Why Are Autism Rates Steadily Rising?

Stefania Sterling with her son Charlie, who was diagnosed at age 3 with autism.
Stefania Sterling was just 21 when she had her son, Charlie. She was young and healthy, with no genetic issues apparent in either her or her husband's family, so she expected Charlie to be typical.
"It is surprising that the prevalence of a significant disorder like autism has risen so consistently over a relatively brief period."
It wasn't until she went to a Mommy and Me music class when he was one, and she saw all the other one-year-olds walking, that she realized how different her son was. He could barely crawl, didn't speak, and made no eye contact. By the time he was three, he was diagnosed as being on the lower functioning end of the autism spectrum.
She isn't sure why it happened – and researchers, too, are still trying to understand the basis of the complex condition. Studies suggest that genes can act together with influences from the environment to affect development in ways that lead to Autism Spectrum Disorder (ASD). But rates of ASD are rising dramatically, making the need to figure out why it's happening all the more urgent.
The Latest News
Indeed, the CDC's latest autism report, released last week, which uses 2016 data, found that the prevalence of ASD in four-year-old children was one in 64 children, or 15.6 affected children per 1,000. That's more than the 14.1 rate they found in 2014, for the 11 states included in the study. New Jersey, as in years past, was the highest, with 25.3 per 1,000, compared to Missouri, which had just 8.8 per 1,000.
The rate for eight-year-olds had risen as well. Researchers found the ASD prevalence nationwide was 18.5 per 1,000, or one in 54, about 10 percent higher than the 16.8 rate found in 2014. New Jersey, again, was the highest, at one in 32 kids, compared to Colorado, which had the lowest rate, at one in 76 kids. For New Jersey, that's a 175 percent rise from the baseline number taken in 2000, when the state had just one in 101 kids.
"It is surprising that the prevalence of a significant disorder like autism has risen so consistently over a relatively brief period," said Walter Zahorodny, an associate professor of pediatrics at Rutgers New Jersey Medical School, who was involved in collecting the data.
The study echoed the findings of a surprising 2011 study in South Korea that found 1 in every 38 students had ASD. That was the the first comprehensive study of autism prevalence using a total population sample: A team of investigators from the U.S., South Korea, and Canada looked at 55,000 children ages 7 to 12 living in a community in South Korea and found that 2.64 percent of them had some level of autism.
Searching for Answers
Scientists can't put their finger on why rates are rising. Some say it's better diagnosis. That is, it's not that more people have autism. It's that we're better at detecting it. Others attribute it to changes in the diagnostic criteria. Specifically, the May 2013 update of the Diagnostic and Statistical Manual of Mental Disorders-5 -- the standard classification of mental disorders -- removed the communication deficit from the autism definition, which made more children fall under that category. Cynical observers believe physicians and therapists are handing out the diagnosis more freely to allow access to services available only to children with autism, but that are also effective for other children.
Alycia Halladay, chief science officer for the Autism Science Foundation in New York, said she wishes there were just one answer, but there's not. While she believes the rising ASD numbers are due in part to factors like better diagnosis and a change in the definition, she does not believe that accounts for the entire rise in prevalence. As for the high numbers in New Jersey, she said the state has always had a higher prevalence of autism compared to other states. It is also one of the few states that does a good job at recording cases of autism in its educational records, meaning that children in New Jersey are more likely to be counted compared to kids in other states.
"Not every state is as good as New Jersey," she said. "That accounts for some of the difference compared to elsewhere, but we don't know if it's all of the difference in prevalence, or most of it, or what."
"What we do know is that vaccinations do not cause autism."
There is simply no defined proven reason for these increases, said Scott Badesch, outgoing president and CEO of the Autism Society of America.
"There are suggestions that it is based on better diagnosis, but there are also suggestions that the incidence of autism is in fact increasing due to reasons that have yet been determined," he said, adding, "What we do know is that vaccinations do not cause autism."
Zahorodny, the pediatrics professor, believes something is going on beyond better detection or evolving definitions.
