Scientists redesign bacteria to tackle the antibiotic resistance crisis
Probiotic bacteria can be engineered to fight antibiotic-resistant superbugs by releasing chemicals that kill them.
In 1945, almost two decades after Alexander Fleming discovered penicillin, he warned that as antibiotics use grows, they may lose their efficiency. He was prescient—the first case of penicillin resistance was reported two years later. Back then, not many people paid attention to Fleming’s warning. After all, the “golden era” of the antibiotics age had just began. By the 1950s, three new antibiotics derived from soil bacteria — streptomycin, chloramphenicol, and tetracycline — could cure infectious diseases like tuberculosis, cholera, meningitis and typhoid fever, among others.
Today, these antibiotics and many of their successors developed through the 1980s are gradually losing their effectiveness. The extensive overuse and misuse of antibiotics led to the rise of drug resistance. The livestock sector buys around 80 percent of all antibiotics sold in the U.S. every year. Farmers feed cows and chickens low doses of antibiotics to prevent infections and fatten up the animals, which eventually causes resistant bacterial strains to evolve. If manure from cattle is used on fields, the soil and vegetables can get contaminated with antibiotic-resistant bacteria. Another major factor is doctors overprescribing antibiotics to humans, particularly in low-income countries. Between 2000 to 2018, the global rates of human antibiotic consumption shot up by 46 percent.
In recent years, researchers have been exploring a promising avenue: the use of synthetic biology to engineer new bacteria that may work better than antibiotics. The need continues to grow, as a Lancet study linked antibiotic resistance to over 1.27 million deaths worldwide in 2019, surpassing HIV/AIDS and malaria. The western sub-Saharan Africa region had the highest death rate (27.3 people per 100,000).
Researchers warn that if nothing changes, by 2050, antibiotic resistance could kill 10 million people annually.
To make it worse, our remedy pipelines are drying up. Out of the 18 biggest pharmaceutical companies, 15 abandoned antibiotic development by 2013. According to the AMR Action Fund, venture capital has remained indifferent towards biotech start-ups developing new antibiotics. In 2019, at least two antibiotic start-ups filed for bankruptcy. As of December 2020, there were 43 new antibiotics in clinical development. But because they are based on previously known molecules, scientists say they are inadequate for treating multidrug-resistant bacteria. Researchers warn that if nothing changes, by 2050, antibiotic resistance could kill 10 million people annually.
The rise of synthetic biology
To circumvent this dire future, scientists have been working on alternative solutions using synthetic biology tools, meaning genetically modifying good bacteria to fight the bad ones.
From the time life evolved on earth around 3.8 billion years ago, bacteria have engaged in biological warfare. They constantly strategize new methods to combat each other by synthesizing toxic proteins that kill competition.
For example, Escherichia coli produces bacteriocins or toxins to kill other strains of E.coli that attempt to colonize the same habitat. Microbes like E.coli (which are not all pathogenic) are also naturally present in the human microbiome. The human microbiome harbors up to 100 trillion symbiotic microbial cells. The majority of them are beneficial organisms residing in the gut at different compositions.
The chemicals that these “good bacteria” produce do not pose any health risks to us, but can be toxic to other bacteria, particularly to human pathogens. For the last three decades, scientists have been manipulating bacteria’s biological warfare tactics to our collective advantage.
In the late 1990s, researchers drew inspiration from electrical and computing engineering principles that involve constructing digital circuits to control devices. In certain ways, every cell in living organisms works like a tiny computer. The cell receives messages in the form of biochemical molecules that cling on to its surface. Those messages get processed within the cells through a series of complex molecular interactions.
Synthetic biologists can harness these living cells’ information processing skills and use them to construct genetic circuits that perform specific instructions—for example, secrete a toxin that kills pathogenic bacteria. “Any synthetic genetic circuit is merely a piece of information that hangs around in the bacteria’s cytoplasm,” explains José Rubén Morones-Ramírez, a professor at the Autonomous University of Nuevo León, Mexico. Then the ribosome, which synthesizes proteins in the cell, processes that new information, making the compounds scientists want bacteria to make. “The genetic circuit remains separated from the living cell’s DNA,” Morones-Ramírez explains. When the engineered bacteria replicates, the genetic circuit doesn’t become part of its genome.
Highly intelligent by bacterial standards, some multidrug resistant V. cholerae strains can also “collaborate” with other intestinal bacterial species to gain advantage and take hold of the gut.
