Can Spare Parts from Pigs Solve Our Organ Shortage?
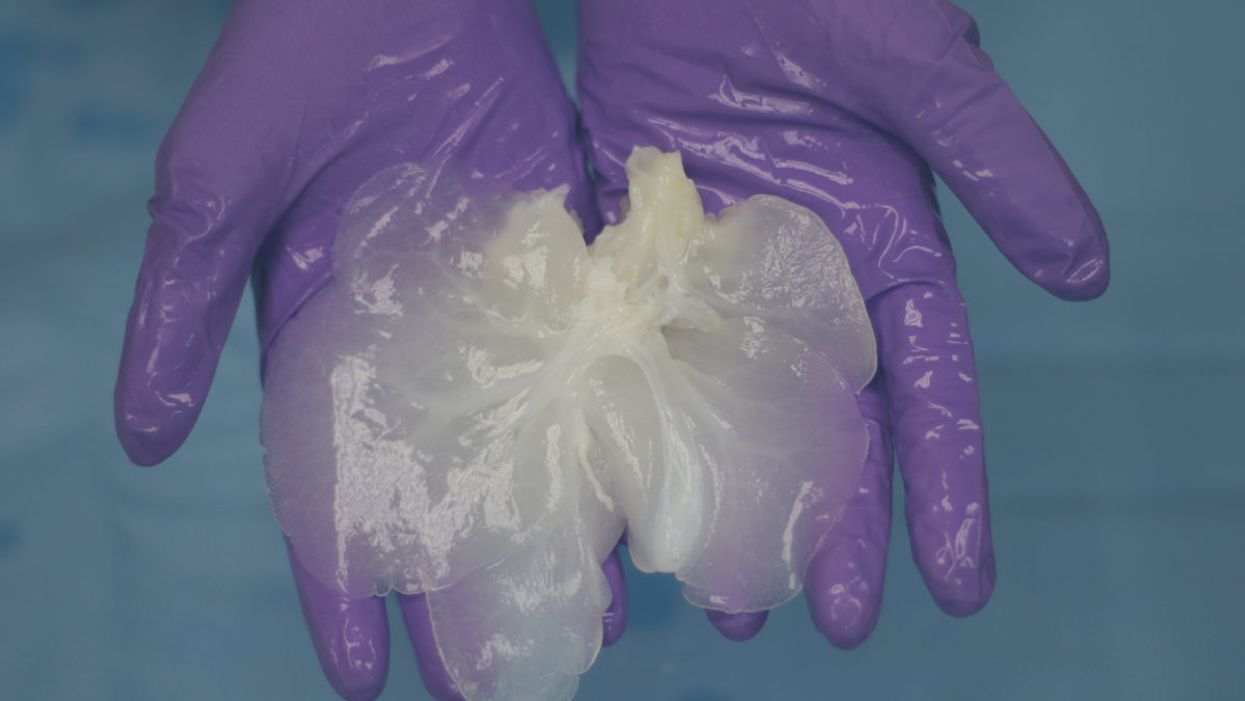
A decellularized small porcine liver.
Jennifer Cisneros was 18 years old, commuting to college from her family's home outside Annapolis, Maryland, when she came down with what she thought was the flu. Over the following weeks, however, her fatigue and nausea worsened, and her weight began to plummet. Alarmed, her mother took her to see a pediatrician. "When I came back with the urine cup, it was orange," Cisneros recalls. "He was like, 'Oh, my God. I've got to send you for blood work.'"
"Eventually, we'll be better off than with a human organ."
Further tests showed that her kidneys were failing, and at Johns Hopkins Hospital, a biopsy revealed the cause: Goodpasture syndrome (GPS), a rare autoimmune disease that attacks the kidneys or lungs. Cisneros was put on dialysis to filter out the waste products that her body could no longer process, and given chemotherapy and steroids to suppress her haywire immune system.
The treatment drove her GPS into remission, but her kidneys were beyond saving. At 19, Cisneros received a transplant, with her mother as donor. Soon, she'd recovered enough to return to school; she did some traveling, and even took up skydiving and parasailing. Then, after less than two years, rejection set in, and the kidney had to be removed.
She went back on dialysis until she was 26, when a stranger learned of her plight and volunteered to donate. That kidney lasted four years, but gave out after a viral infection. Since 2015, Cisneros—now 32, and working as an office administrator between thrice-weekly blood-filtering sessions—has been waiting for a replacement.
