In November 2020, messenger RNA catapulted into the public consciousness when the first COVID-19 vaccines were authorized for emergency use. Around the same time, an equally groundbreaking yet relatively unheralded application of mRNA technology was taking place at a London hospital.
Over the past two decades, there's been increasing interest in harnessing mRNA — molecules present in all of our cells that act like digital tape recorders, copying instructions from DNA in the cell nucleus and carrying them to the protein-making structures — to create a whole new class of therapeutics.
Scientists realized that artificial mRNA, designed in the lab, could be used to instruct our cells to produce certain antibodies, turning our bodies into vaccine-making factories, or to recognize and attack tumors. More recently, researchers recognized that mRNA could also be used to make another groundbreaking technology far more accessible to more patients: gene editing. The gene-editing tool CRISPR has generated plenty of hype for its potential to cure inherited diseases. But delivering CRISPR to the body is complicated and costly.
"Most gene editing involves taking cells out of the patient, treating them and then giving them back, which is an extremely expensive process," explains Drew Weissman, professor of medicine at the University of Pennsylvania, who was involved in developing the mRNA technology behind the COVID-19 vaccines.
But last November, a Massachusetts-based biotech company called Intellia Therapeutics showed it was possible to use mRNA to make the CRISPR system inside the body, eliminating the need to extract cells out of the body and edit them in a lab. Just as mRNA can instruct our cells to produce antibodies against a viral infection, it can also teach them to produce one of the two components that make up CRISPR — a cutting protein that snips out a problem gene.
"The pandemic has really shown that not only are mRNA approaches viable, they could in certain circumstances be vastly superior to more traditional technologies."
In Intellia's London-based clinical trial, the company applied this for the first time in a patient with a rare inherited liver disease known as hereditary transthyretin amyloidosis with polyneuropathy. The disease causes a toxic protein to build up in a person's organs and is typically fatal. In a company press release, Intellia's president and CEO John Leonard swiftly declared that its mRNA-based CRISPR therapy could usher in a "new era of potential genome editing cures."
Weissman predicts that turning CRISPR into an affordable therapy will become the next major frontier for mRNA over the coming decade. His lab is currently working on an mRNA-based CRISPR treatment for sickle cell disease. More than 300,000 babies are born with sickle cell every year, mainly in lower income nations.
"There is a FDA-approved cure, but it involves taking the bone marrow out of the person, and then giving it back which is prohibitively expensive," he says. It also requires a patient to have a matched bone marrow done. "We give an intravenous injection of mRNA lipid nanoparticles that target CRISPR to the bone marrow stem cells in the patient, which is easy, and much less expensive."
Cancer Immunotherapy
Meanwhile, the overwhelming success of the COVID-19 vaccines has focused attention on other ways of using mRNA to bolster the immune system against threats ranging from other infectious diseases to cancer.
The practicality of mRNA vaccines – relatively small quantities are required to induce an antibody response – coupled with their adaptable design, mean companies like Moderna are now targeting pathogens like Zika, chikungunya and cytomegalovirus, or CMV, which previously considered commercially unviable for vaccine developers. This is because outbreaks have been relatively sporadic, and these viruses mainly affect people in low-income nations who can't afford to pay premium prices for a vaccine. But mRNA technology means that jabs could be produced on a flexible basis, when required, at relatively low cost.
Other scientists suggest that mRNA could even provide a means of developing a universal influenza vaccine, a goal that's long been the Holy Grail for vaccinologists around the world.
"The mRNA technology allows you to pick out bits of the virus that you want to induce immunity to," says Michael Mulqueen, vice president of business development at eTheRNA, a Belgium-based biotech that's developing mRNA-based vaccines for malaria and HIV, as well as various forms of cancer. "This means you can get the immune system primed to the bits of the virus that don't vary so much between strains. So you could actually have a single vaccine that protects against a whole raft of different variants of the same virus, offering more universal coverage."
Before mRNA became synonymous with vaccines, its biggest potential was for cancer treatments. BioNTech, the German biotech company that collaborated with Pfizer to develop the first authorized COVID-19 vaccine, was initially founded to utilize mRNA for personalized cancer treatments, and the company remains interested in cancers ranging from melanoma to breast cancer.