In 2000, Boston-based researchers constructed an E.coli with a genetic switch that toggled between turning genes on and off two. Later, they built some safety checks into their bacteria. “To prevent unintentional or deleterious consequences, in 2009, we built a safety switch in the engineered bacteria’s genetic circuit that gets triggered after it gets exposed to a pathogen," says James Collins, a professor of biological engineering at MIT and faculty member at Harvard University’s Wyss Institute. “After getting rid of the pathogen, the engineered bacteria is designed to switch off and leave the patient's body.”
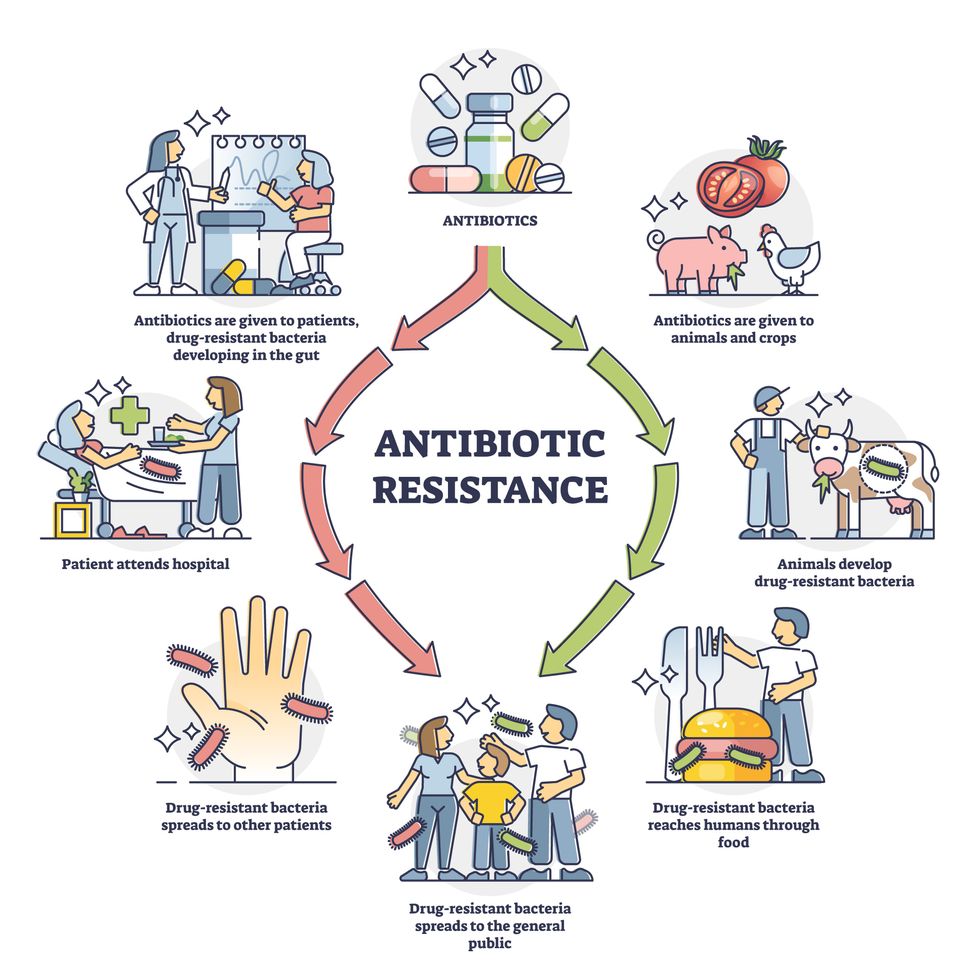
Overuse and misuse of antibiotics causes resistant strains to evolve
Adobe Stock
Seek and destroy
As the field of synthetic biology developed, scientists began using engineered bacteria to tackle superbugs. They first focused on Vibrio cholerae, which in the 19th and 20th century caused cholera pandemics in India, China, the Middle East, Europe, and Americas. Like many other bacteria, V. cholerae communicate with each other via quorum sensing, a process in which the microorganisms release different signaling molecules, to convey messages to its brethren. Highly intelligent by bacterial standards, some multidrug resistant V. cholerae strains can also “collaborate” with other intestinal bacterial species to gain advantage and take hold of the gut. When untreated, cholera has a mortality rate of 25 to 50 percent and outbreaks frequently occur in developing countries, especially during floods and droughts.
