What Will Make the Public Trust a COVID-19 Vaccine?

A successful deployment of an eventual vaccine will mean grappling with ongoing cultural tensions.
With a brighter future hanging on the hopes of an approved COVID-19 vaccine, is it possible to win over the minds of fearful citizens who challenge the value or safety of vaccination?
Globally, nine COVID-19 vaccines so far are being tested for safety in early phase human clinical trials.
It's a decades-old practice. With a dose injected into the arm of a healthy patient, doctors aim to prevent illness with a vaccine shot designed to trigger a person's immune system to fight serious infection without getting the disease.
This week, in fact, the U.S. frontrunner vaccine candidate, developed by Moderna, safely produced an immune response in the first eight healthy volunteers, the company announced. A large efficacy trial is planned to start in July. But if positive signals for safety and efficacy result from that trial, will that be enough to convince the public to broadly embrace a new vaccine?
"Throughout the history of vaccines there has always been a small vocal minority who don't believe vaccines work or don't trust the science," says sociologist and researcher Jennifer Reich, a professor at the University of Colorado in Denver and author of Calling the Shots: Why Parents Reject Vaccines.
Research indicates that only about 2 percent of the population say vaccines aren't necessary under any circumstance. Remarkably, a quarter to one third of American parents delay or reject the shots, not because they are anti-vaccine, but because they disapprove of the recommended timing or administration, says Reich.
Additionally, addressing distrust about how they come to market is key when talking to parents, workers or anyone targeted for a new vaccine, she says.
"When I talk to parents about why they reject vaccines for their kids, a lot of them say that they don't fully trust the process by which vaccines are regulated and tested," says Reich. "They don't trust that vaccine manufacturers -- which are for-profit companies -- are looking out for public health."
Balancing Act
Globally, nine COVID-19 vaccine candidates so far are being tested for safety in early phase human clinical trials and more than 100 are under development as scientists hustle to curtail the disease. Creating a new vaccine at a record pace requires a delicate balance of benefit and risk, says vaccinology expert Dr. Kathryn Edwards, professor of pediatrics in the division of infectious diseases at Vanderbilt University School of Medicine in Nashville, Tenn.
"We take safety very seriously," says Dr. Edwards. "We don't want something bad to happen, but we also realize that we have a terrible outbreak and we have a lot of people dying. We want to figure out how we can stop this."
In the U.S., all vaccine clinical trials have a data safety board of experts who monitor results for adverse reactions and red flags that should halt a study, notes Dr. Edwards. Any candidate that succeeds through safety and efficacy trials still requires review and approval by the Food and Drug Administration before a public launch.
Community vs. Individual
A major challenge to the deployment of a safe and effective coronavirus vaccine goes beyond the technical realm. A persistent all-out anti-vaccine sentiment has found a home and growing community on social media where conspiracies thrive. Main tenets of the movement are that vaccines are ineffective, unsafe and cause autism, despite abundant scientific evidence to the contrary.
Best-case scenario, more than one successful vaccine ascends with competing methods to achieve the same goal of preventing or lessening the severity of the COVID-19 virus.
In fact, widespread use of vaccines is considered by the U.S. Centers of Disease Control and Prevention to be one of the greatest public health achievements of the 20th Century. The World Health Organization estimates that between two million to three million deaths are avoided each year through immunization campaigns that employ vaccination to control life-threatening infectious diseases.
Most people reluctant to give their children vaccines, however, don't oppose them for everyone, but believe that they are a personal choice, says Reich.
"They think that vaccines are one strategy in personal health optimization, but they shouldn't be mandated for participation in any part of civil society," she says.
Vaccine hesitancy, like the teeter totter of social distancing acceptance, reflects the push and pull of individual versus community values, says Reich.
"A lot of people are saying, 'I take personal responsibility for my own health and I don't want a city or a county or state telling me what I should and shouldn't do,'" says Reich. "Then we also see calls for collective responsibility that says 'It's not your personal choice. This is about helping health systems function. This is about making sure vulnerable people are protected.'"
These same debates are likely to continue if a vaccine comes to market, she says.