One of the major hurdles in treating cancer has been the fact that tumors can look very different from one person to the next. It's why conventional approaches, such as chemotherapy or radiation, don't work for every patient. But weaponizing mRNA against cancer primes the immune cells with the tumor's specific genetic sequence, training the patient's body to attack their own unique type of cancer.
"It means you're able to think about personalizing cancer treatments down to specific subgroups of patients," says Mulqueen. "For example, eTheRNA are developing a renal cell carcinoma treatment which will be targeted at around 20% of these patients, who have specific tumor types. We're hoping to take that to human trials next year, but the challenge is trying to identify the right patients for the treatment at an early stage."
Repairing Damaged mRNA
While hopes are high that mRNA could usher in new cancer treatments and make CRISPR more accessible, a growing number of companies are also exploring an alternative to gene editing, known as RNA editing.
In genetic disorders, the mRNA in certain cells is impaired due to a rogue gene defect, and so the body ceases to produce a particular vital protein. Instead of permanently deleting the problem gene with CRISPR, the idea behind RNA editing is to inject small pieces of synthetic mRNA to repair the existing mRNA. Scientists think this approach will allow normal protein production to resume.
Over the past few years, this approach has gathered momentum, as some researchers have recognized that it holds certain key advantages over CRISPR. Companies from Belgium to Japan are now looking at RNA editing to treat all kinds of disorders, from Huntingdon's disease, to amyotrophic lateral sclerosis, or ALS, and certain types of cancer.
"With RNA editing, you don't need to make any changes to the DNA," explains Daniel de Boer, CEO of Dutch biotech ProQR, which is looking to treat rare genetic disorders that cause blindness. "Changes to the DNA are permanent, so if something goes wrong, that may not be desirable. With RNA editing, it's a temporary change, so we dose patients with our drugs once or twice a year."
Last month, ProQR reported a landmark case study, in which a patient with a rare form of blindness called Leber congenital amaurosis, which affects the retina at the back of the eye, recovered vision after three months of treatment.
"We have seen that this RNA therapy restores vision in people that were completely blind for a year or so," says de Boer. "They were able to see again, to read again. We think there are a large number of other genetic diseases we could go after with this technology. There are thousands of different mutations that can lead to blindness, and we think this technology can target approximately 25% of them."
Ultimately, there's likely to be a role for both RNA editing and CRISPR, depending on the disease. "I think CRISPR is ideally suited for illnesses where you would like to permanently correct a genetic defect," says Joshua Rosenthal of the Marine Biology Laboratory in Chicago. "Whereas RNA editing could be used to treat things like pain, where you might want to reset a neural circuit temporarily over a shorter period of time."
Much of this research has been accelerated by the COVID-19 pandemic, which has played a major role in bringing mRNA to the forefront of people's minds as a therapeutic.
"The pandemic has really shown that not only are mRNA approaches viable, they could in certain circumstances be vastly superior to more traditional technologies," says Mulqueen. "In the future, I would not be surprised if many of the top pharma products are mRNA derived."
Eight Big Medical and Science Trends to Watch in 2021
Promising developments underway include advancements in gene and cell therapy, better testing for COVID, and a renewed focus on climate change.
The world as we know it has forever changed. With a greater focus on science and technology than before, experts in the biotech and life sciences spaces are grappling with what comes next as SARS-CoV-2, the coronavirus that causes the COVID-19 illness, has spread and mutated across the world.
Even with vaccines being distributed, so much still remains unknown.
Jared Auclair, Technical Supervisor for the Northeastern University's Life Science Testing Center in Burlington, Massachusetts, guides a COVID testing lab that cranks out thousands of coronavirus test results per day. His lab is also focused on monitoring the quality of new cell and gene therapy products coming to the market.
Here are trends Auclair and other experts are watching in 2021.
Better Diagnostic Testing for COVID
Expect improvements in COVID diagnostic testing and the ability to test at home.
There are currently three types of coronavirus tests. The molecular test—also known as the RT-PCR test, detects the virus's genetic material, and is highly accurate, but it can take days to receive results. There are also antibody tests, done through a blood draw, designed to test whether you've had COVID in the past. Finally, there's the quick antigen test that isn't as accurate as the PCR test, but can identify if people are going to infect others.