Sometimes, however, V. cholerae makes mistakes. In 2008, researchers at Cornell University observed that when quorum sensing V. cholerae accidentally released high concentrations of a signaling molecule called CAI-1, it had a counterproductive effect—the pathogen couldn’t colonize the gut.
So the group, led by John March, professor of biological and environmental engineering, developed a novel strategy to combat V. cholerae. They genetically engineered E.coli to eavesdrop on V. cholerae communication networks and equipped it with the ability to release the CAI-1 molecules. That interfered with V. cholerae progress. Two years later, the Cornell team showed that V. cholerae-infected mice treated with engineered E.coli had a 92 percent survival rate.
These findings inspired researchers to sic the good bacteria present in foods like yogurt and kimchi onto the drug-resistant ones.
Three years later in 2011, Singapore-based scientists engineered E.coli to detect and destroy Pseudomonas aeruginosa, an often drug-resistant pathogen that causes pneumonia, urinary tract infections, and sepsis. Once the genetically engineered E.coli found its target through its quorum sensing molecules, it then released a peptide, that could eradicate 99 percent of P. aeruginosa cells in a test-tube experiment. The team outlined their work in a Molecular Systems Biology study.
“At the time, we knew that we were entering new, uncharted territory,” says lead author Matthew Chang, an associate professor and synthetic biologist at the National University of Singapore and lead author of the study. “To date, we are still in the process of trying to understand how long these microbes stay in our bodies and how they might continue to evolve.”
More teams followed the same path. In a 2013 study, MIT researchers also genetically engineered E.coli to detect P. aeruginosa via the pathogen’s quorum-sensing molecules. It then destroyed the pathogen by secreting a lab-made toxin.
Probiotics that fight
A year later in 2014, a Nature study found that the abundance of Ruminococcus obeum, a probiotic bacteria naturally occurring in the human microbiome, interrupts and reduces V.cholerae’s colonization— by detecting the pathogen’s quorum sensing molecules. The natural accumulation of R. obeum in Bangladeshi adults helped them recover from cholera despite living in an area with frequent outbreaks.
The findings from 2008 to 2014 inspired Collins and his team to delve into how good bacteria present in foods like yogurt and kimchi can attack drug-resistant bacteria. In 2018, Collins and his team developed the engineered probiotic strategy. They tweaked a bacteria commonly found in yogurt called Lactococcus lactis to treat cholera.
Engineered bacteria can be trained to target pathogens when they are at their most vulnerable metabolic stage in the human gut. --José Rubén Morones-Ramírez.
More scientists followed with more experiments. So far, researchers have engineered various probiotic organisms to fight pathogenic bacteria like Staphylococcus aureus (leading cause of skin, tissue, bone, joint and blood infections) and Clostridium perfringens (which causes watery diarrhea) in test-tube and animal experiments. In 2020, Russian scientists engineered a probiotic called Pichia pastoris to produce an enzyme called lysostaphin that eradicated S. aureus in vitro. Another 2020 study from China used an engineered probiotic bacteria Lactobacilli casei as a vaccine to prevent C. perfringens infection in rabbits.
In a study last year, Ramírez’s group at the Autonomous University of Nuevo León, engineered E. coli to detect quorum-sensing molecules from Methicillin-resistant Staphylococcus aureus or MRSA, a notorious superbug. The E. coli then releases a bacteriocin that kills MRSA. “An antibiotic is just a molecule that is not intelligent,” says Ramírez. “On the other hand, engineered bacteria can be trained to target pathogens when they are at their most vulnerable metabolic stage in the human gut.”
Collins and Timothy Lu, an associate professor of biological engineering at MIT, found that engineered E. coli can help treat other conditions—such as phenylketonuria, a rare metabolic disorder, that causes the build-up of an amino acid phenylalanine. Their start-up Synlogic aims to commercialize the technology, and has completed a phase 2 clinical trial.
Circumventing the challenges
The bacteria-engineering technique is not without pitfalls. One major challenge is that beneficial gut bacteria produce their own quorum-sensing molecules that can be similar to those that pathogens secrete. If an engineered bacteria’s biosensor is not specific enough, it will be ineffective.