She's got plenty of company. About 116,000 people in the United States currently need organ transplants, but fewer than 35,000 organs become available every year. On average, 20 people on the waiting list die each day. And despite repeated campaigns to boost donorship, the gap shows no sign of narrowing.
"This is going to revolutionize medicine, in ways we probably can't yet appreciate."
For decades, doctors and scientists have envisioned a radical solution to the shortage: harvesting other species for spare parts. Xenotransplantation, as the practice is known, could provide an unlimited supply of lifesaving organs for patients like Cisneros. Those organs, moreover, could be altered by genetic engineering or other methods to reduce the danger of rejection—and thus to eliminate the need for immunosuppressive drugs, whose potential side effects include infections, diabetes, and cancer. "Eventually, we'll be better off than with a human organ," says David Cooper, MD, PhD, co-director of the xenotransplant program at the University of Alabama School of Medicine. "This is going to revolutionize medicine, in ways we probably can't yet appreciate."
Recently, progress toward that revolution has accelerated sharply. The cascade of advances began in April 2016, when researchers at the National Heart, Lung, and Blood Institute (NHLBI) reported keeping pig hearts beating in the abdomens of five baboons for a record-breaking mean of 433 days, with one lasting more than two-and-a-half years. Then a team at Emory University announced that a pig kidney sustained a rhesus monkey for 435 days before being rejected, nearly doubling the previous record. At the University of Munich, in Germany, researchers doubled the record for a life-sustaining pig heart transplant in a baboon (replacing the animal's own heart) to 90 days. Investigators at the Salk Institute and the University of California, Davis, declared that they'd grown tissue in pig embryos using human stem cells—a first step toward cultivating personalized replacement organs. The list goes on.
Such breakthroughs, along with a surge of cash from biotech investors, have propelled a wave of bullish media coverage. Yet this isn't the first time that xenotransplantation has been touted as the next big thing. Twenty years ago, the field seemed poised to overcome its final hurdles, only to encounter a setback from which it is just now recovering.
Which raises a question: Is the current excitement justified? Or is the hype again outrunning the science?
A History of Setbacks
The idea behind xenotransplantation dates back at least as far as the 17th century, when French physician Jean-Baptiste Denys tapped the veins of sheep and cows to perform the first documented human blood transfusions. (The practice was banned after two of the four patients died, probably from an immune reaction.) In the 19th century, surgeons began transplanting corneas from pigs and other animals into humans, and using skin xenografts to aid in wound healing; despite claims of miraculous cures, medical historians believe those efforts were mostly futile. In the 1920s and '30s, thousands of men sought renewed vigor through testicular implants from monkeys or goats, but the fad collapsed after studies showed the effects to be imaginary.
Research shut down when scientists discovered a virus in pig DNA that could infect human cells.
After the first successful human organ transplant in 1954—of a kidney, passed between identical twin sisters—the limited supply of donor organs brought a resurgence of interest in animal sources. Attention focused on nonhuman primates, our species' closest evolutionary relatives. At Tulane University, surgeon Keith Reemstma performed the first chimpanzee-to-human kidney transplants in 1963 and '64. Although one of the 13 patients lived for nine months, the rest died within a few weeks due to organ rejection or infections. Other surgeons attempted liver and heart xenotransplants, with similar results. Even the advent of the first immunosuppressant drug, cyclosporine, in 1983, did little to improve survival rates.
In the 1980s, Cooper—a pioneering heart transplant surgeon who'd embraced the dream of xenotransplantation—began arguing that apes and monkeys might not be the best donor animals after all. "First of all, there's not enough of them," he explains. "They breed in ones and twos, and take years to grow to full size. Even then, their hearts aren't big enough for a 70-kg. patient." Pigs, he suggested, would be a more practical alternative. But when he tried transplanting pig organs into nonhuman primates (as surrogates for human recipients), they were rejected within minutes.
In 1992, Cooper's team identified a sugar on the surface of porcine cells, called alpha-1,3-galactose (a-gal), as the main target for the immune system's attack. By then, the first genetically modified pigs had appeared, and biotech companies—led by the Swiss-based pharma giant Novartis—began pouring millions of dollars into developing one whose organs could elude or resist the human body's defenses.