Last month, Lucira Health secured the U.S. FDA Emergency Use Authorization for the first prescription molecular diagnostic test for COVID-19 that can be performed at home. On December 15th, the Ellume Covid-19 Home Test received authorization as the first over-the-counter COVID-19 diagnostic antigen test that can be done at home without a prescription. The test uses a nasal swab that is connected to a smartphone app and returns results in 15-20 minutes. Similarly, the BinaxNOW COVID-19 Ag Card Home Test received authorization on Dec. 16 for its 15-minute antigen test that can be used within the first seven days of onset of COIVD-19 symptoms.
Home testing has the possibility to impact the pandemic pretty drastically, Auclair says, but there are other considerations: the type and timing of test that is administered, how expensive is the test (and if it is financially feasible for the general public) and the ability of a home test taker to accurately administer the test.
"The vaccine roll-out will not eliminate the need for testing until late 2021 or early 2022."
Ideally, everyone would frequently get tested, but that would mean the cost of a single home test—which is expected to be around $30 or more—would need to be much cheaper, more in the $5 range.
Auclair expects "innovations in the diagnostic space to explode" with the need for more accurate, inexpensive, quicker COVID tests. Auclair foresees innovations to be at first focused on COVID point-of-care testing, but he expects improvements within diagnostic testing for other types of viruses and diseases too.
"We still need more testing to get the pandemic under control, likely over the next 12 months," Auclair says. "The vaccine roll-out will not eliminate the need for testing until late 2021 or early 2022."
Rise of mRNA-based Vaccines and Therapies
A year ago, vaccines weren't being talked about like they are today.
"But clearly vaccines are the talk of the town," Auclair says. "The reason we got a vaccine so fast was there was so much money thrown at it."
A vaccine can take more than 10 years to fully develop, according to the World Economic Forum. Prior to the new COVID vaccines, which were remarkably developed and tested in under a year, the fastest vaccine ever made was for mumps -- and it took four years.
"Normally you have to produce a protein. This is typically done in eggs. It takes forever," says Catherine Dulac, a neuroscientist and developmental biologist at Harvard University who won the 2021 Breakthrough Prize in Life Sciences. "But an mRNA vaccine just enabled [us] to skip all sorts of steps [compared with burdensome conventional manufacturing] and go directly to a product that can be injected into people."
Non-traditional medicines based on genetic research are in their infancy. With mRNA-based vaccines hitting the market for the first time, look for more vaccines to be developed for whatever viruses we don't currently have vaccines for, like dengue virus and Ebola, Auclair says.
"There's a whole bunch of things that could be explored now that haven't been thought about in the past," Auclair says. "It could really be a game changer."
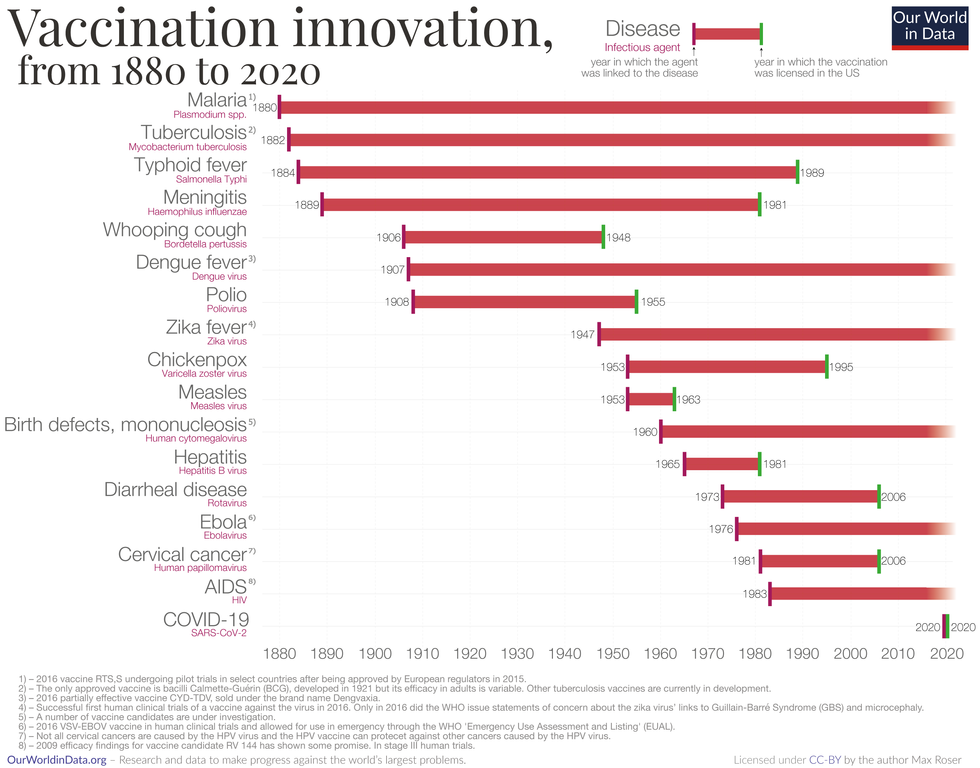
Vaccine Innovation over the last 140 years.
