Blood Money: Paying for Convalescent Plasma to Treat COVID-19
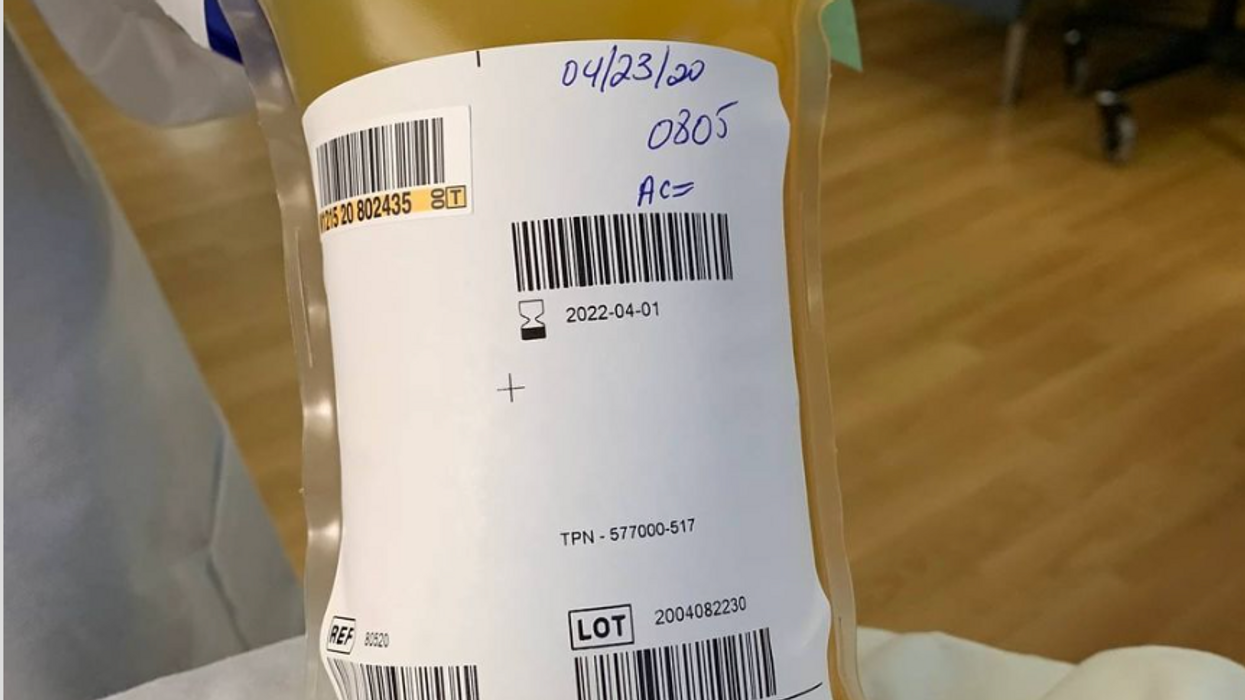
A bag of plasma that Tom Hanks donated back in April 2020 after his coronavirus infection. (He was not paid to donate.)
Convalescent plasma – first used to treat diphtheria in 1890 – has been dusted off the shelf to treat COVID-19. Does it work? Should we rely strictly on the altruism of donors or should people be paid for it?
The biologic theory is that a person who has recovered from a disease has chemicals in their blood, most likely antibodies, that contributed to their recovery, and transferring those to a person who is sick might aid their recovery. Whole blood won't work because there are too few antibodies in a single unit of blood and the body can hold only so much of it.
Plasma comprises about 55 percent of whole blood and is what's left once you take out the red blood cells that carry oxygen and the white blood cells of the immune system. Most of it is water but the rest is a complex mix of fats, salts, signaling molecules and proteins produced by the immune system, including antibodies.
A process called apheresis circulates the donors' blood through a machine that separates out the desired parts of blood and returns the rest to the donor. It takes several times the length of a regular whole blood donation to cycle through enough blood for the process. The end product is a yellowish concentration called convalescent plasma.
Recent History
It was used extensively during the great influenza epidemic off 1918 but fell out of favor with the development of antibiotics. Still, whenever a new disease emerges – SARS, MERS, Ebola, even antibiotic-resistant bacteria – doctors turn to convalescent plasma, often as a stopgap until more effective antibiotic and antiviral drugs are developed. The process is certainly safe when standard procedures for handling blood products are followed, and historically it does seem to be beneficial in at least some patients if administered early enough in the disease.
With few good treatment options for COVID-19, doctors have given convalescent plasma to more than a hundred thousand Americans and tens of thousand of people elsewhere, to mixed results. Placebo-controlled trials could give a clearer picture of plasma's value but it is difficult to enroll patients facing possible death when the flip of a coin will determine who will receive a saline solution or plasma.
