An Astounding Treatment at an Astounding Price: Who Gets to Benefit?

Tony and Kelly Mantoan, with their boys Teddy and Fulton, who both suffer from SMA, a genetic disorder that makes walking, swallowing, and breathing progressively difficult.
Kelly Mantoan was nursing her newborn son, Teddy, in the NICU in a Philadelphia hospital when her doctor came in and silently laid a hand on her shoulder. Immediately, Kelly knew what the gesture meant and started to sob: Teddy, like his one-year-old brother, Fulton, had just tested positive for a neuromuscular condition called spinal muscular atrophy (SMA).
The boys were 8 and 10 when Kelly heard about an experimental new treatment, still being tested in clinical trials, called Spinraza.
"We knew that [SMA] was a genetic disorder, and we knew that we had a 1 in 4 chance of Teddy having SMA," Mantoan recalls. But the idea of having two children with the same severe disability seemed too unfair for Kelly and her husband, Tony, to imagine. "We had lots of well-meaning friends tell us, well, God won't do this to you twice," she says. Except that He, or a cruel trick of nature, had.
In part, the boys' diagnoses were so devastating because there was little that could be done at the time, back in 2009 and 2010, when the boys were diagnosed. Affecting an estimated 1 in 11,000 babies, SMA is a degenerative disease in which the body is deficient in survival motor neuron (SMN) protein, thanks to a genetic mutation or absence of the body's SNM1 gene. So muscles that control voluntary movement – such as walking, breathing, and swallowing – weaken and eventually cease to function altogether.
Babies diagnosed with SMA Type 1 rarely live past toddlerhood, while people diagnosed with SMA Types 2, 3, and 4 can live into adulthood, usually with assistance like ventilators and feeding tubes. Shortly after birth, both Teddy Mantoan and his brother, Fulton, were diagnosed with SMA Type 2.
The boys were 8 and 10 when Kelly heard about an experimental new treatment, still being tested in clinical trials, called Spinraza. Up until then, physical therapy was the only sanctioned treatment for SMA, and Kelly enrolled both her boys in weekly sessions to preserve some of their muscle strength as the disease marched forward. But Spinraza – a grueling regimen of lumbar punctures and injections designed to stimulate a backup survival motor neuron gene to produce more SMN protein – offered new hope.
In clinical trials, after just a few doses of Spinraza, babies with SMA Type 1 began meeting normal developmental milestones – holding up their heads, rolling over, and sitting up. In other trials, Spinraza treatment delayed the need for permanent ventilation, while patients on the placebo arm continued to lose function, and several died. Spinraza was such a success, and so well tolerated among patients, that clinical trials ended early and the drug was fast-tracked for FDA approval in 2016. In January 2017, when Kelly got the call that Fulton and Teddy had been approved by the hospital to start Spinraza infusions, Kelly dropped to her knees in the middle of the kitchen and screamed.
Spinraza, manufactured by Biogen, has been hailed as revolutionary, but it's also not without drawbacks: Priced per injection, just one dose of Spinraza costs $125,000, making it one of the most expensive drugs on the global market. What's worse, treatment requires a "loading dose" of four injections over a four-week period, and then periodic injections every four months, indefinitely. For the first year of treatment, Spinraza treatment costs $750,000 – and then $375,000 for every year thereafter.
Last week, a competitive treatment for SMA Type 1 manufactured by Novartis burst onto the market. The new treatment, called Zolgensma, is a one-time gene therapy intended to be given to infants and is currently priced at $2.125 million, or $425,000 annually for five years, making it the most expensive drug in the world. Like Spinraza, Zolgensma is currently raising challenging questions about how insurers and government payers like Medicaid will be able to afford these treatments without bankrupting an already-strained health care system.
To Biogen's credit, the company provides financial aid for Spinraza patients with private insurance who pay co-pays for treatment, as well as for those who have been denied by Medicaid and Medicare. But getting insurance companies to agree to pay for Spinraza can often be an ordeal in itself. Although Fulton and Teddy Mantoan were approved for treatment over two years ago, a lengthy insurance battle delayed treatment for another eight months – time that, for some SMA patients, can mean a significant loss of muscular function.