Another concern is whether engineered bacteria might mutate after entering the gut. “As with any technology, there are risks where bad actors could have the capability to engineer a microbe to act quite nastily,” says Collins of MIT. But Collins and Ramírez both insist that the chances of the engineered bacteria mutating on its own are virtually non-existent. “It is extremely unlikely for the engineered bacteria to mutate,” Ramírez says. “Coaxing a living cell to do anything on command is immensely challenging. Usually, the greater risk is that the engineered bacteria entirely lose its functionality.”
However, the biggest challenge is bringing the curative bacteria to consumers. Pharmaceutical companies aren’t interested in antibiotics or their alternatives because it’s less profitable than developing new medicines for non-infectious diseases. Unlike the more chronic conditions like diabetes or cancer that require long-term medications, infectious diseases are usually treated much quicker. Running clinical trials are expensive and antibiotic-alternatives aren’t lucrative enough.
“Unfortunately, new medications for antibiotic resistant infections have been pushed to the bottom of the field,” says Lu of MIT. “It's not because the technology does not work. This is more of a market issue. Because clinical trials cost hundreds of millions of dollars, the only solution is that governments will need to fund them.” Lu stresses that societies must lobby to change how the modern healthcare industry works. “The whole world needs better treatments for antibiotic resistance.”
New device can diagnose concussions using AI
A new test called the EyeBox could provide a more objective - and portable - tool to measure whether people have concussions in stadiums and hospitals.
For a long time after Mary Smith hit her head, she was not able to function. Test after test came back normal, so her doctors ruled out the concussion, but she knew something was wrong. Finally, when she took a test with a novel EyeBOX device, recently approved by the FDA, she learned she indeed had been dealing with the aftermath of a concussion.
“I felt like even my husband and doctors thought I was faking it or crazy,” recalls Smith, who preferred not to disclose her real name. “When I took the EyeBOX test it showed that my eyes were not moving together and my BOX score was abnormal.” To her diagnosticians, scientists at the Minneapolis-based company Oculogica who developed the EyeBOX, these markers were concussion signs. “I cried knowing that finally someone could figure out what was wrong with me and help me get better,” she says.
Concussion affects around 42 million people worldwide. While it’s increasingly common in the news because of sports injuries, anything that causes damage to the head, from a fall to a car accident, can result in a concussion. The sudden blow or jolt can disrupt the normal way the brain works. In the immediate aftermath, people may suffer from headaches, lose consciousness and experience dizziness, confusion and vomiting. Some recover but others have side effects that can last for years, particularly affecting memory and concentration.
There is no simple standard-of-care test to confirm a concussion or rule it out. Neither do they appear on MRI and CT scans. Instead, medical professionals use more indirect approaches that test symptoms of concussions, such as assessments of patients’ learning and memory skills, ability to concentrate and problem solving. They also look at balance and coordination. Most tests are in the form of questionnaires or symptom checklists. Consequently, they have limitations, can be biased and may miss a concussion or produce a false positive. Some people suspected of having a concussion may ordinarily have difficulties with literary and problem-solving tests because of language challenges or education levels.
Another problem with current tests is that patients, particularly soldiers who want to return to combat and athletes who would like to keep competing, could try and hide their symptoms to avoid being diagnosed with a brain injury. Trauma physicians who work with concussion patients have the need for a tool that is more objective and consistent.
“This type of assessment doesn’t rely on the patient's education level, willingness to follow instructions or cooperation. You can’t game this.” -- Uzma Samadani, founder of Oculogica
“The importance of having an objective measurement tool for the diagnosis of concussion is of great importance,” says Douglas Powell, associate professor of biomechanics at the University of Memphis, with research interests in sports injury and concussion. “While there are a number of promising systems or metrics, we have yet to develop a system that is portable, accessible and objective for use on the sideline and in the clinic. The EyeBOX may be able to address these issues, though time will be the ultimate test of performance.”
The EyeBOX as a window inside the brain
Using eye movements to diagnose a concussion has emerged as a promising technique since around 2010. Oculogica combined eye movements with AI to develop the EyeBOX to develop an unbiased objective diagnostic tool.
“What’s so great about this type of assessment is it doesn’t rely on the patient's education level, willingness to follow instructions or cooperation,” says Uzma Samadani, a neurosurgeon and brain injury researcher at the University of Minnesota, who founded Oculogica. “You can’t game this. It assesses functions that are prompted by your brain.”