Building Public Confidence
Reich offers solutions to address the conflict between embedded American norms and widespread embrace of an approved COVID-19 vaccine. Long-term goals: Stop blaming people when they get sick, treat illness as a community responsibility, make sick leave common for all workers, and improve public health systems.
"In the shorter run," says Reich, "health authorities and companies that might bring a vaccine to market need to work very hard to explain to the public why they should trust this vaccine and why they should use it."
The rush for a viable vaccine raises questions for consumers. To build public confidence, it's up to FDA reviewers, institutions and pharmaceutical companies to explain "what steps were skipped. What steps moved forward. How rigorous was safety testing. And to make that information clear to the public," says Reich.
Dr. Edwards says clinical trial timelines accelerated to test vaccines in humans make all the safeguards involved in the process that more compelling and important.
"There's no question we need a vaccine," she says. "But we also have to make sure that we don't harm people."
The Road Ahead
Think of manufacturing and distribution as key pitstops to keep the race for a vaccine on the road to the finish line. Both elements require substantial effort and consideration.
The speed of getting a vaccine to those who need it could hinge on the type of technology used to create it. Best-case scenario, more than one successful vaccine ascends with competing methods to achieve the same goal of preventing or lessening the severity of the COVID-19 virus.
Technological platforms fall into two basic camps, those that are proven and licensed for other viruses, and experimental approaches that may hold great promise but lack regulatory approval, says Maria Elena Bottazzi, co-director of Texas Children's Center for Vaccine Development at Baylor College of Medicine in Houston.
Moderna, for instance, employs an experimental technology called messenger RNA (mRNA) that has produced the encouraging early results in human safety trials, although some researchers criticized the company for not making the data public. The mRNA vaccine instructs cells to make copies of the key COVID-19 spike protein, with the goal of then triggering production of immune cells that can recognize and attack the virus if it ever invades the body.
"We were already seeing a lot of dissent around questions of individual freedoms and community responsibilities."
Scientists always look for ways to incorporate new technologies into drug development, says Bottazzi. On the other hand, the more basic and generic the technology, theoretically, the faster production could ramp up if a vaccine proves successful through all phases of clinical trials, she says.
"I don't want to develop a vaccine in my lab, but then I don't have anybody to hand it off to because my process is not suitable" for manufacturing or scalability, says Bottazzi.
Researchers at the Baylor lab hope to repurpose a shelved vaccine developed for the genetically similar SARS virus, with a strategy to leverage what is already known instead of "starting from scratch" to develop a COVID-19 vaccine. A recombinant protein technology similar to that used for an approved Hepatitis B vaccine lets scientists focus on identifying a suitable vaccine target without the added worry of a novel platform, says Bottazzi.
The Finish Line
If and when a COVID-19 vaccine is approved is anyone's guess. Announcing a plan to hasten vaccine development via a program dubbed Operation Warp Speed, President Trump said recently one could be available "hopefully" by the end of the year or early 2021.
Scientists urge caution, noting that safe vaccines can take 10 years or more to develop. If a rushed vaccine turns out to have safety and efficacy issues, that could add ammunition to the anti-vaccine lobby.
Emergence of a successful vaccine requires an "enormous effort" with many complex systems from the lab all the way to manufacturing enough capacity to handle a pandemic, says Bottazzi.
"At the same time, you're developing it, you're really carefully assessing its safety and ability to be effective," she says, so it's important "not to get discouraged" if it takes longer than a year or more.
To gauge if a vaccine works on a broad scale, it would have to be delivered into communities where the virus is active. There are examples in history of life-saving vaccines going first to people who could pay for them and not to those who needed them most, says Reich.
"Agencies are going to have to think about how those distribution decisions are going to be made and who is going to make them and that will go a certain way toward reassuring the public," says Reich.
A Gallup survey last year found that vaccine confidence, in general, remains high, with 86 percent of Americans believing that vaccines are safer than the diseases that they are designed to prevent. Still, recent news organization polls indicate that roughly 20 to 25 percent of Americans say they won't or are unlikely to get a COVID-19 vaccine if one becomes available.
Until the 1980s, every vaccine to hit the market was appreciated; a culture of questioning science didn't exist in the same way as today, notes Reich. Time passed and attitudes changed.