Max Roser/Our World in Data (Creative Commons license)
Advancements in Cell and Gene Therapies
CRISPR, a type of gene editing, is going to be huge in 2021, especially after the Nobel Prize in Chemistry was awarded to Emmanuelle Charpentier and Jennifer Doudna in October for pioneering the technology.
Right now, CRISPR isn't completely precise and can cause deletions or rearrangements of DNA.
"It's definitely not there yet, but over the next year it's going to get a lot closer and you're going to have a lot of momentum in this space," Auclair says. "CRISPR is one of the technologies I'm most excited about and 2021 is the year for it."
Gene therapies are typically used on rare genetic diseases. They work by replacing the faulty dysfunctional genes with corrected DNA codes.
"Cell and gene therapies are really where the field is going," Auclair says. "There is so much opportunity....For the first time in our life, in our existence as a species, we may actually be able to cure disease by using [techniques] like gene editing, where you cut in and out of pieces of DNA that caused a disease and put in healthy DNA," Auclair says.
For example, Spinal Muscular Atrophy is a rare genetic disorder that leads to muscle weakness, paralysis and death in children by age two. As of last year, afflicted children can take a gene therapy drug called Zolgensma that targets the missing or nonworking SMN1 gene with a new copy.
Another recent breakthrough uses gene editing for sickle cell disease. Victoria Gray, a mom from Mississippi who was exclusively followed by NPR, was the first person in the United States to be successfully treated for the genetic disorder with the help of CRISPR. She has continued to improve since her landmark treatment on July 2, 2019 and her once-debilitating pain has greatly eased.
"This is really a life-changer for me," she told NPR. "It's magnificent."
"You are going to see bigger leaps in gene therapies."
Look out also for improvements in cell therapies, but on a much lesser scale.
Cell therapies remove immune cells from a person or use cells from a donor. The cells are modified or cultured in lab, multiplied by the millions and then injected back into patients. These include stem cell therapies as well as CAR-T cell therapies, which are typically therapies of last resort and used in cancers like leukemia, Auclair says.
"You are going to see bigger leaps in gene therapies," Auclair says. "It's being heavily researched and we understand more about how to do gene therapies. Cell therapies will lie behind it a bit because they are so much more difficult to work with right now."
More Monoclonal Antibody Therapies
Look for more customized drugs to personalize medicine even more in the biotechnology space.
In 2019, the FDA anticipated receiving more than 200 Investigational New Drug (IND) applications in 2020. But with COVID, the number of INDs skyrocketed to 6,954 applications for the 2020 fiscal year, which ended September 30, 2020, according to the FDA's online tracker. Look for antibody therapies to play a bigger role.
Monoclonal antibodies are lab-grown proteins that mimic or enhance the immune system's response to fight off pathogens, like viruses, and they've been used to treat cancer. Now they are being used to treat patients with COVID-19.
President Donald Trump received a monoclonal antibody cocktail, called REGEN-COV2, which later received FDA emergency use authorization.
A newer type of monoclonal antibody therapy is Antibody-Drug Conjugates, also called ADCs. It's something we're going to be hearing a lot about in 2021, Auclair says.
"Antibody-Drug Conjugates is a monoclonal antibody with a chemical, we consider it a chemical warhead on it," Auclair says. "The monoclonal antibody binds to a specific antigen in your body or protein and delivers a chemical to that location and kills the infected cell."
Moving Beyond Male-Centric Lab Testing
Scientific testing for biology has, until recently, focused on testing males. Dulac, a Howard Hughes Medical Investigator and professor of molecular and cellular biology at Harvard University, challenged that idea to find brain circuitry behind sex-specific behaviors.