"Changes in awareness and shifts in how children are identified or diagnosed are relevant, but they only take you so far in accounting for an increase of this magnitude," he said. "We don't know what is driving the surge in autism recorded by the ADDM Network and others."
He suggested that the increase in prevalence could be due to non-genetic environmental triggers or risk factors we do not yet know about, citing possibilities including parental age, prematurity, low birth rate, multiplicity, breech presentation, or C-section delivery. It may not be one, but rather several factors combined, he said.
"Increases in ASD prevalence have affected the whole population, so the triggers or risks must be very widely dispersed across all strata," he added.
There are studies that find new risk factors for ASD almost on a daily basis, said Idan Menashe, assistant professor in the Department of Health at Ben-Gurion University of the Negev, the fastest growing research university in Israel.
"There are plenty of studies that find new genetic variants (and new genes)," he said. In addition, various prenatal and perinatal risk factors are associated with a risk of ASD. He cited a study his university conducted last year on the relationship between C-section births and ASD, which found that exposure to general anesthesia may explain the association.
Whatever the cause, health practitioners are seeing the consequences in real time.
"People say rates are higher because of the changes in the diagnostic criteria," said Dr. Roseann Capanna-Hodge, a psychologist in Ridgefield, CT. "And they say it's easier for children to get identified. I say that's not the truth and that I've been doing this for 30 years, and that even 10 years ago, I did not see the level of autism that I do see today."
Sure, we're better at detecting autism, she added, but the detection improvements have largely occurred at the low- to mid- level part of the spectrum. The higher rates of autism are occurring at the more severe end, in her experience.
A Polarizing Theory
Among the more controversial risk factors scientists are exploring is the role environmental toxins may play in the development of autism. Some scientists, doctors and mental health experts suspect that toxins like heavy metals, pesticides, chemicals, or pollution may interrupt the way genes are expressed or the way endocrine systems function, manifesting in symptoms of autism. But others firmly resist such claims, at least until more evidence comes forth. To date, studies have been mixed and many have been more associative than causative.
"Today, scientists are still trying to figure out whether there are other environmental changes that can explain this rise, but studies of this question didn't provide any conclusive answer," said Menashe, who also serves as the scientific director of the National Autism Research Center at BGU.
"It's not everything that makes Charlie. He's just like any other kid."
That inconclusiveness has not dissuaded some doctors from taking the perspective that toxins do play a role. "Autism rates are rising because there is a mismatch between our genes and our environment," said Julia Getzelman, a pediatrician in San Francisco. "The majority of our evolution didn't include the kinds of toxic hits we are experiencing. The planet has changed drastically in just the last 75 years –- it has become more and more polluted with tens of thousands of unregulated chemicals being used by industry that are having effects on our most vulnerable."
She cites BPA, an industrial chemical that has been used since the 1960s to make certain plastics and resins. A large body of research, she says, has shown its impact on human health and the endocrine system. BPA binds to our own hormone receptors, so it may negatively impact the thyroid and brain. A study in 2015 was the first to identify a link between BPA and some children with autism, but the relationship was associative, not causative. Meanwhile, the Food and Drug Administration maintains that BPA is safe at the current levels occurring in food, based on its ongoing review of the available scientific evidence.
Michael Mooney, President of St. Louis-based Delta Genesis, a non-profit organization that treats children struggling with neurodevelopmental delays like autism, suspects a strong role for epigenetics, which refers to changes in how genes are expressed as a result of environmental influences, lifestyle behaviors, age, or disease states.
He believes some children are genetically predisposed to the disorder, and some unknown influence or combination of influences pushes them over the edge, triggering epigenetic changes that result in symptoms of autism.
For Stefania Sterling, it doesn't really matter how or why she had an autistic child. That's only one part of Charlie.
"It's not everything that makes Charlie," she said. "He's just like any other kid. He comes with happy moments. He comes with sad moments. Just like my other three kids."