And the plasma itself isn't some uniform pill stamped out in a factory, it's a natural product that is shaped by the immune history of the donor's body and its encounter not just with SARS-CoV-2 but a lifetime of exposure to different pathogens.
Researchers believe antibodies in plasma are a key factor in directly fighting the virus. But the variety and quantity of antibodies vary from donor to donor, and even over time from the same donor because once the immune system has cleared the virus from the body, it stops putting out antibodies to fight the virus. Often the quality and quantity of antibodies being given to a patient are not measured, making it somewhat hit or miss, which is why several companies have recently developed monoclonal antibodies, a single type of antibody found in blood that is effective against SARS-CoV-2 and that is multiplied in the lab for use as therapy.
Plasma may also contain other unknown factors that contribute to fighting disease, say perhaps signaling molecules that affect gene expression, which might affect the movement of immune cells, their production of antiviral molecules, or the regulation of inflammation. The complexity and lack of standardization makes it difficult to evaluate what might be working or not with a convalescent plasma treatment. Thus researchers are left with few clues about how to make it more effective.
Industrializing Plasma
Many Americans living along the border with Mexico regularly head south to purchase prescription drugs at a significant discount. Less known is the medical traffic the other way, Mexicans who regularly head north to be paid for plasma donations, which are prohibited in their country; the U.S. allows payment for plasma donations but not whole blood. A typical payment is about $35 for a donation but the sudden demand for convalescent plasma from people who have recovered from COVID-19 commands a premium price, sometimes as high as $200. These donors are part of a fast-growing plasma industry that surpassed $25 billion in 2018. The U.S. supplies about three-quarters of the world's needs for plasma.
Payment for whole blood donation in the U.S. is prohibited, and while payment for plasma is allowed, there is a stigma attached to payment and much plasma is donated for free.
The pharmaceutical industry has shied away from natural products they cannot patent but they have identified simpler components from plasma, such as clotting factors and immunoglobulins, that have been turned into useful drugs from this raw material of plasma. While some companies have retooled to provide convalescent plasma to treat COVID-19, often paying those donors who have recovered a premium of several times the normal rate, most convalescent plasma has come as donations through traditional blood centers.
In April the Mayo Clinic, in cooperation with the FDA, created an expanded access program for convalescent plasma to treat COVID-19. It was meant to reduce the paperwork associated with gaining access to a treatment not yet approved by the FDA for that disease. Initially it was supposed to be for 5000 units but it quickly grew to more than twenty times that size. Michael Joyner, the head of the program, discussed that experience in an extended interview in September.
The Centers for Medicare and Medicaid Services (CMS) also created associated reimbursement codes, which became permanent in August.
Mayo published an analysis of the first 35,000 patients as a preprint in August. It concluded, "The relationships between mortality and both time to plasma transfusion, and antibody levels provide a signature that is consistent with efficacy for the use of convalescent plasma in the treatment of hospitalized COVID-19 patients."
It seemed to work best when given early in infection and in larger doses; a similar pattern has been seen in studies of monoclonal antibodies. A revised version will soon be published in a major medical journal. Some criticized the findings as not being from a randomized clinical trial.
Convalescent plasma is not the only intervention that seems to work better when used earlier in the course of disease. Recently the pharmaceutical company Eli Lilly stopped a clinical trial of a monoclonal antibody in hospitalized COVID-19 patients when it became apparent it wasn't helping. It is continuing trials for patients who are less sick and begin treatment earlier, as well as in persons who have been exposed to the virus but not yet diagnosed as infected, to see if it might prevent infection. In November the FDA eased access to this drug outside of clinical trials, though it is not yet approved for sale.
Show Me the Money
The antibodies that seem to give plasma its curative powers are fragile proteins that the body produces to fight the virus. Production shuts down once the virus is cleared and the remaining antibodies survive only for a few weeks before the levels fade. [Vaccines are used to train immune cells to produce antibodies and other defenses to respond to exposure to future pathogens.] So they can be usefully harvested from a recovered patient for only a few short weeks or months before they decline precipitously. The question becomes, how does one mobilize this resource in that short window of opportunity?
The program run by the Mayo Clinic explains the process and criteria for donating convalescent plasma for COVID-19, as well as links to local blood centers equipped to handle those free donations. Commercial plasma centers also are advertising and paying for donations.
A majority of countries prohibit paying donors for blood or blood products, including India. But an investigation by India Today touted a black market of people willing to donate convalescent plasma for the equivalent of several hundred dollars. Officials vowed to prosecute, saying donations should be selfless.