"For the longest time, until now, all the model systems in biology, are male," Dulac says. "The idea is if you do testing on males, you don't need to do testing on females."
Clinical models are done in male animals, as well as fundamental research. Because biological research is always done on male models, Dulac says the outcomes and understanding in biology is geared towards understanding male biology.
"All the drugs currently on the market and diagnoses of diseases are biased towards the understanding of male biology," Dulac says. "The diagnostics of diseases is way weaker in women than men."
That means the treatment isn't necessarily as good for women as men, she says, including what is known and understood about pain medication.
"So pain medication doesn't work well in women," Dulac says. "It works way better in men. It's true for almost all diseases that I know. Why? because you have a science that is dominated by males."
Although some in the scientific community challenge that females are not interesting or too complicated with their hormonal variations, Dulac says that's simply not true.
"There's absolutely no reason to decide 50% of life forms are interesting and the other 50% are not interesting. What about looking at both?" says Dulac, who was awarded the $3 million Breakthrough Prize in Life Sciences in September for connecting specific neural mechanisms to male and female parenting behaviors.
Disease Research on Single Cells
To better understand how diseases manifest in the body's cell and tissues, many researchers are looking at single-cell biology. Cells are the most fundamental building blocks of life. Much still needs to be learned.
"A remarkable development this year is the massive use of analysis of gene expression and chromosomal regulation at the single-cell level," Dulac says.
Much is focused on the Human Cell Atlas (HCA), a global initiative to map all cells in healthy humans and to better identify which genes associated with diseases are active in a person's body. Most estimates put the number of cells around 30 trillion.
Dulac points to work being conducted by the Cell Census Network (BICCN) Brain Initiative, an initiative by the National Institutes of Health to come up with an atlas of cell types in mouse, human and non-human primate brains, and the Chan Zuckerberg Initiative's funding of single-cell biology projects, including those focused on single-cell analysis of inflammation.
"Our body and our brain are made of a large number of cell types," Dulac says. "The ability to explore and identify differences in gene expression and regulation in massively multiplex ways by analyzing millions of cells is extraordinarily important."
Converting Plastics into Food
Yep, you heard it right, plastics may eventually be turned into food. The Defense Advanced Research Projects Agency, better known as DARPA, is funding a project—formally titled "Production of Macronutrients from Thermally Oxo-Degraded Wastes"—and asking researchers how to do this.
"When I first heard about this challenge, I thought it was absolutely absurd," says Dr. Robert Brown, director of the Bioeconomy Institute at Iowa State University and the project's principal investigator, who is working with other research partners at the University of Delaware, Sandia National Laboratories, and the American Institute of Chemical Engineering (AIChE)/RAPID Institute.
But then Brown realized plastics will slowly start oxidizing—taking in oxygen—and microorganisms can then consume it. The oxidation process at room temperature is extremely slow, however, which makes plastics essentially not biodegradable, Brown says.
That changes when heat is applied at brick pizza oven-like temperatures around 900-degrees Fahrenheit. The high temperatures get compounds to oxidize rapidly. Plastics are synthetic polymers made from petroleum—large molecules formed by linking many molecules together in a chain. Heated, these polymers will melt and crack into smaller molecules, causing them to vaporize in a process called devolatilization. Air is then used to cause oxidation in plastics and produce oxygenated compounds—fatty acids and alcohols—that microorganisms will eat and grow into single-cell proteins that can be used as an ingredient or substitute in protein-rich foods.
"The caveat is the microorganisms must be food-safe, something that we can consume," Brown says. "Like supplemental or nutritional yeast, like we use to brew beer and to make bread or is used in Australia to make Vegemite."
What do the microorganisms look like? For any home beer brewers, it's the "gunky looking stuff you'd find at the bottom after the fermentation process," Brown says. "That's cellular biomass. Like corn grown in the field, yeast or other microorganisms like bacteria can be harvested as macro-nutrients."
Brown says DARPA's ReSource program has challenged all the project researchers to find ways for microorganisms to consume any plastics found in the waste stream coming out of a military expeditionary force, including all the packaging of food and supplies. Then the researchers aim to remake the plastic waste into products soldiers can use, including food. The project is in the first of three phases.
"We are talking about polyethylene, polypropylene, like PET plastics used in water bottles and converting that into macronutrients that are food," says Brown.