Kelly didn't notice anything in either boy – positive or negative – for the first few months of Spinraza injections. But one day in November 2017, as Teddy was lowered off his school bus in his wheelchair, he turned to say goodbye to his friends and "dab," – a dance move where one's arms are extended briefly across the chest and in the air. Normally, Teddy would dab by throwing his arms up in the air with momentum, striking a pose quickly before they fell down limp at his sides. But that day, Teddy held his arms rigid in the air. His classmates, along with Kelly, were stunned. "Teddy, look at your arms!" Kelly remembers shrieking. "You're holding them up – you're dabbing!"

Teddy and Fulton Mantoan, who both suffer from spinal muscular atrophy, have seen life-changing results from Spinraza.
(Courtesy of Kelly Mantoan)
Not long after Teddy's dab, the Mantoans started seeing changes in Fulton as well. "With Fulton, we realized suddenly that he was no longer choking on his food during meals," Kelly said. "Almost every meal we'd have to stop and have him take a sip of water and make him slow down and take small bites so he wouldn't choke. But then we realized we hadn't had to do that in a long time. The nurses at school were like, 'it's not an issue anymore.'"
For the Mantoans, this was an enormous relief: Less choking meant less chance of aspiration pneumonia, a leading cause of death for people with SMA Types 1 and 2.
While Spinraza has been life-changing for the Mantoans, it remains painfully out of reach for many others. Thanks to Spinraza's enormous price tag, the threshold for who gets to use it is incredibly high: Adult and pediatric patients, particularly those with state-sponsored insurance, have reported multiple insurance denials, lengthy appeals processes, and endless bureaucracy from insurance and hospitals alike that stand in the way of treatment.
Kate Saldana, a 21-year-old woman with Type 2 SMA, is one of the many adult patients who have been lobbying for the drug. Saldana, who uses a ventilator 20 hours each day, says that Medicaid denied her Spinraza treatments because they mistakenly believed that she used a ventilator full-time. Saldana is currently in the process of appealing their decision, but knows she is fighting an uphill battle.

Kate Saldana, who suffers from Type 2 SMA, has been fighting unsuccessfully for Medicaid to cover Spinraza.
(Courtesy of Saldana)
"Originally, the treatments were studied and created for infants and children," Saldana said in an e-mail. "There is a plethora of data to support the effectiveness of Spinraza in those groups, but in adults it has not been studied as much. That makes it more difficult for insurance to approve it, because they are not sure if it will be as beneficial."
Saldana has been pursuing treatment unsuccessfully since last August – but others, like Kimberly Hill, a 32-year-old with SMA Type 2, have been waiting even longer. Hill, who lives in Oklahoma, has been fighting for treatment since Spinraza went on the U.S. market in December 2016. Because her mobility is limited to the use of her left thumb, Hill is eager to try anything that will enable her to keep working and finish a Master's degree in Fire and Emergency Management.
"Obviously, my family and I were elated with the approval of Spinraza," Hill said in an e-mail. "We thought I would finally have the chance to get a little stronger and healthier." But with Medicare and Medicaid, coverage and eligibility varies wildly by state. Earlier this year, Medicaid approved Spinraza for adult patients only if a clawback clause was attached to the approval, meaning that under certain conditions the Medicaid funds would need to be paid back. Because of the clawback clause, hospitals have been reluctant to take on Spinraza treatments, effectively barring adult Medicaid patients from accessing the drug altogether.
Hill's hospital is currently in negotiations with Medicaid to move forward with Spinraza treatment, but in the meantime, Hill is in limbo. "We keep being told there is nothing we can do, and we are devastated," Hill said.
"I felt extremely sad and honestly a bit forgotten, like adults [with SMA] don't matter."
Between Spinraza and its new competitor, Zolgensma, some are speculating that insurers will start to favor Zolgensma coverage instead, since the treatment is shorter and ultimately cheaper than Spinraza in the long term. But for some adults with SMA who can't access Spinraza and who don't qualify for Zolgensma treatment, the issue of what insurers will cover is moot.
"I was so excited when I heard that Zolgensma was approved by the FDA," said Annie Wilson, an adult SMA patient from Alameda, Calif. who has been fighting for Spinraza since 2017. "When I became aware that it was only being offered to children, I felt extremely sad and honestly a bit forgotten, like adults [with SMA] don't matter."