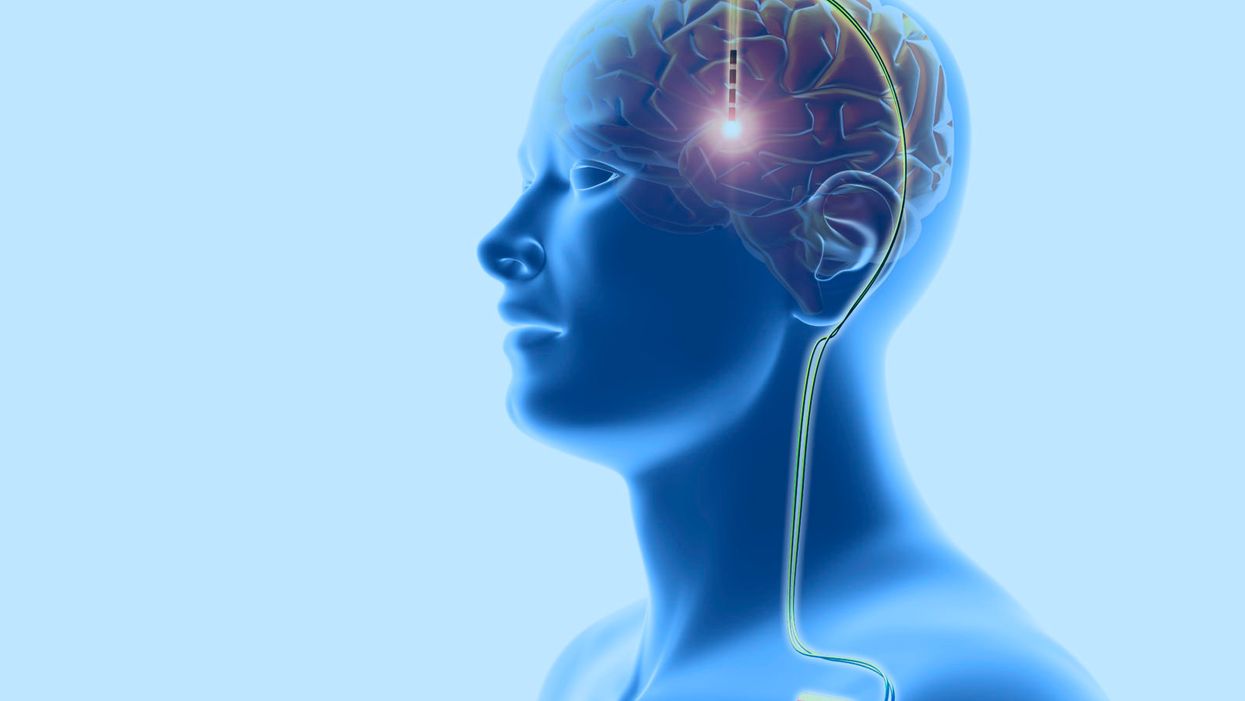
Deep Brain Stimulation for Mental Illnesses Raises Ethical Concerns
Deep brain stimulation: This neurosurgical treatment involves the implantation of electrodes in the cerebral lobes of the brain, linked through the scalp (top) to wires (down right) leading to a battery implanted below the skin. This sends electrical impulses to specific areas of the brain. DBS was developed for the treatment of Parkinson's disease, but is being investigated for use in other conditions.
Imagine that you are one of the hundreds of millions of people who suffer from depression. Medication hasn't helped you, so you're looking for another treatment option. Something powerful enough to change your mood as soon as you need a lift.
"If a participant experiences a personality change, does this change who they are or dehumanize them by altering their nature?"
Enter deep brain stimulation: a type of therapy in which one or more electrodes are inserted into your brain and connected to a surgically implanted, battery-operated medical device in your chest. This device, which is approximately the size of a stopwatch, sends electric pulses to a targeted region of your brain. The idea is to control a variety of neurological symptoms that can't be adequately managed by drugs.
Over the last twenty years, deep brain stimulation, known as DBS, has become an efficient and safe alternative for the treatment of chronic neurological diseases such as epilepsy, Parkinson's disease and neuropathic pain. According to the International Neuromodulation Society, there have been more than 80,000 deep brain stimulation implants performed around the world.