"We were already having robust arguments nationally about what counts as an expert, what's the role of the government in daily life," says Reich. "We were already seeing a lot of dissent around questions of individual freedoms and community responsibilities. COVID-19 did not create those conflicts, but they've definitely become more visible since we've moved into this pandemic."
Dr. May Edward Chinn, Kizzmekia Corbett, PhD., and Alice Ball, among others, have been behind some of the most important scientific work of the last century.
If you look back on the last century of scientific achievements, you might notice that most of the scientists we celebrate are overwhelmingly white, while scientists of color take a backseat. Since the Nobel Prize was introduced in 1901, for example, no black scientists have landed this prestigious award.
The work of black women scientists has gone unrecognized in particular. Their work uncredited and often stolen, black women have nevertheless contributed to some of the most important advancements of the last 100 years, from the polio vaccine to GPS.
Here are five black women who have changed science forever.
Dr. May Edward Chinn
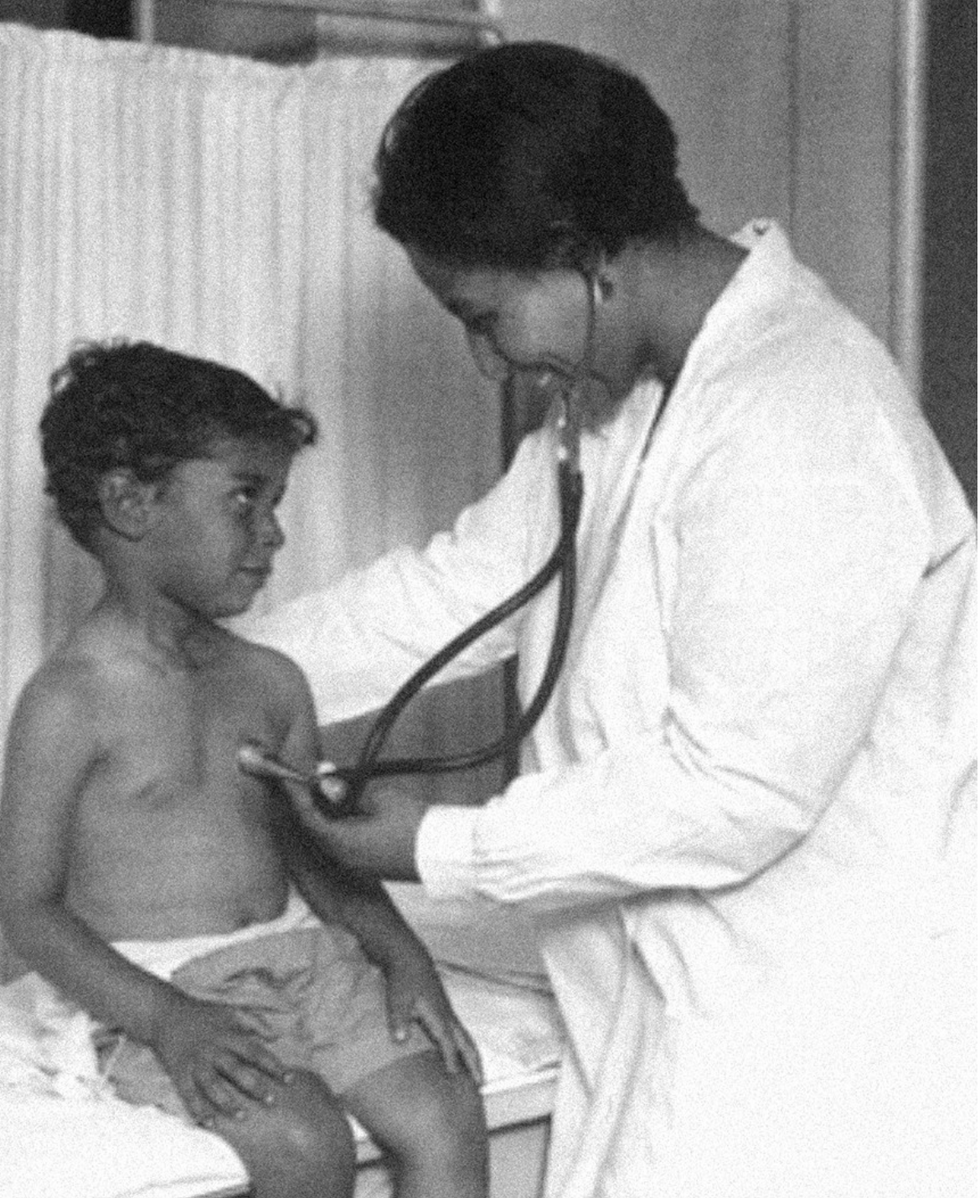
Dr. May Edward Chinn practicing medicine in Harlem
George B. Davis, PhD.