According to information from a Biogen representative, more than 7500 people worldwide have been treated with Spinraza to date, one third of whom are adults.
While Spinraza has been revolutionary for thousands of patients, it's unclear how many more lives state agencies and insurance companies will allow it to save.
People With This Rare Disease Can Barely Eat Protein. Biotechnology May Change That.
The Brown family at the Grand Tetons (2019). Clockwise from left, Christine, Kevin, Keagan, Connor, and Kellen.
Imagine that the protein in bread, eggs, steak, even beans is not the foundation for a healthy diet, but a poison to your brain. That is the reality for people living with Phenylketonuria, or PKU. This cluster of rare genetic variations affects the ability to digest phenylalanine (Phe), one of the chemical building blocks of protein. The toxins can build up in the brain causing severe mental retardation.
Can a probiotic help digest the troublesome proteins before they can enter the bloodstream and travel to the brain? A Boston area biotech start up, Synlogic, believes it can. Their starting point is an E. coli bacterium that has been used as a probiotic for more than a century. The company then screened thousands of gene variants to identify ones that produced enzymes most efficient at slicing and dicing the target proteins and optimized them further through directed evolution. The results have been encouraging.
But Christine Brown knew none of this when the hospital called saying that standard newborn screening of her son Connor had come back positive for PKU. It was urgent that they visit a special metabolic clinic the next day, which was about a three-hour drive away.
“I was told not to go on the Internet,” Christine recalls, “So when somebody tells you not to go on the Internet, what do you do? Even back in 2005, right.” What she saw were the worst examples of retardation, which was a common outcome from PKU before newborn screening became routine. “We were just in complete shell shock, our whole world just kind of shattered and went into a tail spin.”
“I remember feeding him the night before our clinic visit and almost feeling like I was feeding him poison because I knew that breast milk must have protein in it,” she says.
“Some of my first memories are of asking, ‘Mommy, can I eat this? There were yes foods and no foods.'"
Over the next few days the dedicated staff of the metabolic clinic at the Waisman Center at the University of Wisconsin Madison began to walk she and husband Kevin back from that nightmare. They learned that a simple blood test to screen newborns had been developed in the early 1960s to detect PKU and that the condition could be managed with stringent food restrictions and vigilant monitoring of Phe levels.
Everything in Your Mouth Counts
PKU can be successfully managed with a severely restricted diet. That simple statement is factually true, but practically impossible to follow, as it requires slashing protein consumption by about 90 percent. To compensate for the missing protein, several times a day PKU patients take a medical formula – commonly referred to simply as formula – containing forms of proteins that are digestible to their bodies. Several manufacturers now add vitamins and minerals and offer a variety of formats and tastes to make it more consumer friendly, but that wasn't always the case.
“When I was a kid, it tasted horrible, was the consistency of house paint. I didn't think about it, I just drank it. I didn't like it but you get used to it after a while,” recalls Jeff Wolf, the twang of Appalachia still strong in his voice. Now age 50, he grew up in Ashland, Kentucky and was part of the first wave of persons with PKU who were identified at birth as newborn screening was rolled out across the US. He says the options of taste and consistency have improved tremendously over the years.
Some people with PKU are restricted to as little as 8 grams of protein a day from food. That's a handful of almonds or a single hard-boiled egg; a skimpy 4-ounce hamburger and slice of cheese adds up to half of their weekly protein ration. Anything above that daily allowance is more than the body can handle and toxic levels of Phe begin to accumulate in the brain.
“Some of my first memories are of asking, ‘Mommy, can I eat this? There were yes foods and no foods,’” recalls Les Clark. He has never eaten a hamburger, steak, or ribs, practically a sacrilege for someone raised in Stanton, a small town in northeastern Nebraska, a state where the number of cattle and hogs are several-fold those of people.
His grandmother learned how to make low protein bread, but it looked and tasted different. His mom struggled making birthday cakes. “I learned some bad words at a very young age” as mom struggled applying icing that would pull the cake apart or a slice would collapse into a heap of crumbs, Les recalls.

Les Clark with a birthday cake.
Courtesy Clark
Controlling the diet “is not so bad when you are a baby” because that's all you know, says Jerry Vockley, Director of the Center for Rare Disease Therapy at Children's Hospital of Pittsburgh. “But after a while, as you get older and you start tasting other things and you say, Well, gee, this stuff tastes way better than what you're giving me. What's the deal? It becomes harder to maintain the diet.”