The Food and Drug Administration approved DBS as a treatment for essential tremor and Parkinson's in 1997, dystonia in 2003 and obsessive compulsive disorder in 2009. Since doctors can use drugs and treatments "off-label" (not approved by the FDA) to treat patients with any disease, DBS is now also being investigated as a treatment for chronic pain, PTSD and major depression.
And these new applications are raising profound ethical questions about individuality, personality, and even what it means to be human.
"These patients are essentially having a computer that can modify and influence emotional processing, mood and motor outputs inserted into the brain," said Gabriel Lazaro-Munoz, an assistant professor at The Center for Medical Ethics and Health Policy at Baylor College of Medicine. "These responses define us as human beings and dictate our autonomy. If a participant experiences a personality change, does this change who they are or dehumanize them by altering their nature? These are some of the questions we have to consider."
"When we are not in control of ourselves, are we ourselves?"
The U.S. government has similar concerns about DBS. The National Institutes of Health recently awarded grants to study the neuroethical issues surrounding the use of DBS in neuropsychiatric and movement disorders and appropriate consent for brain research. The grants are part of the Brain Research through Advancing Innovative Neurotechnologies (BRAIN) Initiative. Walter Koroshetz, director of NIH's National Institute of Neurological Disorders and Stroke said, "Neuroscience is rapidly moving toward a new frontier of research on human brains that may have long-lasting and unforeseen effects. These new awards signal our commitment to research conducted in a responsible way as to anticipate all potential consequences, and to ensure that research subjects have a clear understanding of the potential benefits and risks of participating in studies."
Dr. Lazaro-Munoz's Center was awarded one of the grants to identify and evaluate the ethical, legal and social concerns with adaptive deep brain stimulation (aDBS) technologies. Adaptive DBS is a relatively new version of the technology that enables recording of brain cell activity that is then used to regulate the brain in real time. He and his team will closely observe researchers conducting aDBS studies and administering in-depth interviews to trial participants, their caregivers, and researchers, as well as individuals who declined to participate in such studies. The goal is to gain a better understanding of the ethical concerns at stake in order to guide responsible research.
Dr. Lazaro-Munoz said one of the concerns is dehumanization. "By using this technology are we compromising what makes us human? When we are not in control of ourselves, are we ourselves?" He notes that similar concerns were raised about pharmaceutical treatments for illnesses. "Both change behaviors and emotional processing. However, there is a difference. Culturally we are more used to using drugs, not implanting devices into brain and computer interfaces. Many people think of it as science fiction."
The changes in behavior due to DBS can be dramatic, perhaps none more so than with Parkinson's disease; patients may see their chronic tremors suddenly vanish.
Pills for OCD and depression take longer than DBS to see significant improvement, sometimes months. "A DBS device is either on or off. And patients and families see changes immediately," Dr. Lazaro-Munoz said. "Family members are often startled by these changes, as are the patients." He's observed that patients feel more in control with pills because they can alter and "play" with the dose or even skip a dose.
The changes in behavior due to DBS can be dramatic, perhaps none more so than with Parkinson's disease; patients may see their chronic tremors suddenly vanish, like in this must-see video.
But surgical procedures to treat motor symptoms are also increasingly being implicated as a cause of behavioral changes, both positive and negative, in patients with Parkinson's. The personality changes reported in patients who undergo DBS include hypermania, pathological gambling, hypersexuality, impulsivity and aggressiveness. One patient who suffered from OCD fell in love with the music of Johnny Cash when his brain was stimulated. On the positive side, patients report memory enhancement.
One patient who is pleased with DBS is Greg Barstead, who was diagnosed with Parkinson's in 2003, when he was the president of Colonial Penn Life Insurance Company. He also has dystonia, which affects his neck and shoulders. Barstead said that DBS has been helpful for a range of symptoms: "My shoulder is a lot less stiff and my neck hurts less. And my tremors are under control. It is not perfect, as it doesn't relieve all the Parkinson's symptoms, but it does enough of a good job that both my wife and I are very happy I had DBS."
