with our crowning achievement being the successful eradication of smallpox.
The purpose of vaccine documentation is to provide proof of an individual's protection from either becoming infected or transmitting a vaccine-preventable disease. Vouching for these protections requires a firm knowledge about the epidemiology of the disease, as well as scientific knowledge concerning the efficacy of the vaccine. The vaccines we currently require be documented have met these tests; the vaccine for COVID-19 has not yet been proven to do so.
Let's acknowledge that the term "vaccine passport" is a poor choice of words. Passports are a legal travel document created by nations and governed by law for identification of the bearer to control entry and exit from nation states. They often serve as legal forms of identification and as a record of international travel. They are generally very sophisticated documents that have been created in a secure manner and may include a range of electronic and, in some cases, biometric measures such as fingerprints to ensure the holder is indeed who they say they are. Vaccine passports are medical documents used to document the vaccination status of an individual. They do not undergo the same level of administrative scrutiny and cannot be used to verify that the presenter is indeed the vaccinated individual. Some companies do have electronic methods to address concerns about verification; however, most people currently have paper records that can be easily falsified.
"Vaccine passports" as currently proposed risk giving people a false sense of security.
Successful disease control from vaccination programs relies on the ability to vaccinate at a level that prevents large-scale disease spread and the ability to rapidly identify the presence of disease outbreaks. It requires reliable, safe, and effective vaccines that are easily delivered in clinical and nonclinical settings. Keeping vaccination information as a part of the medical record, and even having a separate specialized vaccine record for personal use, is a time-honored tradition.
Keeping a vaccination record provides a method to keep track of the many shots one receives and serves as a visual reminder to help ensure the appropriate vaccine shot schedule is maintained for vaccines requiring multiple doses. The vaccine record, when combined with vaccine safety monitoring systems, serves as a mechanism to track adverse events to monitor and ensure the safety of vaccines as a consumer product. The record also serves as the official record of vaccination when required for administrative or legally prescribed purposes.
"Vaccine passports" as currently proposed risk giving people a false sense of security. In the case of the COVID-19 vaccines currently approved for use, many of the essential questions remain unanswered. While we do know the current three vaccines are highly protective against severe disease and death, and there is some evidence that these vaccinations do reduce infections and virus transmission of SARS-CoV-2, we do not yet know the full degree to which this occurs.
For example, we know there have been some cases of people that have been infected in close proximity to getting their full vaccination and rare cases of breakthrough reinfections. A breakthrough infection in a restaurant is a challenge for contact tracing, but an outbreak from a movie theater exposure or a baseball game could spark a major outbreak at our current level of vaccination. Current CDC guidance recommends continued mask wearing in order to address these concerns.
We also do not yet know how long the protections will last and if or when a booster or revaccination is required. In effect, it is too soon to know. Should an annual booster shot be required, then a vaccine passport would require annual updating, a process more frequent than renewal of a driver's license.
We also know that the current SARS-CoV-2 virus is mutating briskly. While the current approved vaccines have remained effective overall, there is evidence of some degree of degradation in vaccine effectiveness against some of the circulating strains. We also have sparse data on many of the other emerging strains of concern because we have not had the surveillance capacity in the U.S. to gain an adequate sense of how the virus is changing to fully align vaccine effectiveness with viral capabilities.
The risk of people misusing these "passports" is troubling. The potential for using these documents for hiring, firing or job limitation is a serious concern. Unvaccinated workers are at risk of this form of discrimination even from well-meaning employers or supervisors. Health insurers are prohibited by the Affordable Care Act from discriminating based on preexisting conditions, but they could probably charge a higher premium for unvaccinated individuals. There also is a risk of stigmatizing individuals who are not vaccinated or have left their vaccine documentation at home. Another concern: the opportunity to discriminate based on race, gender, sexual orientation, or religion, using one's vaccination status as an excuse.
These "passports" are being discussed as a "ticket verification" for entry to many activities, including dining at restaurants, flying domestically and/or internationally, going to movie theaters and sporting events, etc. These are all activities we already are doing at reduced levels and for which wearing a mask, hand hygiene and physical distancing are effective disease control practices. COVID-19 vaccines are indeed the measure that will make the ability to totally reopen our society complete, but we are not there yet. Documentation of one's COVID-19 vaccine status may be useful in selected situations in the future. That remains to be seen.