First is the lure of forbidden foods as children venture into the community away from the watchful eyes of parents. The support system weakens further when they leave home for college or work. “Pizza was mighty tasty,” Wolf' says of his first slice.
Vockley estimates that about 90 percent of adults with PKU are off of treatment. Moving might mean finding a new metabolic clinic that treats PKU. A lapse in insurance coverage can be a factor. Finally there is plain fatigue from multiple daily dosing of barely tolerable formula, monitoring protein intake, and simply being different in terms of food restrictions. Most people want to fit in and not be defined by their medical condition.
Jeff Wolf was one of those who dropped out in his twenties and thirties. He stopped going to clinic, monitoring his Phe levels, and counting protein. But the earlier experience of living with PKU never completely left the back of his mind; he listened to his body whenever eating too much protein left him with the “fuzzy brain" of a protein hangover. About a decade ago he reconnected with a metabolic clinic, began taking formula and watching his protein intake. He still may go over his allotment for a single day but he tries to compensate on subsequent days so that his Phe levels come back into balance.

Jeff Wolf on a boat.
Courtesy Wolf
One of the trickiest parts of trying to manage phenylalanine intake is the artificial sweetener aspartame. The chemical is ubiquitous in diet and lite foods and drinks. Gum too, you don't even have to swallow to receive a toxic dose of Phe. Most PKUers say it is easier to simply avoid these products entirely rather than try to count their Phe content.
Treatments
Most rare diseases have no treatment. There are two drugs for PKU that provide some benefit to some portion of patients but those drugs often have their own burdens.
KUVAN® (sapropterin dihydrochloride) is a pill or powder that helps correct a protein folding error so that food proteins can be digested. It is approved for most types of PKU in adults and children one month and older, and often is used along with a protein-restricted diet.
“The problem is that it doesn't work for every [patient's genetic] mutation, and there are hundreds of mutations that have been identified with PKU. Two to three percent of patients will have a very dramatic response and if you're one of those small number of patients, it's great,” says Vockley. “If you have one of the other mutation, chances are pretty good you still are going to end up on a restricted diet.”
PALYNZIQ® (pegvaliase-pqpz) “has the potential to lower the Phe to normal levels, it's a real breakthrough in the field,” says Vockley. “But is a very hard drug to use. Most folks have to take either one or two 2ml injections a day of something that is basically a gel, and some individuals have to take three.”
Many PKUers have reactions at the site of the injection and some develop anaphylaxis, a severe potentially life-threatening allergic reaction that can happen within seconds and can occur at any time, even after long term use. Many patients using Palynziq carry an EpiPen, a self-injection devise containing a form of adrenaline that can reverse some of the symptoms of anaphylaxis.
Then there is the cost. With the Kuvan dosing for an adult, “you're talking between $100,000 and $200,000 a year. And Palynziq is three times that,” says Vockley. Insurance coverage through a private plan or a state program is essential. Some state programs provide generous coverage while others are skimpy. Most large insurance company plans cover the drugs, sometimes with significant copays, but companies that are self-insured are under no legal obligation to provide coverage.
Les Clark found that out the hard way when the company he worked for was sold. The new owner was self-insured and declined to continue covering his drugs. Almost immediately he was out of pocket an additional $1500 a month for formula, and that was with a substantial discount through the manufacturer's patient support program. He says, “If you don't have an insurance policy that will cover the formula, it's completely unaffordable.” He quickly began to look for a new job.
Hope
It's easy to see why PKUers are eager for advances that will make managing their condition more effect, easier, and perhaps more affordable. Synlogic's efforts have drawn their attention and raised hopes.
Just before Thanksgiving Jerry Vockley presented the latest data to a metabolism conference meeting in Australia. There were only 8 patients in this group of a phase 2 trial using the original version of the company's lead E. coli product, SYNB1618, but they were intensely studied. Each was given the probiotic and then a challenge meal. Vockley saw a 40% reduction in Phe absorption and later a 20% reduction in mean fasting Phe levels in the blood. The product was easy to use and tolerate.