But that enforcement threat seemed to be undercut when the health minister of the state of Assam declared "plasma donors will get preference in several government schemes including the government job interview." It appeared to be a form of compensation that far surpassed simple cash.
The small city of Rexburg, Idaho, with a population a bit over 50,000, overwhelmingly Mormon and home to a campus of Brigham Young University, at one point had one of the highest per capita rates of COVID-19 in the current wave of infection. Rumors circulated that some students were intentionally trying to become infected so they could later sell their plasma for top dollar, potentially as much as $200 a visit.
Troubled university officials investigated the allegations but could come up with nothing definitive; how does one prove intentionality with such an omnipresent yet elusive virus? They chalked it up to idle chatter, perhaps an urban legend, which might be associated with alcohol use on some other campus.
Doctors, hospitals, and drug companies are all rightly praised for their altruism in the fight against COVID-19, but they also get paid. Payment for whole blood donation in the U.S. is prohibited, and while payment for plasma is allowed, there is a stigma attached to payment and much plasma is donated for free. "Why do we expect the donors [of convalescent plasma] to be the only uncompensated people in the process? It really makes no sense," argues Mark Yarborough, an ethicist at the UC Davis School of Medicine in Sacramento.
"When I was in grad school, two of my closest friends, at least once a week they went and gave plasma. That was their weekend spending money," Yarborough recalls. He says upper and middle-income people may have the luxury of donating blood products but prohibiting people from selling their plasma is a bit paternalistic and doesn't do anything to improve the economic status of poor people.
"Asking people to dedicate two hours a week for an entire year in exchange for cookies and milk is demonstrably asking too much," says Peter Jaworski, an ethicist who teaches at Georgetown University.
He notes that companies that pay plasma donors have much lower total costs than do operations that rely solely on uncompensated donations. The companies have to spend less to recruit and retain donors because they increase payments to encourage regular repeat donations. They are able to more rationally schedule visits to maximize use of expensive apheresis equipment and medical personnel used for the collection.
It seems that COVID-19 has been with us forever, but in reality it is less than a year. We have learned much over that short time, can now better manage the disease, and have lower mortality rates to prove it. Just how much convalescent plasma may have contributed to that remains an open question. Access to vaccines is months away for many people, and even then some people will continue to get sick. Given the lack of proven treatments, it makes sense to keep plasma as part of the mix, and not close the door to any legitimate means to obtain it.
Autonomous, indoor farming gives a boost to crops
Artificial Intelligence is already helping to grow some of the food we eat.
The glass-encased cabinet looks like a display meant to hold reasonably priced watches, or drugstore beauty creams shipped from France. But instead of this stagnant merchandise, each of its five shelves is overgrown with leaves — moss-soft pea sprouts, spikes of Lolla rosa lettuces, pale bok choy, dark kale, purple basil or red-veined sorrel or green wisps of dill. The glass structure isn’t a cabinet, but rather a “micro farm.”
The gadget is on display at the Richmond, Virginia headquarters of Babylon Micro-Farms, a company that aims to make indoor farming in the U.S. more accessible and sustainable. Babylon’s soilless hydroponic growing system, which feeds plants via nutrient-enriched water, allows chefs on cruise ships, cafeterias and elsewhere to provide home-grown produce to patrons, just seconds after it’s harvested. Currently, there are over 200 functioning systems, either sold or leased to customers, and more of them are on the way.
The chef-farmers choose from among 45 types of herb and leafy-greens seeds, plop them into grow trays, and a few weeks later they pick and serve. While success is predicated on at least a small amount of these humans’ care, the systems are autonomously surveilled round-the-clock from Babylon’s base of operations. And artificial intelligence is helping to run the show.
Babylon piloted the use of specialized cameras that take pictures in different spectrums to gather some less-obvious visual data about plants’ wellbeing and alert people if something seems off.
Imagine consistently perfect greens and tomatoes and strawberries, grown hyper-locally, using less water, without chemicals or environmental contaminants. This is the hefty promise of controlled environment agriculture (CEA) — basically, indoor farms that can be hydroponic, aeroponic (plant roots are suspended and fed through misting), or aquaponic (where fish play a role in fertilizing vegetables). But whether they grow 4,160 leafy-green servings per year, like one Babylon farm, or millions of servings, like some of the large, centralized facilities starting to supply supermarkets across the U.S., they seek to minimize failure as much as possible.

Babylon’s soilless hydroponic growing system
Courtesy Babylon Micro-Farms
Here, AI is starting to play a pivotal role. CEA growers use it to help “make sense of what’s happening” to the plants in their care, says Scott Lowman, vice president of applied research at the Institute for Advanced Learning and Research (IALR) in Virginia, a state that’s investing heavily in CEA companies. And although these companies say they’re not aiming for a future with zero human employees, AI is certainly poised to take a lot of human farming intervention out of the equation — for better and worse.
