Can You Trust Your Gut for Food Advice?
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.

Which foods are actually healthy for your individual gut microbiome? Several companies are offering personalized dietary guidance based on your test results, but their answers in one experiment turned up with some conflicting advice.
I recently got on the scale to weigh myself, thinking I've got to eat better. With so many trendy diets today claiming to improve health, from Keto to Paleo to Whole30, it can be confusing to figure out what we should and shouldn't eat for optimal nutrition.
A number of companies are now selling the concept of "personalized" nutrition based on the genetic makeup of your individual gut bugs.
My next thought was: I've got to lose a few pounds.
Consider a weird factoid: In addition to my fat, skin, bone and muscle, I'm carrying around two or three pounds of straight-up bacteria. Like you, I am the host to trillions of micro-organisms that live in my gut and are collectively known as my microbiome. An explosion of research has occurred in the last decade to try to understand exactly how these microbial populations, which are unique to each of us, may influence our overall health and potentially even our brains and behavior.
Lots of mysteries still remain, but it is established that these "bugs" are crucial to keeping our body running smoothly, performing functions like stimulating the immune system, synthesizing important vitamins, and aiding digestion. The field of microbiome science is evolving rapidly, and a number of companies are now selling the concept of "personalized" nutrition based on the genetic makeup of your individual gut bugs. The two leading players are Viome and DayTwo, but the landscape includes the newly launched startup Onegevity Health and others like Thryve, which offers customized probiotic supplements in addition to dietary recommendations.
The idea has immediate appeal – if science could tell you exactly what to make for lunch and what to avoid, you could forget about the fad diets and go with your own bespoke food pyramid. Wondering if the promise might be too good to be true, I decided to perform my own experiment.
Last fall, I sent the identical fecal sample to both Viome (I paid $425, but the price has since dropped to $299) and DayTwo ($349). A couple of months later, both reports finally arrived, and I eagerly opened each app to compare their recommendations.
First, I examined my results from Viome, which was founded in 2016 in Cupertino, Calif., and declares without irony on its website that "conflicting food advice is now obsolete."
I learned I have "average" metabolic fitness and "average" inflammatory activity in my gut, which are scores that the company defines based on a proprietary algorithm. But I have "low" microbial richness, with only 62 active species of bacteria identified in my sample, compared with the mean of 157 in their test population. I also received a list of the specific species in my gut, with names like Lactococcus and Romboutsia.
But none of it meant anything to me without actionable food advice, so I clicked through to the Recommendations page and found a list of My Superfoods (cranberry, garlic, kale, salmon, turmeric, watermelon, and bone broth) and My Foods to Avoid (chickpeas, kombucha, lentils, and rice noodles). There was also a searchable database of many foods that had been categorized for me, like "bell pepper; minimize" and "beef; enjoy."
"I just don't think sufficient data is yet available to make reliable personalized dietary recommendations based on one's microbiome."
Next, I looked at my results from DayTwo, which was founded in 2015 from research out of the Weizmann Institute of Science in Israel, and whose pitch to consumers is, "Blood sugar made easy. The algorithm diet personalized to you."
This app had some notable differences. There was no result about my metabolic fitness, microbial richness, or list of the species in my sample. There was also no list of superfoods or foods to avoid. Instead, the app encouraged me to build a meal by searching for foods in their database and combining them in beneficial ways for my blood sugar. Two slices of whole wheat bread received a score of 2.7 out of 10 ("Avoid"), but if combined with one cup of large curd cottage cheese, the score improved to 6.8 ("Limit"), and if I added two hard-boiled eggs, the score went up to 7.5 ("Good").
Perusing my list of foods with "Excellent" scores, I noticed some troubling conflicts with the other app. Lentils, which had been a no-no according to Viome, received high marks from DayTwo. Ditto for Kombucha. My purported superfood of cranberry received low marks. Almonds got an almost perfect score (9.7) while Viome told me to minimize them. I found similarly contradictory advice for foods I regularly eat, including navel oranges, peanuts, pork, and beets.