Finally, inadequate vaccine supply and disparities in vaccine delivery have created enormous challenges in providing equal access to vaccination. Also, the amount of misinformation, disinformation, and lingering vaccine hesitancy continue to limit the speed at which we will reach the level of vaccination of the population that would make this documentation meaningful. The requirement for "vaccine passports" is already alienating people who are opposed to vaccinations for a variety of reasons, paradoxically risking reduced vaccine uptake. This politicization of the vaccination effort is of concern. There are indeed people who, due to medical contraindications or legal exemptions, will not be vaccinated, and we do not yet have a national framework on how to address this.
Vaccine passports are not the solution for reopening our society — a robust vaccination program is. The requirement to document one's vaccination status for COVID-19 may one day have its place. For now, it is an idea whose time has not yet come.
Editor's Note: This op/ed is part of a "Big Question" series on the ethics of vaccine passports. Read the flip side argument here.
Scientists Are Working to Decipher the Puzzle of ‘Broken Heart Syndrome’
Elaine Kamil had just returned home after a few days of business meetings in 2013 when she started having chest pains. At first Kamil, then 66, wasn't worried—she had had some chest pain before and recently went to a cardiologist to do a stress test, which was normal.
"I can't be having a heart attack because I just got checked," she thought, attributing the discomfort to stress and high demands of her job. A pediatric nephrologist at Cedars-Sinai Hospital in Los Angeles, she takes care of critically ill children who are on dialysis or are kidney transplant patients. Supporting families through difficult times and answering calls at odd hours is part of her daily routine, and often leaves her exhausted.
She figured the pain would go away. But instead, it intensified that night. Kamil's husband drove her to the Cedars-Sinai hospital, where she was admitted to the coronary care unit. It turned out she wasn't having a heart attack after all. Instead, she was diagnosed with a much less common but nonetheless dangerous heart condition called takotsubo syndrome, or broken heart syndrome.
A heart attack happens when blood flow to the heart is obstructed—such as when an artery is blocked—causing heart muscle tissue to die. In takotsubo syndrome, the blood flow isn't blocked, but the heart doesn't pump it properly. The heart changes its shape and starts to resemble a Japanese fishing device called tako-tsubo, a clay pot with a wider body and narrower mouth, used to catch octopus.
"The heart muscle is stunned and doesn't function properly anywhere from three days to three weeks," explains Noel Bairey Merz, the cardiologist at Cedar Sinai who Kamil went to see after she was discharged.
"The heart muscle is stunned and doesn't function properly anywhere from three days to three weeks."
But even though the heart isn't permanently damaged, mortality rates due to takotsubo syndrome are comparable to those of a heart attack, Merz notes—about 4-5 percent of patients die from the attack, and 20 percent within the next five years. "It's as bad as a heart attack," Merz says—only it's much less known, even to doctors. The condition affects only about 1 percent of people, and there are around 15,000 new cases annually. It's diagnosed using a cardiac ventriculogram, an imaging test that allows doctors to see how the heart pumps blood.
Scientists don't fully understand what causes Takotsubo syndrome, but it usually occurs after extreme emotional or physical stress. Doctors think it's triggered by a so-called catecholamine storm, a phenomenon in which the body releases too much catecholamines—hormones involved in the fight-or-flight response. Evolutionarily, when early humans lived in savannas or forests and had to either fight off predators or flee from them, these hormones gave our ancestors the needed strength and stamina to take either action. Released by nerve endings and by the adrenal glands that sit on top of the kidneys, these hormones still flood our bodies in moments of stress, but an overabundance of them could sometimes be damaging.

Elaine Kamil
A study by scientists at Harvard Medical School linked increased risk of takotsubo to higher activity in the amygdala, a brain region responsible for emotions that's involved in responses to stress. The scientists believe that chronic stress makes people more susceptible to the syndrome. Notably, one small study suggested that the number of Takotsubo cases increased during the COVID-19 pandemic.
There are no specific drugs to treat takotsubo, so doctors rely on supportive therapies, which include medications typically used for high blood pressure and heart failure. In most cases, the heart returns to its normal shape within a few weeks. "It's a spontaneous recovery—the catecholamine storm is resolved, the injury trigger is removed and the heart heals itself because our bodies have an amazing healing capacity," Merz says. It also helps that tissues remain intact. 'The heart cells don't die, they just aren't functioning properly for some time."
That's the good news. The bad news is that takotsubo is likely to strike again—in 5-20 percent of patients the condition comes back, sometimes more severe than before.
That's exactly what happened to Kamil. After getting her diagnosis in 2013, she realized that she actually had a previous takotsubo episode. In 2010, she experienced similar symptoms after her son died. "The night after he died, I was having severe chest pain at night, but I was too overwhelmed with grief to do anything about it," she recalls. After a while, the pain subsided and didn't return until three years later.