The company also presented early results for SYNB1934, a follow on version that further genetically tweaked the E. coli to roughly double the capacity to chop up the target proteins. Synlogic is recruiting patients for studies to determine the best dosing, which they are planning for next year.
“It's an exciting approach,” says Lex Cowsert, Director of Research Development at the National PKU Alliance, a nonprofit that supports the patient, family, and research communities involved with PKU. “Every patient is different, every patient has a different tolerance for the type of therapy that they are willing to pursue,” and if it pans out, it will be a welcome addition, either alone or in combination with other approaches, to living with PKU.
Author's Note: Reporting this story was made possible by generous support from the National Press Foundation and the Fondation Ipsen. Thanks to the people who so generously shared their time and stories in speaking with me.
Are Brain Implants the Future of Treatment for Depression and Anxiety?
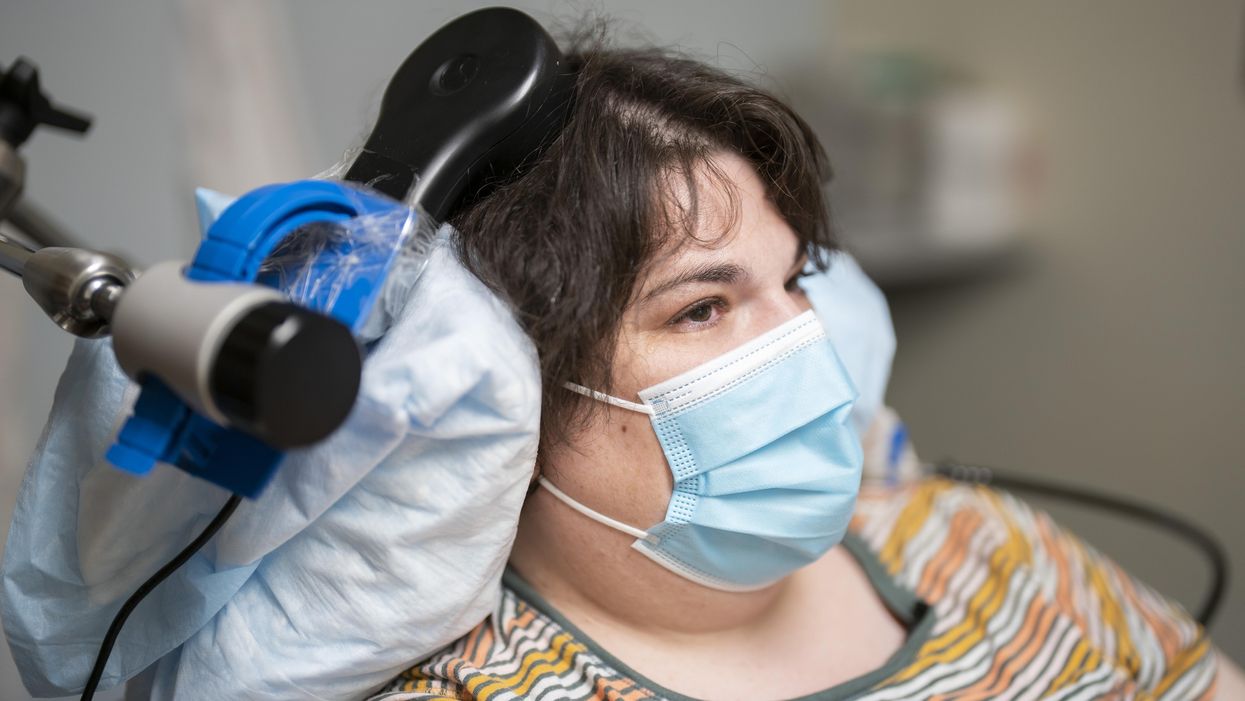
Sarah, clinical trial participant, at an appointment with Katherine Scangos, MD, PhD, at UCSF’s Langley Porter Psychiatric Institute.
When she woke up after a procedure involving drilling small holes in her skull, a woman suffering from chronic depression reported feeling “euphoric”. The holes were made to fit the wires that connected her brain with a matchbox-sized electrical implant; this would deliver up to 300 short-lived electricity bursts per day to specific parts of her brain.