"We are not exactly sure what part of the brain causes depression. Doctors have not identified where to implant the device."
He said he hasn't noticed any personality changes, but noted that the disease itself can cause such changes. In fact, studies have shown that it can cause many psychiatric problems including depression and hallucinations. And, approximately a third of Parkinson's patients develop dementia.
Arthur L. Caplan, founding head of the Division of Medical Ethics at NYU School of Medicine, notes that unlike psychosurgery, DBS can be turned on and off and the device can be removed. "There are less ethical concerns around treating patients with Parkinson's disease than other illnesses because surgeons know exactly where to implant the device and have many years of experience with it," he said, adding that he is concerned about using DBS for other illnesses, such as depression. "We are not exactly sure what part of the brain causes depression. Doctors have not identified where to implant the device. And I would certainly not advocate its use in patients with mild depression."
Dr. Lazaro-Munoz said of the personality changes possible with DBS, physicians need to consider how the patients were functioning without it. "Patients who are candidates for DBS typically used many medications as well as psychotherapy before opting for DBS," he explained. "To me, the question is what is the net result of using this technology? Does the patient have regrets? Are the changes in personality significant or not? Although most DBS patients report being happy they underwent the procedure, some say they don't feel like themselves after DBS. Others feel they are more like themselves, especially if there are dramatic improvements in movement problems or relief of OCD symptoms."
And then there is the question of money. The costs of DBS are covered by most insurance companies and Medicare only for FDA-approved targets like Parkinson's. Off-label uses are not covered, at least for now.
Caplan reminds people that DBS devices are manufactured by companies that are interested in making money and the average cost per treatment is around $50,000. "I am interested in seeing DBS move forward," he said. "But we must be careful and not allow industry to make it go too fast, or be used on too many people, before we know it is effective."
Why Aren’t Gene Editing Treatments Available Yet For People With Genetic Disorders? 
Lynn Julian Crisci, a 40-year-old actress with Ehlers-Danlos syndrome, is eager for gene editing treatments; she is pictured here with her service dog, a Maltese named Lil Stinker.
Lynn Julian Crisci, 40, is an actress, a singer-songwriter, and an ambassador for the U.S. Pain Foundation. She is also a Boston Marathon bombing survivor. Crisci has a genetic disorder called Ehlers-Danlos syndrome (EDS), which has magnified the impact of the traumatic brain injury she sustained as a result of the attack that occurred almost five years ago. Having EDS means that her brain tissue is weaker and more prone to injury.
"I would love to learn more about gene editing and the possibilities of using it to lessen the symptoms of EDS, or cure it completely."
"EDS is a genetic tissue disorder that forces the body to make defective collagen," Crisci told LeapsMag. Since collagen is the main component of connective tissue (bones, blood vessels, the gastrointestinal tract, skin, cartilage, etc.), and is the most abundant protein in mammals, EDS can affect virtually every part of the body. "This results in widespread joint pain, usually due to hypermobility, sometimes along with digestive issues such as inflammatory bowel disease, and prolapsed organs."
If life was difficult with Ehlers-Danlos syndrome alone, the addition of the brain injury has made Crisci's life feel unbearable at times. Amidst her week's back-to-back doctor's visits, Crisci said that she would "love to learn more about gene editing and the possibilities of using it to lessen the symptoms of Ehlers-Danlos syndrome, or cure it completely."
With all of the excitement these days around CRISPR, a precise and efficient way to edit DNA that has taken the world by storm, such treatments seem tantalizingly within reach. But is it fair to present the hope of such cures to those with life-limiting genetic disorders?
"From the experience that we've had from gene therapy — we're 20, almost 30 years past some of the initial gene therapy stuff — and there's still not a huge number of applications for it," said Scott Weissman, founder of Chicago Genetic Consultants, a company that provides genetic counseling services to patients. "Unfortunately, we have to wait and see if this is something that's truly viable, or if it's really just hype."