Chinn was born to poor parents in New York City just before the start of the 20th century. Although she showed great promise as a pianist, playing with the legendary musician Paul Robeson throughout the 1920s, she decided to study medicine instead. Chinn, like other black doctors of the time, were barred from studying or practicing in New York hospitals. So Chinn formed a private practice and made house calls, sometimes operating in patients’ living rooms, using an ironing board as a makeshift operating table.
Chinn worked among the city’s poor, and in doing this, started to notice her patients had late-stage cancers that often had gone undetected or untreated for years. To learn more about cancer and its prevention, Chinn begged information off white doctors who were willing to share with her, and even accompanied her patients to other clinic appointments in the city, claiming to be the family physician. Chinn took this information and integrated it into her own practice, creating guidelines for early cancer detection that were revolutionary at the time—for instance, checking patient health histories, checking family histories, performing routine pap smears, and screening patients for cancer even before they showed symptoms. For years, Chinn was the only black female doctor working in Harlem, and she continued to work closely with the poor and advocate for early cancer screenings until she retired at age 81.
Alice Ball

Pictorial Press Ltd/Alamy
Alice Ball was a chemist best known for her groundbreaking work on the development of the “Ball Method,” the first successful treatment for those suffering from leprosy during the early 20th century.
In 1916, while she was an undergraduate student at the University of Hawaii, Ball studied the effects of Chaulmoogra oil in treating leprosy. This oil was a well-established therapy in Asian countries, but it had such a foul taste and led to such unpleasant side effects that many patients refused to take it.
So Ball developed a method to isolate and extract the active compounds from Chaulmoogra oil to create an injectable medicine. This marked a significant breakthrough in leprosy treatment and became the standard of care for several decades afterward.
Unfortunately, Ball died before she could publish her results, and credit for this discovery was given to another scientist. One of her colleagues, however, was able to properly credit her in a publication in 1922.
Henrietta Lacks

onathan Newton/The Washington Post/Getty
The person who arguably contributed the most to scientific research in the last century, surprisingly, wasn’t even a scientist. Henrietta Lacks was a tobacco farmer and mother of five children who lived in Maryland during the 1940s. In 1951, Lacks visited Johns Hopkins Hospital where doctors found a cancerous tumor on her cervix. Before treating the tumor, the doctor who examined Lacks clipped two small samples of tissue from Lacks’ cervix without her knowledge or consent—something unthinkable today thanks to informed consent practices, but commonplace back then.
As Lacks underwent treatment for her cancer, her tissue samples made their way to the desk of George Otto Gey, a cancer researcher at Johns Hopkins. He noticed that unlike the other cell cultures that came into his lab, Lacks’ cells grew and multiplied instead of dying out. Lacks’ cells were “immortal,” meaning that because of a genetic defect, they were able to reproduce indefinitely as long as certain conditions were kept stable inside the lab.
Gey started shipping Lacks’ cells to other researchers across the globe, and scientists were thrilled to have an unlimited amount of sturdy human cells with which to experiment. Long after Lacks died of cervical cancer in 1951, her cells continued to multiply and scientists continued to use them to develop cancer treatments, to learn more about HIV/AIDS, to pioneer fertility treatments like in vitro fertilization, and to develop the polio vaccine. To this day, Lacks’ cells have saved an estimated 10 million lives, and her family is beginning to get the compensation and recognition that Henrietta deserved.