In 2010, Samadani was working on a clinical trial to improve the outcome of brain injuries. The team needed some way to measure if seriously brain injured patients were improving. One thing patients could do was watch TV. So Samadani designed and patented an AI-based algorithm that tracks the relationship between eye movement and concussion.
The EyeBOX test requires patients to watch movie or music clips for 220 seconds. An eye tracking camera records subconscious eye movements, tracking eye positions 500 times per seconds as patients watch the video. It collects over 100,000 data points. The device then uses AI to assess whether there’s any disruptions from the normal way the eyes move.
Cranial nerves are responsible for transmitting information between the brain and the body. Many are involved in eye movement. Pressure caused by a concussion can affect how these nerves work. So tracking how the eyes move can indicate if there’s anything wrong with the cranial nerves and where the problem lies.
If someone is healthy, their eyes should be able to focus on an object, follow movement and both eyes should be coordinated with each other. The EyeBox can detect abnormalities. For example, if a patient’s eyes are coordinated but they are not moving as they should, that indicates issues in the central brain stem, whilst only one eye moving abnormally suggests that a particular nerve section is affected.

Uzma Samadani with the EyeBOX device
Courtesy Oculogica
“The EyeBOX is a monitor for cranial nerves,” says Samadani. “Essentially it’s a form of digital neurological exam. “Several other eye-tracking techniques already exist, but they rely on subjective self-reported symptoms. Many also require a baseline, a measure of how patients reacted when they were healthy, which often isn’t available.
VOMS (Vestibular Ocular Motor Screen) is one of the most accurate diagnostic tests used in clinics in combination with other tests, but it is subjective. It involves a therapist getting patients to move their head or eyes as they focus or follow a particular object. Patients then report their symptoms.
The King-Devick test measures how fast patients can read numbers and compares it to a baseline. Since it is mainly used for athletes, the initial test is completed before the season starts. But participants can manipulate it. It also cannot be used in emergency rooms because the majority of patients wouldn’t have prior baseline tests.
Unlike these tests, EyeBOX doesn’t use a baseline and is objective because it doesn’t rely on patients’ answers. “It shows great promise,” says Thomas Wilcockson, a senior lecturer of psychology in Loughborough University, who is an expert in using eye tracking techniques in neurological disorders. “Baseline testing of eye movements is not always possible. Alternative measures of concussion currently in development, including work with VR headsets, seem to currently require it. Therefore the EyeBOX may have an advantage.”
A technology that’s still evolving
In their last clinical trial, Oculogica used the EyeBOX to test 46 patients who had concussion and 236 patients who did not. The sensitivity of the EyeBOX, or the probability of it correctly identifying the patient’s concussion, was 80.4 percent. Meanwhile, the test accurately ruled out a concussion in 66.1 percent of cases. This is known as its specificity score.
While the team is working on improving the numbers, experts who treat concussion patients find the device promising. “I strongly support their use of eye tracking for diagnostic decision making,” says Douglas Powell. “But for diagnostic tests, we would prefer at least one of the sensitivity or specificity values to be greater than 90 percent. Powell compares EyeBOX with the Buffalo Concussion Treadmill Test, which has sensitivity and specificity values of 73 and 78 percent, respectively. The VOMS also has shown greater accuracy than the EyeBOX, at least for now. Still, EyeBOX is competitive with the best diagnostic testing available for concussion and Powell hopes that its detection prowess will improve. “I anticipate that the algorithms being used by Oculogica will be under continuous revision and expect the results will improve within the next several years.”
“The color of your skin can have a huge impact in how quickly you are triaged and managed for brain injury. People of color have significantly worse outcomes after traumatic brain injury than people who are white.” -- Uzma Samadani, founder of Oculogica
Powell thinks the EyeBOX could be an important complement to other concussion assessments.
“The Oculogica product is a viable diagnostic tool that supports clinical decision making. However, concussion is an injury that can present with a wide array of symptoms, and the use of technology such as the Oculogica should always be a supplement to patient interaction.”
Ioannis Mavroudis, a consultant neurologist at Leeds Teaching Hospital, agrees that the EyeBOX has promise, but cautions that concussions are too complex to rely on the device alone. For example, not all concussions affect how eyes move. “I believe that it can definitely help, however not all concussions show changes in eye movements. I believe that if this could be combined with a cognitive assessment the results would be impressive.”