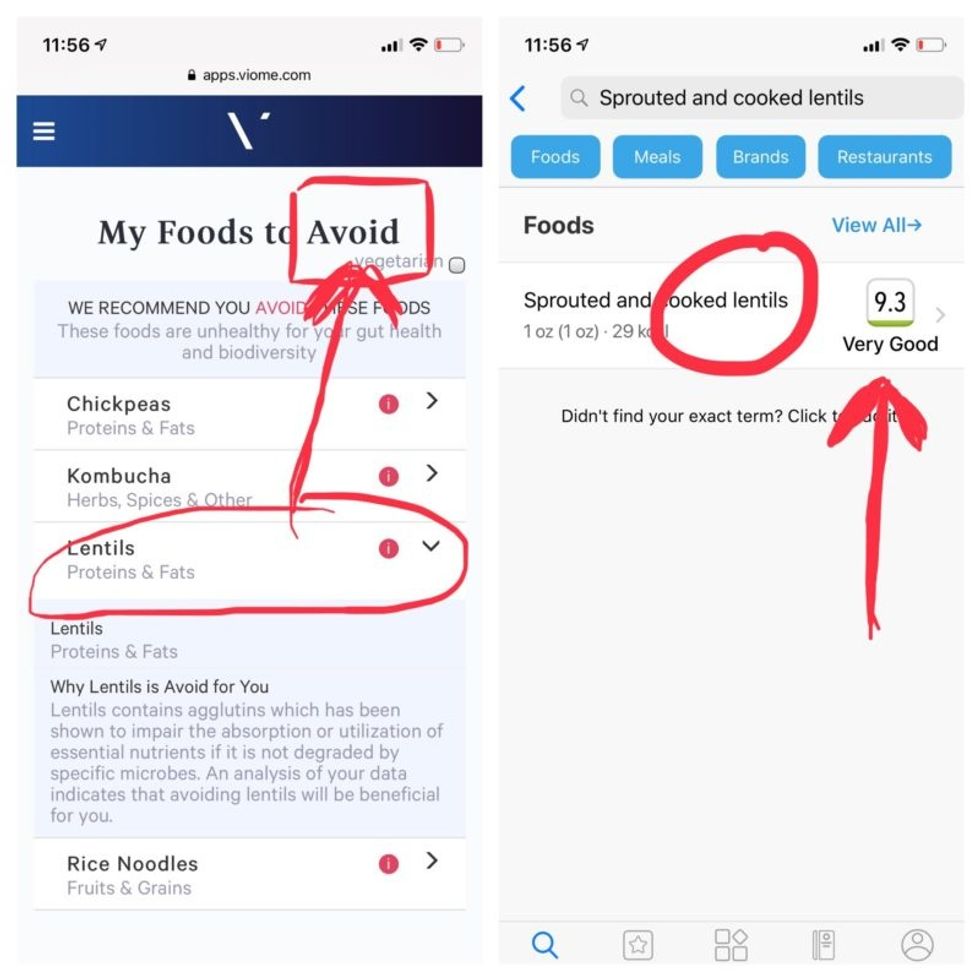
Contradictory dietary guidance that Kira Peikoff received from Viome (left) and DayTwo from an identical sample.
To be sure, there was some overlap. Both apps agreed on rice noodles (bad), chickpeas (bad), honey (bad), carrots (good), and avocado (good), among other foods.
But still, I was left scratching my head. Which set of recommendations should I trust, if either? And what did my results mean for the accuracy of this nascent field?
I called a couple of experts to find out.
"I have worked on the microbiome and nutrition for the last 20 years and I would be absolutely incapable of finding you evidence in the scientific literature that lentils have a detrimental effect based on the microbiome," said Dr. Jens Walter, an Associate Professor and chair for Nutrition, Microbes, and Gastrointestinal Health at the University of Alberta. "I just don't think sufficient data is yet available to make reliable personalized dietary recommendations based on one's microbiome. And even if they would have proprietary algorithms, at least one of them is not doing it right."
There is definite potential for personalized nutrition based on the microbiome, he said, but first, predictive models must be built and standardized, then linked to clinical endpoints, and tested in a large sample of healthy volunteers in order to enable extrapolations for the general population.
"It is mindboggling what you would need to do to make this work," he observed. "There are probably hundreds of relevant dietary compounds, then the microbiome has at least a hundred relevant species with a hundred or more relevant genes each, then you'd have to put all this together with relevant clinical outcomes. And there's a hundred-fold variation in that information between individuals."
However, Walter did acknowledge that the companies might be basing their algorithms on proprietary data that could potentially connect all the dots. I reached out to them to find out.
Amir Golan, the Chief Commercial Officer of DayTwo, told me, "It's important to emphasize this is a prediction, as the microbiome field is in a very early stage of research." But he added, "I believe we are the only company that has very solid science published in top journals and we can bring very actionable evidence and benefit to our uses."