Most of these companies are compiling their own data sets to identify anything that might block the success of their systems. Babylon had already integrated sensor data into its farms to measure heat and humidity, the nutrient content of water, and the amount of light plants receive. Last year, they got a National Science Foundation grant that allowed them to pilot the use of specialized cameras that take pictures in different spectrums to gather some less-obvious visual data about plants’ wellbeing and alert people if something seems off. “Will this plant be healthy tomorrow? Are there things…that the human eye can't see that the plant starts expressing?” says Amandeep Ratte, the company’s head of data science. “If our system can say, Hey, this plant is unhealthy, we can reach out to [users] preemptively about what they’re doing wrong, or is there a disease at the farm?” Ratte says. The earlier the better, to avoid crop failures.

Natural light accounts for 70 percent of Greenswell Growers’ energy use on a sunny day.
Courtesy Greenswell Growers
IALR’s Lowman says that other CEA companies are developing their AI systems to account for the different crops they grow — lettuces come in all shapes and sizes, after all, and each has different growing needs than, for example, tomatoes. The ways they run their operations differs also. Babylon is unusual in its decentralized structure. But centralized growing systems with one main location have variabilities, too. AeroFarms, which recently declared bankruptcy but will continue to run its 140,000-square foot vertical operation in Danville, Virginia, is entirely enclosed and reliant on the intense violet glow of grow lights to produce microgreens.
Different companies have different data needs. What data is essential to AeroFarms isn’t quite the same as for Greenswell Growers located in Goochland County, Virginia. Raising four kinds of lettuce in a 77,000-square-foot automated hydroponic greenhouse, the vagaries of naturally available light, which accounts for 70 percent of Greenswell’s energy use on a sunny day, affect operations. Their tech needs to account for “outside weather impacts,” says president Carl Gupton. “What adjustments do we have to make inside of the greenhouse to offset what's going on outside environmentally, to give that plant optimal conditions? When it's 85 percent humidity outside, the system needs to do X, Y and Z to get the conditions that we want inside.”
AI will help identify diseases, as well as when a plant is thirsty or overly hydrated, when it needs more or less calcium, phosphorous, nitrogen.
Nevertheless, every CEA system has the same core needs — consistent yield of high quality crops to keep up year-round supply to customers. Additionally, “Everybody’s got the same set of problems,” Gupton says. Pests may come into a facility with seeds. A disease called pythium, one of the most common in CEA, can damage plant roots. “Then you have root disease pressures that can also come internally — a change in [growing] substrate can change the way the plant performs,” Gupton says.
AI will help identify diseases, as well as when a plant is thirsty or overly hydrated, when it needs more or less calcium, phosphorous, nitrogen. So, while companies amass their own hyper-specific data sets, Lowman foresees a time within the next decade “when there will be some type of [open-source] database that has the most common types of plant stress identified” that growers will be able to tap into. Such databases will “create a community and move the science forward,” says Lowman.
In fact, IALR is working on assembling images for just such a database now. On so-called “smart tables” inside an Institute lab, a team is growing greens and subjects them to various stressors. Then, they’re administering treatments while taking images of every plant every 15 minutes, says Lowman. Some experiments generate 80,000 images; the challenge lies in analyzing and annotating the vast trove of them, marking each one to reflect outcome—for example increasing the phosphate delivery and the plant’s response to it. Eventually, they’ll be fed into AI systems to help them learn.
For all the enthusiasm surrounding this technology, it’s not without downsides. Training just one AI system can emit over 250,000 pounds of carbon dioxide, according to MIT Technology Review. AI could also be used “to enhance environmental benefit for CEA and optimize [its] energy consumption,” says Rozita Dara, a computer science professor at the University of Guelph in Canada, specializing in AI and data governance, “but we first need to collect data to measure [it].”

The chef-farmers can choose from 45 types of herb and leafy-greens seeds.
Courtesy Babylon Micro-Farms
Any system connected to the Internet of Things is also vulnerable to hacking; if CEA grows to the point where “there are many of these similar farms, and you're depending on feeding a population based on those, it would be quite scary,” Dara says. And there are privacy concerns, too, in systems where imaging is happening constantly. It’s partly for this reason, says Babylon’s Ratte, that the company’s in-farm cameras all “face down into the trays, so the only thing [visible] is pictures of plants.”