The Oculogica team submitted their clinical data for FDA approval and received it in 2018. Now, they’re working to bring the test to the commercial market and using the device clinically to help diagnose concussions for clients. They also want to look at other areas of brain health in the next few years. Samadani believes that the EyeBOX could possibly be used to detect diseases like multiple sclerosis or other neurological conditions. “It’s a completely new way of figuring out what someone’s neurological exam is and we’re only beginning to realize the potential,” says Samadani.
One of Samadani’s biggest aspirations is to help reduce inequalities in healthcare because of skin color and other factors like money or language barriers. From that perspective, the EyeBOX’s greatest potential could be in emergency rooms. It can help diagnose concussions in addition to the questionnaires, assessments and symptom checklists, currently used in the emergency departments. Unlike these more subjective tests, EyeBOX can produce an objective analysis of brain injury through AI when patients are admitted and assessed, unrelated to their socioeconomic status, education, or language abilities. Studies suggest that there are racial disparities in how patients with brain injuries are treated, such as how quickly they're assessed and get a treatment plan.
“The color of your skin can have a huge impact in how quickly you are triaged and managed for brain injury,” says Samadani. “As a result of that, people of color have significantly worse outcomes after traumatic brain injury than people who are white. The EyeBOX has the potential to reduce inequalities,” she explains.
“If you had a digital neurological tool that you could screen and triage patients on admission to the emergency department you would potentially be able to make sure that everybody got the same standard of care,” says Samadani. “My goal is to change the way brain injury is diagnosed and defined.”
Catching colds may help protect kids from Covid
A new study shows that the immune system's response to colds can help prepare it to defend against COVID-19 - but only in the very young.
A common cold virus causes the immune system to produce T cells that also provide protection against SARS-CoV-2, according to new research. The study, published last month in PNAS, shows that this effect is most pronounced in young children. The finding may help explain why most young people who have been exposed to the cold-causing coronavirus have not developed serious cases of COVID-19.
One curiosity stood out in the early days of the COVID-19 pandemic – why were so few kids getting sick. Generally young children and the elderly are the most vulnerable to disease outbreaks, particularly viral infections, either because their immune systems are not fully developed or they are starting to fail.
But solid information on the new infection was so scarce that many public health officials acted on the precautionary principle, assumed a worst-case scenario, and applied the broadest, most restrictive policies to all people to try to contain the coronavirus SARS-CoV-2.
One early thought was that lockdowns worked and kids (ages 6 months to 17 years) simply were not being exposed to the virus. So it was a shock when data started to come in showing that well over half of them carried antibodies to the virus, indicating exposure without getting sick. That trend grew over time and the latest tracking data from the CDC shows that 96.3 percent of kids in the U.S. now carry those antibodies.
Antibodies are relatively quick and easy to measure, but some scientists are exploring whether the reactions of T cells could serve as a more useful measure of immune protection.
But that couldn't be the whole story because antibody protection fades, sometimes as early as a month after exposure and usually within a year. Additionally, SARS-CoV-2 has been spewing out waves of different variants that were more resistant to antibodies generated by their predecessors. The resistance was so significant that over time the FDA withdrew its emergency use authorization for a handful of monoclonal antibodies with earlier approval to treat the infection because they no longer worked.
Antibodies got most of the attention early on because they are part of the first line response of the immune system. Antibodies can bind to viruses and neutralize them, preventing infection. They are relatively quick and easy to measure and even manufacture, but as SARS-CoV-2 showed us, often viruses can quickly evolve to become more resistant to them. Some scientists are exploring whether the reactions of T cells could serve as a more useful measure of immune protection.
Kids, colds and T cells
T cells are part of the immune system that deals with cells once they have become infected. But working with T cells is much more difficult, takes longer, and is more expensive than working with antibodies. So studies often lags behind on this part of the immune system.
A group of researchers led by Annika Karlsson at the Karolinska Institute in Sweden focuses on T cells targeting virus-infected cells and, unsurprisingly, saw that they can play a role in SARS-CoV-2 infection. Other labs have shown that vaccination and natural exposure to the virus generates different patterns of T cell responses.
The Swedes also looked at another member of the coronavirus family, OC43, which circulates widely and is one of several causes of the common cold. The molecular structure of OC43 is similar to its more deadly cousin SARS-CoV-2. Sometimes a T cell response to one virus can produce a cross-reactive response to a similar protein structure in another virus, meaning that T cells will identify and respond to the two viruses in much the same way. Karlsson looked to see if T cells for OC43 from a wide age range of patients were cross-reactive to SARS-CoV-2.