Disaster struck five years later, when scientists reported that a virus whose genetic code was written into pig DNA could infect human cells in lab experiments. Although there was no evidence that the virus, known as PERV (for porcine endogenous retrovirus) could cause disease in people, the discovery stirred fears that xenotransplants might unleash a deadly epidemic. Facing scrutiny from government regulators and protests from anti-GMO and animal-rights activists, Novartis "pulled out completely," Cooper recalls. "They slaughtered all their pigs and closed down their research facility." Competitors soon followed suit.
The riddles surrounding animal-to-human transplants are far from fully solved.
A New Chapter – With New Questions
Yet xenotransplantation's visionaries labored on, aided by advances in genetic engineering and immunosuppression, as well as in the scientific understanding of rejection. In 2003, a team led by Cooper's longtime colleague David Sachs, at Harvard Medical School, developed a pig lacking the gene for a-gal; over the next few years, other scientists knocked out genes expressing two more problematic sugars. In 2013, Muhammad Mohiuddin, then chief of the transplantation section at the NHLBI, further modified a group of triple-knockout pigs, adding genes that code for two human proteins: one that shields cells from attack by an immune mechanism known as the complement system; another that prevents harmful coagulation. (It was those pigs whose hearts recently broke survival records when transplanted into baboon bellies. Mohiuddin has since become director of xenoheart transplantation at the University of Maryland's new Center for Cardiac Xenotransplantation Research.) And in August 2017, researchers at Harvard Medical School, led by George Church and Luhan Yang, announced that they'd used CRISPR-cas9—an ultra-efficient new gene-editing technique—to disable 62 PERV genes in fetal pig cells, from which they then created cloned embryos. Of the 37 piglets born from this experiment, none showed any trace of the virus.
Still, the riddles surrounding animal-to-human transplants are far from fully solved. One open question is what further genetic manipulations will be necessary to eliminate all rejection. "No one is so naïve as to think, 'Oh, we know all the genes—let's put them in and we are done,'" biologist Sean Stevens, another leading researcher, told the The New York Times. "It's an iterative process, and no one that I know can say whether we will do two, or five, or 100 iterations." Adding traits can be dangerous as well; pigs engineered to express multiple anticoagulation proteins, for example, often die of bleeding disorders. "We're still finding out how many you can do, and what levels are acceptable," says Cooper.
Another question is whether PERV really needs to be disabled. Cooper and some of his colleagues note that pig tissue has long been used for various purposes, such as artificial heart valves and wound-repair products, without incident; requiring the virus to be eliminated, they argue, will unnecessarily slow progress toward creating viable xenotransplant organs and the animals that can provide them. Others disagree. "You cannot do anything with pig organs if you do not remove them," insists bioethicist Jeantine Lunshof, who works with Church and Yang at Harvard. "The risk is simply too big."
"We've removed the cells, so we don't have to worry about latent viruses."
Meanwhile, over the past decade, other approaches to xenotransplantation have emerged. One is interspecies blastocyst complementation, which could produce organs genetically identical to the recipient's tissues. In this method, genes that produce a particular organ are knocked out in the donor animal's embryo. The embryo is then injected with pluripotent stem cells made from the tissue of the intended recipient. The stem cells move in to fill the void, creating a functioning organ. This technique has been used to create mouse pancreases in rats, which were then successfully transplanted into mice. But the human-pig "chimeras" recently created by scientists were destroyed after 28 days, and no one plans to bring such an embryo to term anytime soon. "The problem is that cells don't stay put; they move around," explains Father Kevin FitzGerald, a bioethicist at Georgetown University. "If human cells wind up in a pig's brain, that leads to a really interesting conundrum. What if it's self-aware? Are you going to kill it?"
Much further along, and less ethically fraught, is a technique in which decellularized pig organs act as a scaffold for human cells. A Minnesota-based company called Miromatrix Medical is working with Mayo Clinic researchers to develop this method. First, a mild detergent is pumped through the organ, washing away all cellular material. The remaining structure, composed mainly of collagen, is placed in a bioreactor, where it's seeded with human cells. In theory, each type of cell that normally populates the organ will migrate to its proper place (a process that naturally occurs during fetal development, though it remains poorly understood). One potential advantage of this system is that it doesn't require genetically modified pigs; nor will the animals have to be raised under controlled conditions to avoid exposure to transmissible pathogens. Instead, the organs can be collected from ordinary slaughterhouses.