He was referring to pioneering work out of the Weizmann Institute that was published in 2015 in the journal Cell, which logged the glycemic responses of 800 people in response to nearly 50,000 meals; adding information about the subjects' microbiomes enabled more accurate glycemic response predictions. Since then, Golan said, additional trials have been conducted, most recently with the Mayo Clinic, to duplicate the results, and other studies are ongoing whose results have not yet been published.
He also pointed out that the microbiome was merely one component that goes into building a client's profile, in addition to medical records, including blood glucose levels. (I provided my HbA1c levels, a measure of average blood sugar over the previous several months.)
"We are not saying we want to improve your gut microbiome. We provide a dynamic tool to help guide what you should eat to control your blood sugar and think about combinations," he said. "If you eat one thing, or with another, it will affect you in a different way."
Viome acknowledged that the two companies are taking very different approaches.
"DayTwo is primarily focused on the glycemic response," Naveen Jain, the CEO, told me. "If you can only eat butter for rest of your life, you will have no glycemic response but will probably die of a heart attack." He laughed. "Whereas we came from very different angle – what is happening inside the gut at a microbial level? When you eat food like spinach, how will that be metabolized in the gut? Will it produce the nutrients you need or cause inflammation?"
He said his team studied 1000 people who were on continuous glucose monitoring and fed them 45,000 meals, then built a proprietary data prediction model, looking at which microbes existed and how they actively broke down the food.
Jain pointed out that DayTwo sequences the DNA of the microbes, while Viome sequences the RNA – the active expression of DNA. That difference, in his opinion, is key to making accurate predictions.
"DNA is extremely stable, so when you eat any food and measure the DNA [in a fecal sample], you get all these false positives--you get DNA from plant food and meat, and you have no idea if those organisms are dead and simply transient, or actually exist. With RNA, you see what is actually alive in the gut."
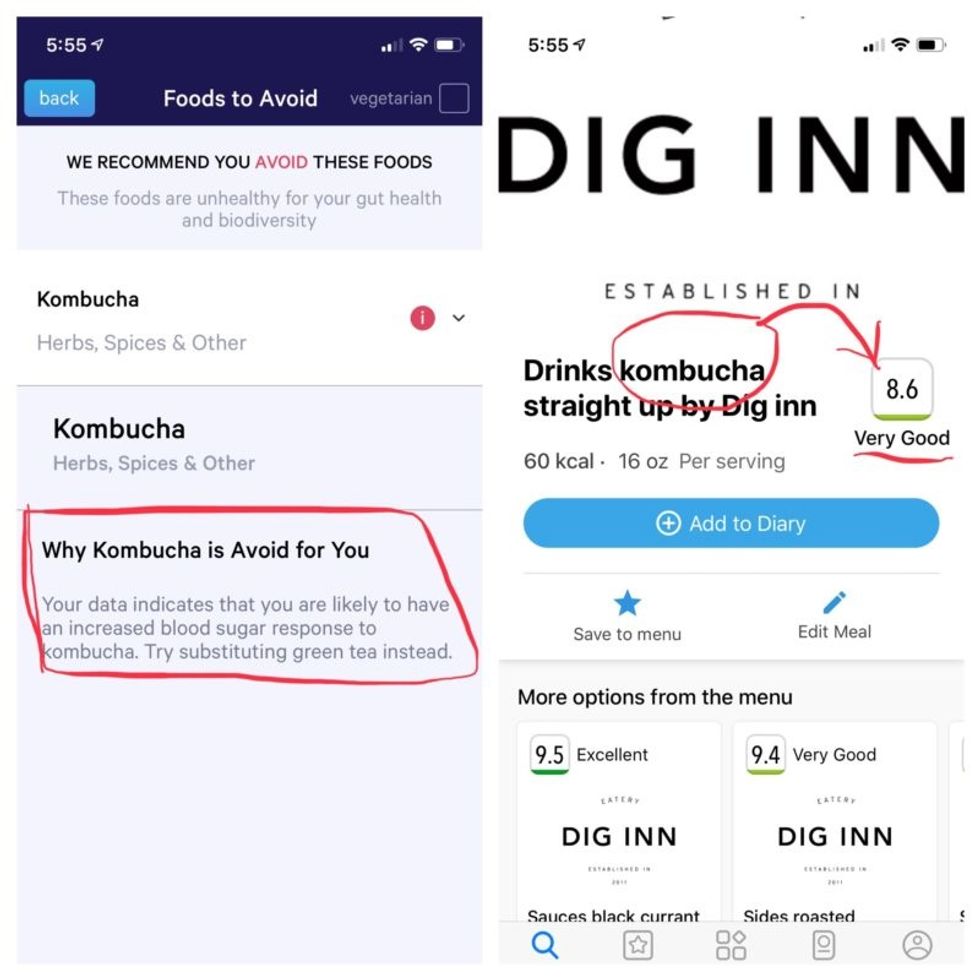
More contradictory food advice from Viome (left) and DayTwo.