"I expect five years from now we'll look back and say, 'Wow, we were just scratching the surface.'"
Defining Our Terms
The terms "gene therapy" and "gene editing" are often used interchangeably, but not everyone agrees with this usage.
According to Editas Medicine, a leader in CRISPR technology, gene therapy involves the transfer of a new gene into a patient's cells to augment a defective gene, instead of using drugs or surgery to treat a condition. After a teenager's death in 1999 effectively shut down gene therapy research in the U.S., subsequent studies helped the field make a comeback, and the first such treatment for an inherited disease was approved by the FDA just a few weeks ago, for a rare form of vision loss. Called Luxturna, it is for treatment of patients with RPE65-mediated inherited retinal disease (IRD).
Since those with RPE65-mediated IRD typically become blind in childhood and have no pharmacologic treatment options, the FDA's approval of Luxturna is "a significant moment for patients," said Jeffrey Marrazzo, the chief executive officer of the company behind the product, Spark Therapeutics. Two other gene therapy treatments were also approved in the last five months, both for specific cancers.
Gene editing, on the other hand, refers to a group of technologies that enables scientists to precisely and directly change an organism's genes by adding, removing, or altering particular segments of DNA. Gene editing tools include Zinc Finger Nucleases (ZFNs), Transcription Activator-Like Effector Nucleases (TALENs), and CRISPR/Cas9. The first treatment using ZFNs happened in November in California, when a 44-year-old man with a metabolic ailment called Hunter syndrome was injected with gene editing tools. Results are not yet known.

Dr. David Valle, director of the Institute of Genetic Medicine at Johns Hopkins, said that gene therapy's "significant therapeutic misadventures" have actually been beneficial. They've helped us learn to "be rigorous in our thinking about what we can do and what we can't do with CRISPR" and other gene editing tools.
"It appears like we are really beginning to have, for the first time, some meaningful and good results from gene therapy — it's moving into the clinic now in a meaningful way," Valle said. "I expect five years from now we'll look back and say, 'Wow, we were just at this point in 2017 — we were just scratching the surface.'"
Over 2300 gene therapy clinical trials are planned, ongoing, or have been completed so far. As for gene editing, no treatments are commercially available anywhere in the world. The expectation, however, is that many treatments that are "currently in or soon to enter clinical trials will come up for approval in coming years," according to a November 2016 report by the American Society of Gene & Cell Therapy.
CRISPR Therapeutics of Cambridge, Massachusetts will begin a European gene editing trial this year, with the hopes of creating a treatment for beta thalassemia, an inherited blood disorder. The company will also request approval from the FDA to begin a clinical trial using CRISPR for sickle-cell disease. And Stanford University School of Medicine researchers are planning a similar CRISPR clinical trial for sickle-cell disease. They hope to begin their trial in 2019.
Jim Burns, the president and chief executive officer of Casebia Therapeutics, told Leapsmag that the company will start animal research this year using CRISPR to treat autoimmune diseases, hemophilia A, and retinal diseases. They expect to begin clinical research in humans in 2019 or 2020. [Disclosure: Casebia Therapeutics is a novel joint venture between CRISPR Therapeutics and Leapsmag's founder, Leaps by Bayer, though Leapsmag is editorially independent of Bayer.]
Efforts are well underway to take genome-targeted treatments from the scientist's bench to the patient's bedside.
The Technology Isn't There Yet
Unlike germline gene editing — when egg and sperm cell DNA is edited in an embryo — somatic cell gene editing in adults is not very controversial, because the edits are not heritable. Since somatic cells contribute to the various tissues of the body but not to eggs or sperm cells, changes made to somatic cells are limited to the treated individual.
The number one reason that gene therapy and gene editing treatments are not yet widely available to the adult population is that the technology is not advanced enough. But it's getting there. Efforts are well underway to take genome-targeted treatments from the scientist's bench to the patient's bedside — especially in the case of monogenic diseases.