Over a year later, Sarah, 36, says the brain implant has turned her life around. A sense of alertness and energy have replaced suicidal thoughts and feelings of despair, which had persisted despite antidepressants and electroconvulsive therapy. Sarah is the first person to have received a brain implant to treat depression, a breakthrough that happened during an experimental study published recently in Nature Medicine.
“What we did was use deep-brain stimulation (DBS), a technique used in the treatment of epilepsy,” says Andrew Krystal, professor of psychiatry at University of California, San Francisco (UCSF), and one of the study’s researchers. DBS typically involves implanting electrodes into specific areas of the brain to reduce seizures not controlled with medication or to remove the part of the brain that causes the seizures. Instead of choosing and stimulating a single brain site though, the UCSF team took a different approach.
They first used 10 electrodes to map Sarah’s brain activity, a phase that lasted 10 days, during which they developed a neural biomarker, a specific pattern of brain activity that indicated the onset of depression symptoms (in Sarah, this was detected in her amygdala, an almondlike structure located near the base of the brain). But they also saw that delivering a tiny burst of electricity to the patient’s ventral striatum, an area of the brain that sits in the center, above and behind the ears, dramatically improved these symptoms. What they had to do was outfit Sara’s brain with a DBS-device programmed to propagate small waves of electricity to the ventral striatum only when it discerned the pattern.
“We are not trying to take away normal responses to the world. We are just trying to eliminate this one thing, which is depression, which impedes patients’ ability to function and deal with normal stuff.”
“It was a personalized treatment not only in where to stimulate, but when to stimulate,” Krystal says. Sarah’s depression translated to low amounts of energy, loss of pleasure and interest in life, and feelings of sluggishness. Those symptoms went away when scientists stimulated her ventral capsule area. When the same area was manipulated by electricity when Sarah’s symptoms “were not there” though, she was feeling more energetic, but this sudden flush of energy soon gave way to feelings of overstimulation and anxiety. “This is a very tangible illustration of why it's best to simulate only when you need it,” says Krystal.
We have the tendency to lump together depression symptoms, but, in reality, they are quite diverse; some people feel sad and lethargic, others stay up all night; some overeat, others don’t eat at all. “This happens because people have different underlying dysfunctions in different parts of their brain. Our approach is targeting the specific brain circuit that modulates different kinds of symptoms. Simply, where we stimulate depends on the specific set of problems a person has,” Krystal says. Such tailormade brain stimulation for patients with long-term, drug-resistant depression, which would be easy to use at home, could be transformative, the UCSF researcher concludes.
In the U.S., 12.7 percent of the population is on antidepressants. Almost exactly the same percentage of Australians–12.5–take similar drugs every day. With 13 percent of its population being on antidepressants, Iceland is the world’s highest antidepressant consumer. And quite away from Scandinavia, the Southern European country of Portugal is the world’s third strongest market for corresponding medication.
By 2020, nearly 15.5 million people had been consuming antidepressants for a time period exceeding five years. Between 40 and 60 percent of them saw improvements. “For those people, it was absolutely what they needed, whether that was increased serotonin, or increased norepinephrine or increased dopamine, ” says Frank Anderson, a psychiatrist who has been administering antidepressants in his private practice “for a long time”, and author of Transcending Trauma, a book about resolving complex and dissociative trauma.
Yet the UCSF study brings to the mental health field a specificity it has long lacked. “A lot of the traditional medications only really work on six neurotransmitters, when there are over 100 neurotransmitters in the brain,” Anderson says. Drugs are changing the chemistry of a single system in the brain, but brain stimulation is essentially changing the very architecture of the brain, says James Giordano, professor of neurology and biochemistry at Georgetown University Medical Center in Washington and a neuroethicist. It is a far more elegant approach to treating brain disorders, with the potential to prove a lifesaver for the 40 to 50 percent of patients who see no benefits at all with antidepressants, Giordano says. It is neurofeedback, on steroids, adds Anderson. But it comes with certain risks.
Even if the device generating the brain stimulation sits outside the skull and could be easily used at home, the whole process still involves neurosurgery. While the sophistication and precision of brain surgeries has significantly improved over the last years, says Giordano, they always carry risks, such as an allergic reaction to anesthesia, bleeding in the brain, infection at the wound site, blood clots, even coma. Non-invasive brain stimulation (NIBS), a technology currently being developed by the Defense Advanced Research Projects Agency (DARPA), could potentially tackle this. Patients could wear a cap, helmet, or visor that transmits electrical signals from the brain to a computer system and back, in a brain-computer interface that would not need surgery.