Renewed Focus on Climate Change
The Union of Concerned Scientists say carbon dioxide levels are higher today than any point in at least 800,000 years.
"Climate science is so important for all of humankind. It is critical because the quality of life of humans on the planet depends on it."
Look for technology to help locate large-scale emitters of carbon dioxide, including sensors on satellites and artificial intelligence to optimize energy usage, especially in data centers.
Other technologies focus on alleviating the root cause of climate change: emissions of heat-trapping gasses that mainly come from burning fossil fuels.
Direct air carbon capture, an emerging effort to capture carbon dioxide directly from ambient air, could play a role.
The technology is in the early stages of development and still highly uncertain, says Peter Frumhoff, director of science and policy at Union of Concerned Scientists. "There are a lot of questions about how to do that at sufficiently low costs...and how to scale it up so you can get carbon dioxide stored in the right way," he says, and it can be very energy intensive.
One of the oldest solutions is planting new forests, or restoring old ones, which can help convert carbon dioxide into oxygen through photosynthesis. Hence the Trillion Trees Initiative launched by the World Economic Forum. Trees are only part of the solution, because planting trees isn't enough on its own, Frumhoff says. That's especially true, since 2020 was the year that human-made, artificial stuff now outweighs all life on earth.
More research is also going into artificial photosynthesis for solar fuels. The U.S. Department of Energy awarded $100 million in 2020 to two entities that are conducting research. Look also for improvements in battery storage capacity to help electric vehicles, as well as back-up power sources for solar and wind power, Frumhoff says.
Another method to combat climate change is solar geoengineering, also called solar radiation management, which reflects sunlight back to space. The idea stems from a volcanic eruption in 1991 that released a tremendous amount of sulfate aerosol particles into the stratosphere, reflecting the sunlight away from Earth. The planet cooled by a half degree for nearly a year, Frumhoff says. However, he acknowledges, "there's a lot of things we don't know about the potential impacts and risks" involved in this controversial approach.
Whatever the approach, scientific solutions to climate change are attracting renewed attention. Under President Trump, the White House Office of Science and Technology Policy didn't have an acting director for almost two years. Expect that to change when President-elect Joe Biden takes office.
"Climate science is so important for all of humankind," Dulac says. "It is critical because the quality of life of humans on the planet depends on it."
The Brave New World of Using DNA to Store Data
A panoramic view of DNA
Netscape co-founder-turned-venture capitalist billionaire investor Marc Andreessen once posited that software was eating the world. He was right, and the takeover of software resulted in many things. One of them is data. Lots and lots and lots of data. In the previous two years, humanity created more data than it did during its entire existence combined, and the amount will only increase. Think about it: The hundreds of 50KB emails you write a day, the dozens of 10MB photos, the minute-long, 350MB 4K video you shoot on your iPhone X add up to vast quantities of information. All that information needs to be stored. And that's becoming an issue as data volume outpaces storage space.
The race is on to find another medium capable of storing massive amounts of information in as small a space as possible.
"There won't be enough silicon to store all the data we need. It's unlikely that we can make flash memory smaller. We have reached the physical limits," Victor Zhirnov, chief scientist at the Semiconductor Research Corporation, says. "We are facing a crisis that's comparable to the oil crisis in the 1970s. By 2050, we're going to need to store 10 to the 30 bits, compared to 10 to the 23 bits in 2016." That amount of storage space is equivalent to each of the world's seven billion people owning almost six trillion -- that's 10 to the 12th power -- iPhone Xs with 256GB storage space.
The race is on to find another medium capable of storing massive amounts of information in as small a space as possible. Zhirnov and other scientists are looking at the human body, looking to DNA. "Nature has nailed it," Luis Ceze, a professor in the Department of Computer Science and Engineering at the University of Washington, says. "DNA is a molecular storage medium that is remarkable. It's incredibly dense, many, many thousands of times denser than the densest technology that we have today. And DNA is remarkably general. Any information you can map in bits you can store in DNA." It's so dense -- able to store a theoretical maximum of 215 petabytes (215 million gigabytes) in a single gram -- that all the data ever produced could be stored in the back of a tractor trailer truck.
Writing DNA can be an energy-efficient process, too. Consider how the human body is constantly writing and rewriting DNA, and does so on a couple thousand calories a day. And all it needs for storage is a cool, dark place, a significant energy savings when compared to server farms that require huge amounts of energy to run and even more energy to cool.