For weeks after her second attack, she felt exhausted, listless and anxious. "You lose confidence in your body," she says. "You have these little twinges on your chest, or if you start having arrhythmia, and you wonder if this is another episode coming up. It's really unnerving because you don't know how to read these cues." And that's very typical, Merz says. Even when the heart muscle appears to recover, patients don't return to normal right away. They have shortens of breath, they can't exercise, and they stay anxious and worried for a while.
Women over the age of 50 are diagnosed with takotsubo more often than other demographics. However, it happens in men too, although it typically strikes after physical stress, such as a triathlon or an exhausting day of cycling. Young people can also get takotsubo. Older patients are hospitalized more often, but younger people tend to have more severe complications. It could be because an older person may go for a jog while younger one may run a marathon, which would take a stronger toll on the body of a person who's predisposed to the condition.
Notably, the emotional stressors don't always have to be negative—the heart muscle can get out of shape from good emotions, too. "There have been case reports of takotsubo at weddings," Merz says. Moreover, one out of three or four takotsubo patients experience no apparent stress, she adds. "So it could be that it's not so much the catecholamine storm itself, but the body's reaction to it—the physiological reaction deeply embedded into out physiology," she explains.
Merz and her team are working to understand what makes people predisposed to takotsubo. They think a person's genetics play a role, but they haven't yet pinpointed genes that seem to be responsible. Genes code for proteins, which affect how the body metabolizes various compounds, which, in turn, affect the body's response to stress. Pinning down the protein involved in takotsubo susceptibility would allow doctors to develop screening tests and identify those prone to severe repeating attacks. It will also help develop medications that can either prevent it or treat it better than just waiting for the body to heal itself.
Researchers at the Imperial College London found that elevated levels of certain types of microRNAs—molecules involved in protein production—increase the chances of developing takotsubo.
In one study, researchers tried treating takotsubo in mice with a drug called suberanilohydroxamic acid, or SAHA, typically used for cancer treatment. The drug improved cardiac health and reversed the broken heart in rodents. It remains to be seen if the drug would have a similar effect on humans. But identifying a drug that shows promise is progress, Merz says. "I'm glad that there's research in this area."
This article was originally published by Leaps.org on July 28, 2021.
Lina Zeldovich has written about science, medicine and technology for Popular Science, Smithsonian, National Geographic, Scientific American, Reader’s Digest, the New York Times and other major national and international publications. A Columbia J-School alumna, she has won several awards for her stories, including the ASJA Crisis Coverage Award for Covid reporting, and has been a contributing editor at Nautilus Magazine. In 2021, Zeldovich released her first book, The Other Dark Matter, published by the University of Chicago Press, about the science and business of turning waste into wealth and health. You can find her on http://linazeldovich.com/ and @linazeldovich.
Did Anton the AI find a new treatment for a deadly cancer?
Researchers used a supercomputer to learn about the subtle movement of a cancer-causing molecule, and then they found the precise drug that can recognize that motion.
Bile duct cancer is a rare and aggressive form of cancer that is often difficult to diagnose. Patients with advanced forms of the disease have an average life expectancy of less than two years.
Many patients who get cancer in their bile ducts – the tubes that carry digestive fluid from the liver to the small intestine – have mutations in the protein FGFR2, which leads cells to grow uncontrollably. One treatment option is chemotherapy, but it’s toxic to both cancer cells and healthy cells, failing to distinguish between the two. Increasingly, cancer researchers are focusing on biomarker directed therapy, or making drugs that target a particular molecule that causes the disease – FGFR2, in the case of bile duct cancer.
A problem is that in targeting FGFR2, these drugs inadvertently inhibit the FGFR1 protein, which looks almost identical. This causes elevated phosphate levels, which is a sign of kidney damage, so doses are often limited to prevent complications.
In recent years, though, a company called Relay has taken a unique approach to picking out FGFR2, using a powerful supercomputer to simulate how proteins move and change shape. The team, leveraging this AI capability, discovered that FGFR2 and FGFR1 move differently, which enabled them to create a more precise drug.
Preliminary studies have shown robust activity of this drug, called RLY-4008, in FGFR2 altered tumors, especially in bile duct cancer. The drug did not inhibit FGFR1 or cause significant side effects. “RLY-4008 is a prime example of a precision oncology therapeutic with its highly selective and potent targeting of FGFR2 genetic alterations and resistance mutations,” says Lipika Goyal, assistant professor of medicine at Harvard Medical School. She is a principal investigator of Relay’s phase 1-2 clinical trial.