“This could counter the implantation of hardware into the brain and body, around which there is also a lot of public hesitance,” says Giordano, who is working on such techniques at DARPA.
Embedding a chip in your head is one of the finest examples of biohacking, an umbrella word for all the practices aimed at hacking one’s body and brain to enhance performance –a citizen do-it-yourself biology. It is also a word charged enough to set off a public backlash. Large segments of the population will simply refuse to allow that level of invasiveness in their heads, says Laura Cabrera, an associate professor of neuroethics at the Center for Neural Engineering, Department of Engineering Science and Mechanics at Penn State University. Cabrera urges caution when it comes to DBS’s potential.
“We've been using it for Parkinson's for over two decades, hoping that now that they get DBS, patients will get off medications. But people have continued taking their drugs, even increasing them,” she says. What the UCSF found is a proof of concept that DBS worked in one depressed person, but there’s a long way ahead until we can confidently say this finding is generalizable to a large group of patients. Besides, as a society, we are not there yet, says Cabrera. “Most people, at least in my research, say they don't want to have things in their brain,” she says. But what could really go wrong if we biohacked our own brains anyway?
In 2014, a man who had received a deep brain implant for a movement disorder started developing an affection for Johnny Cash’s music when he had previously been an avid country music fan. Many protested that the chip had tampered with his personality. Could sparking the brain with electricity generated by a chip outside it put an end to our individuality, messing with our musical preferences, unique quirks, our deeper sense of ego?
“What we found is that when you stimulate a region, you affect people’s moods, their energies,” says Krystal. You are neither changing their personality nor creating creatures of eternal happiness, he says. “’Being on a phone call would generally be a setting that would normally trigger symptoms of depression in me,’” Krystal reports his patient telling him. ‘I now know bad things happen, but am not affected by them in the same way. They don’t trigger the depression.’” Of the research, Krystal continues: “We are not trying to take away normal responses to the world. We are just trying to eliminate this one thing, which is depression, which impedes patients’ ability to function and deal with normal stuff.”
Yet even change itself shouldn't be seen as threatening, especially if the patient had probably desired it in the first place. “The intent of therapy in psychiatric disorders is to change the personality, because a psychiatric disorder by definition is a disorder of personality,” says Cabrera. A person in therapy wants to restore the lost sense of “normal self”. And as for this restoration altering your original taste in music, Cabrera says we are talking about rarities, extremely scarce phenomena that are possible with medication as well.
Maybe it is the allure of dystopian sci-fi films: people have a tendency to worry about dark forces that will spread malice across the world when the line between human and machine has blurred. Such mind-control through DBS would probably require a decent leap of logic with the tools science has--at least to this day. “This would require an understanding of the parameters of brain stimulation we still don't have,” says Cabrera. Still, brain implants are not fully corrupt-proof.
“Hackers could shut off the device or change the parameters of the patient's neurological function enhancing symptoms or creating harmful side-effects,” says Giordano.
There are risks, but also failsafe ways to tackle them, adds Anderson. “Just like medications are not permanent, we could ensure the implants are used for a specific period of time,” he says. And just like people go in for checkups when they are under medication, they could periodically get their personal brain implants checked to see if they have been altered or not, he continues. “It is what my research group refers to as biosecurity by design,” says Giordano. “It is important that we proactively design systems that cannot be corrupted.”
Two weeks after receiving the implant, Sarah scored 14 out of 54 on the Montgomery-Åsberg Depression Rating Scale, a ten-item questionnaire psychiatrists use to measure the severity of depressive episodes. She had initially scored 36. Today she scores under 10. She would have had to wait between four and eight weeks to see positive results had she taken the antidepressant road, says Krystal.
He and his team have enrolled two other patients in the trials and hope to add nine more. They already have some preliminary evidence that there's another place that works better in the brain of another patient, because that specific patient had been experiencing more anxiety as opposed to despondency. Almost certainly, we will have different biomarkers for different people, and brain stimulation will be tailored to a person’s unique situation, says Krystal. “Each brain is different, just like each face is different.”