Picture it: tiny specks of inert DNA made from silicon or another material, stored in cool, dark, dry areas, preserved for all time.
Researchers first succeeded in encoding data onto DNA in 2012, when Harvard University geneticists George Church and Sri Kosuri wrote a 52,000-word book on A, C, G, and T base pairs. Their method only produced 1.28 petabytes per gram of DNA, however, a volume exceeded the next year when a group encoded all 154 Shakespeare sonnets and a 26-second clip of Martin Luther King's "I Have A Dream" speech. In 2017, Columbia University researchers Yaniv Erlich and Dina Zielinski made the process 60 percent more efficient.
The limiting factor today is cost. Erlich said the work his team did cost $7,000 to encode and decode two megabytes of data. To become useful in a widespread way, the price per megabyte needs to plummet. Even advocates concede this point. "Of course it is expensive," Zhirnov says. "But look how much magnetic storage cost in the 1980s. What you store today in your iPhone for virtually nothing would cost many millions of dollars in 1982." There's reason to think the price will continue to fall. Genome readers are improving, getting cheaper, faster, and smaller, and genome sequencing becomes cheaper every year, too. Picture it: tiny specks of inert DNA made from silicon or another material, stored in cool, dark, dry areas, preserved for all time.
"It just takes a few minutes to double a sample. A few more minutes, you double it again. Very quickly, you have thousands or millions of new copies."
Plus, DNA has another advantage over more traditional forms of storage: It's very easy to reproduce. "If you want a second copy of a hard disk drive, you need components for a disk drive, hook both drives up to a computer, and copy. That's a pain," Nick Goldman, a researcher at the European Bioinformatics Institute, says. "DNA, once you have that first sample, it's a process that is absolutely routine in thousands of laboratories around the world to multiply that using polymerase chain reaction [which uses temperature changes or other processes]. It just takes a few minutes to double a sample. A few more minutes, you double it again. Very quickly, you have thousands or millions of new copies."
This ability to duplicate quickly and easily is a positive trait. But, of course, there's also the potential for danger. Does encoding on DNA, the very basis for life, present ethical issues? Could it get out of control and fundamentally alter life as we know it?
The chance is there, but it's remote. The first reason is that storage could be done with only two base pairs, which would serve as replacements for the 0 and 1 digits that make up all digital data. While doing so would decrease the possible density of the storage, it would virtually eliminate the risk that the sequences would be compatible with life.
But even if scientists and researchers choose to use four base pairs, other safeguards are in place that will prevent trouble. According to Ceze, the computer science professor, the snippets of DNA that they write are very short, around 150 nucleotides. This includes the title, the information that's being encoded, and tags to help organize where the snippet should fall in the larger sequence. Furthermore, they generally avoid repeated letters, which dramatically reduces the chance that a protein could be synthesized from the snippet.
"In the future, we'll know enough about someone from a sample of their DNA that we could make a specific poison. That's the danger, not those of us who want to encode DNA for storage."
Inevitably, some DNA will get spilt. "But it's so unlikely that anything that gets created for storage would have a biological interpretation that could interfere with the mechanisms going on in a living organism that it doesn't worry me in the slightest," Goldman says. "We're not of concern for the people who are worried about the ethical issues of synthetic DNA. They are much more concerned about people deliberately engineering anthrax. In the future, we'll know enough about someone from a sample of their DNA that we could make a specific poison. That's the danger, not those of us who want to encode DNA for storage."
In the end, the reality of and risks surrounding encoding on DNA are the same as any scientific advancement: It's another system that is vulnerable to people with bad intentions but not one that is inherently unethical.
"Every human action has some ethical implications," Zhirnov says. "I can use a hammer to build a house or I can use it to harm another person. I don't see why DNA is in any way more or less ethical."
If that house can store all the knowledge in human history, it's worth learning how to build it.
Editor's Note: In response to readers' comments that silicon is one of the earth's most abundant materials, we reached back out to our source, Dr. Victor Zhirnov. He stands by his statement about a coming shortage of silicon, citing this research. The silicon oxide found in beach sand is unsuitable for semiconductors, he says, because the cost of purifying it would be prohibitive. For use in circuit-making, silicon must be refined to a purity of 99.9999999 percent. So the process begins by mining for pure quartz, which can only be found in relatively few places around the world.