Recellularized livers in bioreactors
(Courtesy of Miromatrix)
"We've removed the cells, so we don't have to worry about latent viruses," explains CEO Jeff Ross, who describes his future product as a bioengineered human organ rather than a xeno-organ. That makes PERV a nonissue. To shorten the pathway to approval by the Food and Drug Administration, the replacement cells will initially come from human organs not suitable for transplant. But eventually, they'll be taken from the recipient (as in blastocyst complementation), which should eliminate the need for immunosuppression.
Clinical trials in xenotransplantation may begin as early as 2020.
Miromatrix plans to offer livers first, followed by kidneys, hearts, and eventually lungs and pancreases. The company recently succeeded in seeding several decellularized pig livers with human and porcine endothelial cells, which flocked obediently to the blood vessels. Transplanted into young pigs, the organs showed unimpaired circulation, with no sign of clotting. The next step is to feed all four liver cell types back into decellularized livers, and see if the transplanted organs will keep recipient pigs alive.
Ross hopes to launch clinical trials by 2020, and several other groups (including Cooper's, which plans to start with kidneys) envision a similar timeline. Investors seem to share their confidence. The biggest backer of xenotransplantation efforts is United Therapeutics, whose founder and co-CEO, Martine Rothblatt, has a daughter with a lung condition that may someday require a transplant; since 2011, the biotech firm has poured at least $100 million into companies pursuing such technologies, while supporting research by Cooper, Mohiuddin, and other leaders in the field. Church and Yang, at Harvard, have formed their own company, eGenesis, bringing in a reported $40 million in funding; Miromatrix has raised a comparable amount.
It's impossible to predict who will win the xenotransplantation race, or whether some new obstacle will stop the competition in its tracks. But Jennifer Cisneros is rooting for all the contestants. "These technologies could save my life," she says. If she hasn't found another kidney before trials begin, she has just one request: "Sign me up."
Bivalent Boosters for Young Children Are Elusive. The Search Is On for Ways to Improve Access.
Theo, an 18-month-old in rural Nebraska, walks with his father in their backyard. For many toddlers, the barriers to accessing COVID-19 vaccines are many, such as few locations giving vaccines to very young children.
It’s Theo’s* first time in the snow. Wide-eyed, he totters outside holding his father’s hand. Sarah Holmes feels great joy in watching her 18-month-old son experience the world, “His genuine wonder and excitement gives me so much hope.”
In the summer of 2021, two months after Theo was born, Holmes, a behavioral health provider in Nebraska lost her grandparents to COVID-19. Both were vaccinated and thought they could unmask without any risk. “My grandfather was a veteran, and really trusted the government and faith leaders saying that COVID-19 wasn’t a threat anymore,” she says.” The state of emergency in Louisiana had ended and that was the message from the people they respected. “That is what killed them.”
The current official public health messaging is that regardless of what variant is circulating, the best way to be protected is to get vaccinated. These warnings no longer mention masking, or any of the other Swiss-cheese layers of mitigation that were prevalent in the early days of this ongoing pandemic.
The problem with the prevailing, vaccine centered strategy is that if you are a parent with children under five, barriers to access are real. In many cases, meaningful tools and changes that would address these obstacles are lacking, such as offering vaccines at more locations, mandating masks at these sites, and providing paid leave time to get the shots.
Children are at risk
Data presented at the most recent FDA advisory panel on COVID-19 vaccines showed that in the last year infants under six months had the third highest rate of hospitalization. “From the beginning, the message has been that kids don’t get COVID, and then the message was, well kids get COVID, but it’s not serious,” says Elias Kass, a pediatrician in Seattle. “Then they waited so long on the initial vaccines that by the time kids could get vaccinated, the majority of them had been infected.”
A closer look at the data from the CDC also reveals that from January 2022 to January 2023 children aged 6 to 23 months were more likely to be hospitalized than all other vaccine eligible pediatric age groups.
“We sort of forced an entire generation of kids to be infected with a novel virus and just don't give a shit, like nobody cares about kids,” Kass says. In some cases, COVID has wreaked havoc with the immune systems of very young children at his practice, making them vulnerable to other illnesses, he said. “And now we have kids that have had COVID two or three times, and we don’t know what is going to happen to them.”