Boosts from AI and a billionaire
Traditional drug design has been very much a case of trial and error, as scientists investigate many molecules to see which ones bind to the intended target and bind less to other targets.
“It’s being done almost blindly, without really being guided by structure, so it fails very often,” says Olivier Elemento, associate director of the Institute for Computational Biomedicine at Cornell. “The issue is that they are not sampling enough molecules to cover some of the chemical space that would be specific to the target of interest and not specific to others.”
Relay’s unique hardware and software allow simulations that could never be achieved through traditional experiments, Elemento says.
Some scientists have tried to use X-rays of crystallized proteins to look at the structure of proteins and design better drugs. But they have failed to account for an important factor: proteins are moving and constantly folding into different shapes.
David Shaw, a hedge fund billionaire, wanted to help improve drug discovery and understood that a key obstacle was that computer models of molecular dynamics were limited; they simulated motion for less than 10 millionths of a second.
In 2001, Shaw set up his own research facility, D.E. Shaw Research, to create a supercomputer that would be specifically designed to simulate protein motion. Seven years later, he succeeded in firing up a supercomputer that can now conduct high speed simulations roughly 100 times faster than others. Called Anton, it has special computer chips to enable this speed, and its software is powered by AI to conduct many simulations.
After creating the supercomputer, Shaw teamed up with leading scientists who were interested in molecular motion, and they founded Relay Therapeutics.
Elemento believes that Relay’s approach is highly beneficial in designing a better drug for bile duct cancer. “Relay Therapeutics has a cutting-edge approach for molecular dynamics that I don’t believe any other companies have, at least not as advanced.” Relay’s unique hardware and software allow simulations that could never be achieved through traditional experiments, Elemento says.
How it works
Relay used both experimental and computational approaches to design RLY-4008. The team started out by taking X-rays of crystallized versions of both their intended target, FGFR2, and the almost identical FGFR1. This enabled them to get a 3D snapshot of each of their structures. They then fed the X-rays into the Anton supercomputer to simulate how the proteins were likely to move.
Anton’s simulations showed that the FGFR1 protein had a flap that moved more frequently than FGFR2. Based on this distinct motion, the team tried to design a compound that would recognize this flap shifting around and bind to FGFR2 while steering away from its more active lookalike.
For that, they went back Anton, using the supercomputer to simulate the behavior of thousands of potential molecules for over a year, looking at what made a particular molecule selective to the target versus another molecule that wasn’t. These insights led them to determine the best compounds to make and test in the lab and, ultimately, they found that RLY-4008 was the most effective.
Promising results so far
Relay began phase 1-2 trials in 2020 and will continue until 2024. Preliminary results showed that, in the 17 patients taking a 70 mg dose of RLY-4008, the drug worked to shrink tumors in 88 percent of patients. This was a significant increase compared to other FGFR inhibitors. For instance, Futibatinib, which recently got FDA approval, had a response rate of only 42 percent.
Across all dose levels, RLY-4008 shrank tumors by 63 percent in 38 patients. In more good news, the drug didn’t elevate their phosphate levels, which suggests that it could be taken without increasing patients’ risk for kidney disease.
“Objectively, this is pretty remarkable,” says Elemento. “In a small patient study, you have a molecule that is able to shrink tumors in such a high fraction of patients. It is unusual to see such good results in a phase 1-2 trial.”
A simulated future
The research team is continuing to use molecular dynamic simulations to develop other new drug, such as one that is being studied in patients with solid tumors and breast cancer.
As for their bile duct cancer drug, RLY-4008, Relay plans by 2024 to have tested it in around 440 patients. “The mature results of the phase 1-2 trial are highly anticipated,” says Goyal, the principal investigator of the trial.
Sameek Roychowdhury, an oncologist and associate professor of internal medicine at Ohio State University, highlights the need for caution. “This has early signs of benefit, but we will look forward to seeing longer term results for benefit and side effect profiles. We need to think a few more steps ahead - these treatments are like the ’Whack-a-Mole game’ where cancer finds a way to become resistant to each subsequent drug.”
“I think the issue is going to be how durable are the responses to the drug and what are the mechanisms of resistance,” says Raymond Wadlow, an oncologist at the Inova Medical Group who specializes in gastrointestinal and haematological cancer. “But the results look promising. It is a much more selective inhibitor of the FGFR protein and less toxic. It’s been an exciting development.”