Roughly 10,000 genetic illnesses are monogenic, meaning that they result from a disease-causing variant in a single gene. Some monogenic diseases that have gene editing treatments currently in development for use in clinical trials include cystic fibrosis, Huntington's disease, Tay-Sachs disease, and sickle cell anemia.
Marrazzo of Spark Therapeutics told LeapsMag that his company is working on gene therapies for monogenic diseases that affect the eye, like the retinal disease that Luxturna targets, as well as neurodegenerative and liver diseases.
But most illnesses are polygenic, meaning that they result from multiple gene mutations that have a combined influence on disease progression. Polygenic diseases, like high blood pressure and diabetes, would therefore be more challenging to treat with genome-targeted interventions. As a result, most research is currently focused on monogenic diseases.
"We don't really know how to target the gene editing to a specific organ in the body once it's fully developed and matured."
A major hurdle of gene editing is the risk of off-target effects. Editing the genome "can have unpredictable effects on gene expression and unintended effects on neighboring genes," wrote Morgan Maeder and Charles Gersbach in a March 2016 article in Molecular Therapy. One such unintended effect is the development of leukemia when a new gene unintentionally activates a cancer gene.
And since there are roughly 37 trillion cells in the adult human body, getting the gene editing machinery to enough cells or target tissues to create a lasting and significant change is a daunting task.
"We don't really know how to target the gene editing to a specific organ in the body once it's fully developed and matured," said Weissman, the genetic counseling expert. If you take an adult patient with known BRCA1 or BRCA2 mutations, for example, how do you then "get the [gene editing] system in the breast so that it accurately cuts out the mutation in every single breast cell that could potentially turn into breast cancer, or in every single ovarian cell that could turn into ovarian cancer? We don't know how to target it like that, and I think that's the biggest reason you're not seeing it more somatically at this point in time."
Approval and Access
Debra Mathews, assistant director for science programs for the Johns Hopkins Berman Institute of Bioethics, told LeapsMag that pre-existing regulatory frameworks surrounding gene therapy have been sufficient for addressing ethical and regulatory concerns surrounding gene editing. A bigger concern, she said, centers around access to future genome-targeted treatments.
"We know more about the genetics of Caucasian populations than other populations," Mathews explained, due to how genomic data is gathered. This "could lead to problems not just of financial but of biological access to new therapies." In other words, she said, "if you're of European ancestry, there may be a greater chance that there's a relevant genetically-targeted therapy for you than if you're of non-European ancestry."
Ensuring that genome-targeted treatments are accessible to all will require increased cooperation and data-sharing among key stakeholders around the world, as well as increased public engagement that is inclusive of a wide range of voices.
"It's important to be realistic in our predictions to the public."
The Coming Wave of Gene Editing Treatments
Ehlers-Danlos syndrome alone has 13 monogenic subtypes, each with its own genetic basis and set of clinical criteria. Though several of the gene mutations causing EDS subtypes have been identified, the genetic basis for the most common subtype that Lynn Julian Crisci has — hypermobile EDS — remains unknown. What this means, according to Valle, the doctor from Johns Hopkins, is that a gene therapy or gene editing approach "really cannot be contemplated because we don't know what we're trying to fix" yet. This is the case for many genetic illnesses.
Efforts are ongoing in gene discovery by organizations such as the Baylor-Hopkins Center for Mendelian Genomics, of which Valle is the principal investigator. "Our objective," he said, "is to identify the genes and variants responsible" in monogenic disorders.
While Valle is optimistic about the coming wave of commercially available gene therapy and gene editing treatments, he also thinks that "it's important to be realistic in our predictions to the public." As eager as physicians are to offer cures to their patients, "we have to make sure that we're rigorous in our thinking and our ideas are well-buttressed with results."
Estimates vary for how long Crisci and others with genetic illnesses will have to wait for genome-targeted treatment options. Depending on the illness, viable gene editing treatments could hit the market within the next ten years. Though patients have already waited a long while, the revolutionary technology allowing us to fix nature's mistakes could make up for lost time and lost hope.