Jumping through hurdles
Children under five were the last group to have an emergency use authorization (EUA) granted for the COVID-19 vaccine, a year and a half after adult vaccine approval. In June 2022, 30,000 sites were initially available for children across the country. Six months later, when boosters became available, there were only 5,000.
Currently, only 3.8% of children under two have completed a primary series, according to the CDC. An even more abysmal 0.2% under two have gotten a booster.
Ariadne Labs, a health center affiliated with Harvard, is trying to understand why these gaps exist. In conjunction with Boston Children’s Hospital, they have created a vaccine equity planner that maps the locations of vaccine deserts based on factors such as social vulnerability indexes and transportation access.
“People are having to travel farther because the sites are just few and far between,” says Benjy Renton, a research assistant at Ariadne.
Michelle Baltes-Breitwisch, a pharmacist, and her two-year-old daughter, Charlee, live in Iowa. When the boosters first came out she expected her toddler could get it close to home, but her husband had to drive Charlee four hours roundtrip.
This experience hasn’t been uncommon, especially in rural parts of the U.S. If parents wanted vaccines for their young children shortly after approval, they faced the prospect of loading babies and toddlers, famous for their calm demeanor, into cars for lengthy rides. The situation continues today. Mrs. Smith*, a grant writer and non-profit advisor who lives in Idaho, is still unable to get her child the bivalent booster because a two-hour one-way drive in winter weather isn’t possible.
It can be more difficult for low wage earners to take time off, which poses challenges especially in a number of rural counties across the country, where weekend hours for getting the shots may be limited.
Protect Their Future (PTF), a grassroots organization focusing on advocacy for the health care of children, hears from parents several times a week who are having trouble finding vaccines. The vaccine rollout “has been a total mess,” says Tamara Lea Spira, co-founder of PTF “It’s been very hard for people to access vaccines for children, particularly those under three.”
Seventeen states have passed laws that give pharmacists authority to vaccinate as young as six months. Under federal law, the minimum age in other states is three. Even in the states that allow vaccination of toddlers, each pharmacy chain varies. Some require prescriptions.
It takes time to make phone calls to confirm availability and book appointments online. “So it means that the parents who are getting their children vaccinated are those who are even more motivated and with the time and the resources to understand whether and how their kids can get vaccinated,” says Tiffany Green, an associate professor in population health sciences at the University of Wisconsin at Madison.
Green adds, “And then we have the contraction of vaccine availability in terms of sites…who is most likely to be affected? It's the usual suspects, children of color, disabled children, low-income children.”
It can be more difficult for low wage earners to take time off, which poses challenges especially in a number of rural counties across the country, where weekend hours for getting the shots may be limited. In Bibb County, Ala., vaccinations take place only on Wednesdays from 1:45 to 3:00 pm.
“People who are focused on putting food on the table or stressed about having enough money to pay rent aren't going to prioritize getting vaccinated that day,” says Julia Raifman, assistant professor of health law, policy and management at Boston University. She created the COVID-19 U.S. State Policy Database, which tracks state health and economic policies related to the pandemic.
Most states in the U.S. lack paid sick leave policies, and the average paid sick days with private employers is about one week. Green says, “I think COVID should have been a wake-up call that this is necessary.”
Maskless waiting rooms
For her son, Holmes spent hours making phone calls but could uncover no clear answers. No one could estimate an arrival date for the booster. “It disappoints me greatly that the process for locating COVID-19 vaccinations for young children requires so much legwork in terms of time and resources,” she says.
In January, she found a pharmacy 30 minutes away that could vaccinate Theo. With her son being too young to mask, she waited in the car with him as long as possible to avoid a busy, maskless waiting room.
Kids under two, such as Theo, are advised not to wear masks, which make it too hard for them to breathe. With masking policies a rarity these days, waiting rooms for vaccines present another barrier to access. Even in healthcare settings, current CDC guidance only requires masking during high transmission or when treating COVID positive patients directly.
“This is a group that is really left behind,” says Raifman. “They cannot wear masks themselves. They really depend on others around them wearing masks. There's not even one train car they can go on if their parents need to take public transportation… and not risk COVID transmission.”