And that is what they found, as reported in the PNAS study last month; there was cross-reactive activity, but it depended on a person’s age. A subset of a certain type of T cells, called mCD4+,, that recognized various protein parts of the cold-causing virus, OC43, expressed on the surface of an infected cell – also recognized those same protein parts from SARS-CoV-2. The T cell response was lower than that generated by natural exposure to SARS-CoV-2, but it was functional and thus could help limit the severity of COVID-19.
“One of the most politicized aspects of our pandemic response was not accepting that children are so much less at risk for severe disease with COVID-19,” because usually young children are among the most vulnerable to pathogens, says Monica Gandhi, professor of medicine at the University of California San Francisco.
“The cross-reactivity peaked at age six when more than half the people tested have a cross-reactive immune response,” says Karlsson, though their sample is too small to say if this finding applies more broadly across the population. The vast majority of children as young as two years had OC43-specific mCD4+ T cell responses. In adulthood, the functionality of both the OC43-specific and the cross-reactive T cells wane significantly, especially with advanced age.
“Considering that the mortality rate in children is the lowest from ages five to nine, and higher in younger children, our results imply that cross-reactive mCD4+ T cells may have a role in the control of SARS-CoV-2 infection in children,” the authors wrote in their paper.
“One of the most politicized aspects of our pandemic response was not accepting that children are so much less at risk for severe disease with COVID-19,” because usually young children are among the most vulnerable to pathogens, says Monica Gandhi, professor of medicine at the University of California San Francisco and author of the book, Endemic: A Post-Pandemic Playbook, to be released by the Mayo Clinic Press this summer. The immune response of kids to SARS-CoV-2 stood our expectations on their head. “We just haven't seen this before, so knowing the mechanism of protection is really important.”
Why the T cell immune response can fade with age is largely unknown. With some viruses such as measles, a single vaccination or infection generates life-long protection. But respiratory tract infections, like SARS-CoV-2, cause a localized infection - specific to certain organs - and that response tends to be shorter lived than systemic infections that affect the entire body. Karlsson suspects the elderly might be exposed to these localized types of viruses less often. Also, frequent continued exposure to a virus that results in reactivation of the memory T cell pool might eventually result in “a kind of immunosenescence or immune exhaustion that is associated with aging,” Karlsson says. https://leaps.org/scientists-just-started-testing-a-new-class-of-drugs-to-slow-and-even-reverse-aging/particle-3 This fading protection is why older people need to be repeatedly vaccinated against SARS-CoV-2.
Policy implications
Following the numbers on COVID-19 infections and severity over the last three years have shown us that healthy young people without risk factors are not likely to develop serious disease. This latest study points to a mechanism that helps explain why. But the inertia of existing policies remains. How should we adjust policy recommendations based on what we know today?
The World Health Organization (WHO) updated their COVID-19 vaccination guidance on March 28. It calls for a focus on vaccinating and boosting those at risk for developing serious disease. The guidance basically shrugged its shoulders when it came to healthy children and young adults receiving vaccinations and boosters against COVID-19. It said the priority should be to administer the “traditional essential vaccines for children,” such as those that protect against measles, rubella, and mumps.
“As an immunologist and a mother, I think that catching a cold or two when you are a kid and otherwise healthy is not that bad for you. Children have a much lower risk of becoming severely ill with SARS-CoV-2,” says Karlsson. She has followed public health guidance in Sweden, which means that her young children have not been vaccinated, but being older, she has received the vaccine and boosters. Gandhi and her children have been vaccinated, but they do not plan on additional boosters.
The WHO got it right in “concentrating on what matters,” which is getting traditional childhood immunizations back on track after their dramatic decline over the last three years, says Gandhi. Nor is there a need for masking in schools, according to a study from the Catalonia region of Spain. It found “no difference in masking and spread in schools,” particularly since tracking data indicate that nearly all young people have been exposed to SARS-CoV-2.
Both researchers lament that public discussion has overemphasized the quickly fading antibody part of the immune response to SARS-CoV-2 compared with the more durable T cell component. They say developing an efficient measure of T cell response for doctors to use in the clinic would help to monitor immunity in people at risk for severe cases of COVID-19 compared with the current method of toting up potential risk factors.