Dr. Gladys West

Andre West
Gladys West was a mathematician who helped invent something nearly everyone uses today. West started her career in the 1950s at the Naval Surface Warfare Center Dahlgren Division in Virginia, and took data from satellites to create a mathematical model of the Earth’s shape and gravitational field. This important work would lay the groundwork for the technology that would later become the Global Positioning System, or GPS. West’s work was not widely recognized until she was honored by the US Air Force in 2018.
Dr. Kizzmekia "Kizzy" Corbett

TIME Magazine
At just 35 years old, immunologist Kizzmekia “Kizzy” Corbett has already made history. A viral immunologist by training, Corbett studied coronaviruses at the National Institutes of Health (NIH) and researched possible vaccines for coronaviruses such as SARS (Severe Acute Respiratory Syndrome) and MERS (Middle East Respiratory Syndrome).
At the start of the COVID pandemic, Corbett and her team at the NIH partnered with pharmaceutical giant Moderna to develop an mRNA-based vaccine against the virus. Corbett’s previous work with mRNA and coronaviruses was vital in developing the vaccine, which became one of the first to be authorized for emergency use in the United States. The vaccine, along with others, is responsible for saving an estimated 14 million lives.On today’s episode of Making Sense of Science, I’m honored to be joined by Dr. Paul Song, a physician, oncologist, progressive activist and biotech chief medical officer. Through his company, NKGen Biotech, Dr. Song is leveraging the power of patients’ own immune systems by supercharging the body’s natural killer cells to make new treatments for Alzheimer’s and cancer.
Whereas other treatments for Alzheimer’s focus directly on reducing the build-up of proteins in the brain such as amyloid and tau in patients will mild cognitive impairment, NKGen is seeking to help patients that much of the rest of the medical community has written off as hopeless cases, those with late stage Alzheimer’s. And in small studies, NKGen has shown remarkable results, even improvement in the symptoms of people with these very progressed forms of Alzheimer’s, above and beyond slowing down the disease.
In the realm of cancer, Dr. Song is similarly setting his sights on another group of patients for whom treatment options are few and far between: people with solid tumors. Whereas some gradual progress has been made in treating blood cancers such as certain leukemias in past few decades, solid tumors have been even more of a challenge. But Dr. Song’s approach of using natural killer cells to treat solid tumors is promising. You may have heard of CAR-T, which uses genetic engineering to introduce cells into the body that have a particular function to help treat a disease. NKGen focuses on other means to enhance the 40 plus receptors of natural killer cells, making them more receptive and sensitive to picking out cancer cells.
Paul Y. Song, MD is currently CEO and Vice Chairman of NKGen Biotech. Dr. Song’s last clinical role was Asst. Professor at the Samuel Oschin Cancer Center at Cedars Sinai Medical Center.
Dr. Song served as the very first visiting fellow on healthcare policy in the California Department of Insurance in 2013. He is currently on the advisory board of the Pritzker School of Molecular Engineering at the University of Chicago and a board member of Mercy Corps, The Center for Health and Democracy, and Gideon’s Promise.
Dr. Song graduated with honors from the University of Chicago and received his MD from George Washington University. He completed his residency in radiation oncology at the University of Chicago where he served as Chief Resident and did a brachytherapy fellowship at the Institute Gustave Roussy in Villejuif, France. He was also awarded an ASTRO research fellowship in 1995 for his research in radiation inducible gene therapy.
With Dr. Song’s leadership, NKGen Biotech’s work on natural killer cells represents cutting-edge science leading to key findings and important pieces of the puzzle for treating two of humanity’s most intractable diseases.
Show links
- Paul Song LinkedIn
- NKGen Biotech on Twitter - @NKGenBiotech
- NKGen Website: https://nkgenbiotech.com/
- NKGen appoints Paul Song
- Patient Story: https://pix11.com/news/local-news/long-island/promising-new-treatment-for-advanced-alzheimers-patients/
- FDA Clearance: https://nkgenbiotech.com/nkgen-biotech-receives-ind-clearance-from-fda-for-snk02-allogeneic-natural-killer-cell-therapy-for-solid-tumors/Q3 earnings data: https://www.nasdaq.com/press-release/nkgen-biotech-inc.-reports-third-quarter-2023-financial-results-and-business