Yet another challenge is presented for those who don’t speak English or Spanish. According to Translators without Borders, 65 million people in America speak a language other than English. Most state departments of health have a COVID-19 web page that redirects to the federal vaccines.gov in English, with an option to translate to Spanish only.
The main avenue for accessing information on vaccines relies on an internet connection, but 22 percent of rural Americans lack broadband access. “People who lack digital access, or don’t speak English…or know how to navigate or work with computers are unable to use that service and then don’t have access to the vaccines because they just don’t know how to get to them,” Jirmanus, an affiliate of the FXB Center for Health and Human Rights at Harvard and a member of The People’s CDC explains. She sees this issue frequently when working with immigrant communities in Massachusetts. “You really have to meet people where they’re at, and that means physically where they’re at.”
Equitable solutions
Grassroots and advocacy organizations like PTF have been filling a lot of the holes left by spotty federal policy. “In many ways this collective care has been as important as our gains to access the vaccine itself,” says Spira, the PTF co-founder.
PTF facilitates peer-to-peer networks of parents that offer support to each other. At least one parent in the group has crowdsourced information on locations that are providing vaccines for the very young and created a spreadsheet displaying vaccine locations. “It is incredible to me still that this vacuum of information and support exists, and it took a totally grassroots and volunteer effort of parents and physicians to try and respond to this need.” says Spira.
Kass, who is also affiliated with PTF, has been vaccinating any child who comes to his independent practice, regardless of whether they’re one of his patients or have insurance. “I think putting everything on retail pharmacies is not appropriate. By the time the kids' vaccines were released, all of our mass vaccination sites had been taken down.” A big way to help parents and pediatricians would be to allow mixing and matching. Any child who has had the full Pfizer series has had to forgo a bivalent booster.
“I think getting those first two or three doses into kids should still be a priority, and I don’t want to lose sight of all that,” states Renton, the researcher at Ariadne Labs. Through the vaccine equity planner, he has been trying to see if there are places where mobile clinics can go to improve access. Renton continues to work with local and state planners to aid in vaccine planning. “I think any way we can make that process a lot easier…will go a long way into building vaccine confidence and getting people vaccinated,” Renton says.

Michelle Baltes-Breitwisch, a pharmacist, and her two-year-old daughter, Charlee, live in Iowa. Her husband had to drive four hours roundtrip to get the boosters for Charlee.
Michelle Baltes-Breitwisch
Other changes need to come from the CDC. Even though the CDC “has this historic reputation and a mission of valuing equity and promoting health,” Jirmanus says, “they’re really failing. The emphasis on personal responsibility is leaving a lot of people behind.” She believes another avenue for more equitable access is creating legislation for upgraded ventilation in indoor public spaces.
Given the gaps in state policies, federal leadership matters, Raifman says. With the FDA leaning toward a yearly COVID vaccine, an equity lens from the CDC will be even more critical. “We can have data driven approaches to using evidence based policies like mask policies, when and where they're most important,” she says. Raifman wants to see a sustainable system of vaccine delivery across the country complemented with a surge preparedness plan.
With the public health emergency ending and vaccines going to the private market sometime in 2023, it seems unlikely that vaccine access is going to improve. Now more than ever, ”We need to be able to extend to people the choice of not being infected with COVID,” Jirmanus says.
*Some names were changed for privacy reasons.
Last month, a paper published in Cell by Harvard biologist David Sinclair explored root cause of aging, as well as examining whether this process can be controlled. We talked with Dr. Sinclair about this new research.
What causes aging? In a paper published last month, Dr. David Sinclair, Professor in the Department of Genetics at Harvard Medical School, reports that he and his co-authors have found the answer. Harnessing this knowledge, Dr. Sinclair was able to reverse this process, making mice younger, according to the study published in the journal Cell.
I talked with Dr. Sinclair about his new study for the latest episode of Making Sense of Science. Turning back the clock on mouse age through what’s called epigenetic reprogramming – and understanding why animals get older in the first place – are key steps toward finding therapies for healthier aging in humans. We also talked about questions that have been raised about the research.
Show links:
Dr. Sinclair's paper, published last month in Cell.
Recent pre-print paper - not yet peer reviewed - showing that mice treated with Yamanaka factors lived longer than the control group.
Dr. Sinclair's podcast.
Previous research on aging and DNA mutations.
Dr. Sinclair's book, Lifespan.


Harvard Medical School