Tweaks to improve AI for CEA are happening all the time. Greenswell made its first harvest in 2022 and now has annual data points they can use to start making more intelligent choices about how to feed, water, and supply light to plants, says Gupton. Ratte says he’s confident Babylon’s system can already “get our customers reliable harvests. But in terms of how far we have to go, it's a different problem,” he says. For example, if AI could detect whether the farm is mostly empty—meaning the farm’s user hasn’t planted a new crop of greens—it can alert Babylon to check “what's going on with engagement with this user?” Ratte says. “Do they need more training? Did the main person responsible for the farm quit?”
Lowman says more automation is coming, offering greater ability for systems to identify problems and mitigate them on the spot. “We still have to develop datasets that are specific, so you can have a very clear control plan, [because] artificial intelligence is only as smart as what we tell it, and in plant science, there's so much variation,” he says. He believes AI’s next level will be “looking at those first early days of plant growth: when the seed germinates, how fast it germinates, what it looks like when it germinates.” Imaging all that and pairing it with AI, “can be a really powerful tool, for sure.”
Scientists make progress with growing organs for transplants
Researchers from the University of Cambridge have laid the foundations for growing synthetic embryos that could develop a beating heart, gut and brain.
Story by Big Think
For over a century, scientists have dreamed of growing human organs sans humans. This technology could put an end to the scarcity of organs for transplants. But that’s just the tip of the iceberg. The capability to grow fully functional organs would revolutionize research. For example, scientists could observe mysterious biological processes, such as how human cells and organs develop a disease and respond (or fail to respond) to medication without involving human subjects.
Recently, a team of researchers from the University of Cambridge has laid the foundations not just for growing functional organs but functional synthetic embryos capable of developing a beating heart, gut, and brain. Their report was published in Nature.
The organoid revolution
In 1981, scientists discovered how to keep stem cells alive. This was a significant breakthrough, as stem cells have notoriously rigorous demands. Nevertheless, stem cells remained a relatively niche research area, mainly because scientists didn’t know how to convince the cells to turn into other cells.
Then, in 1987, scientists embedded isolated stem cells in a gelatinous protein mixture called Matrigel, which simulated the three-dimensional environment of animal tissue. The cells thrived, but they also did something remarkable: they created breast tissue capable of producing milk proteins. This was the first organoid — a clump of cells that behave and function like a real organ. The organoid revolution had begun, and it all started with a boob in Jello.
For the next 20 years, it was rare to find a scientist who identified as an “organoid researcher,” but there were many “stem cell researchers” who wanted to figure out how to turn stem cells into other cells. Eventually, they discovered the signals (called growth factors) that stem cells require to differentiate into other types of cells.
For a human embryo (and its organs) to develop successfully, there needs to be a “dialogue” between these three types of stem cells.
By the end of the 2000s, researchers began combining stem cells, Matrigel, and the newly characterized growth factors to create dozens of organoids, from liver organoids capable of producing the bile salts necessary for digesting fat to brain organoids with components that resemble eyes, the spinal cord, and arguably, the beginnings of sentience.
Synthetic embryos
Organoids possess an intrinsic flaw: they are organ-like. They share some characteristics with real organs, making them powerful tools for research. However, no one has found a way to create an organoid with all the characteristics and functions of a real organ. But Magdalena Żernicka-Goetz, a developmental biologist, might have set the foundation for that discovery.
Żernicka-Goetz hypothesized that organoids fail to develop into fully functional organs because organs develop as a collective. Organoid research often uses embryonic stem cells, which are the cells from which the developing organism is created. However, there are two other types of stem cells in an early embryo: stem cells that become the placenta and those that become the yolk sac (where the embryo grows and gets its nutrients in early development). For a human embryo (and its organs) to develop successfully, there needs to be a “dialogue” between these three types of stem cells. In other words, Żernicka-Goetz suspected the best way to grow a functional organoid was to produce a synthetic embryoid.
As described in the aforementioned Nature paper, Żernicka-Goetz and her team mimicked the embryonic environment by mixing these three types of stem cells from mice. Amazingly, the stem cells self-organized into structures and progressed through the successive developmental stages until they had beating hearts and the foundations of the brain.
“Our mouse embryo model not only develops a brain, but also a beating heart [and] all the components that go on to make up the body,” said Żernicka-Goetz. “It’s just unbelievable that we’ve got this far. This has been the dream of our community for years and major focus of our work for a decade and finally we’ve done it.”
If the methods developed by Żernicka-Goetz’s team are successful with human stem cells, scientists someday could use them to guide the development of synthetic organs for patients awaiting transplants. It also opens the door to studying how embryos develop during pregnancy.
This article originally appeared on Big Think, home of the brightest minds and biggest ideas of all time.