Note that controversy exists over how it is possible with a fecal sample to effectively measure RNA, which degrades within minutes, though Jain said that his company has the technology to keep RNA stable for fourteen days.
Viome's approach, Jain maintains, is 90 percent accurate, based on as-yet unpublished data; a patent was filed just last week. DayTwo's approach is 66 percent accurate according to the latest published research.
Natasha Haskey, a registered dietician and doctoral student conducting research in the field of microbiome science and nutrition, is skeptical of both companies. "We can make broad statements, like eat more fruits and vegetables and fiber, but when it comes to specific foods, the science is just not there yet," she said. "I think there is a future, and we will be doing that someday, but not yet. Maybe we will be closer in ten years."
Professor Walter wholeheartedly agrees with Haskey, and suggested that if people want to eat a gut-healthy diet, they should focus on beneficial oils, fruits and vegetables, fish, a variety of whole grains, poultry and beans, and limit red meat and cheese, as well as avoid processed meats.
"These services are far over the tips of their science skis," Arthur Caplan, the founding head of New York University's Division of Medical Ethics, said in an email. "We simply don't know enough about the gut microbiome, its fluctuations and variability from person to person to support general [direct-to-consumer] testing. This is simply premature. We need standards for accuracy, specificity, and sensitivity, plus mandatory competent counseling for all such testing. They don't exist. Neither should DTC testing—yet."
Meanwhile, it's time for lunch. I close out my Viome and DayTwo apps and head to the kitchen to prepare a peanut butter sandwich. My gut tells me I'll be just fine.
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.
They received retinal implants to restore their vision. Then the company turned its back on them.
A company called Second Sight made an implant that partially restored vision to people who'd been blind for decades. But when Second Sight pivoted, it stopped servicing its product, leaving many in the dark.
The first thing Jeroen Perk saw after he partially regained his sight nearly a decade ago was the outline of his guide dog Pedro.
“There was a white floor, and the dog was black,” recalls Perk, a 43-year-old investigator for the Dutch customs service. “I was crying. It was a very nice moment.”
Perk was diagnosed with retinitis pigmentosa as a child and had been blind since early adulthood. He has been able to use the implant placed into his retina in 2013 to help identify street crossings, and even ski and pursue archery. A video posted by the company that designed and manufactured the device indicates he’s a good shot.
Less black-and-white has been the journey Perk and others have been on after they were implanted with the Argus II, a second-generation device created by a Los Angeles-based company called Second Sight Medical Devices.
The Argus II uses the implant and a video camera embedded in a special pair of glasses to provide limited vision to those with retinitis pigmentosa, a genetic disease that causes cells in the retina to deteriorate. The camera feeds information to the implant, which sends electrical impulses into the retina to recapitulate what the camera sees. The impulses appear in the Argus II as a 60-pixel grid of blacks, grays and whites in the user’s eye that can render rough outlines of objects and their motion.
Smartphone and computer manufacturers typically stop issuing software upgrades to their devices after two or three years, eventually rendering them bricks. But is the smartphone approach acceptable for a device that helps restore the most crucial sense a human being possesses?
Ross Doerr, a retired disability rights attorney in Maine who received an Argus II in 2019, describes the field of vision as the equivalent of an index card held at arm’s length. Perk often brings objects close to his face to decipher them. Moreover, users must swivel their heads to take in visual data; moving their eyeballs does not work.
Despite its limitations, the Argus II beats the alternative. Perk no longer relies on his guide dog. Doerr was uplifted when he was able to see the outlines of Christmas trees at a holiday show.
“The fairy godmother department sort of reaches out and taps you on the shoulder once in a while,” Doerr says of his implant, which came about purely by chance. A surgeon treating his cataracts was partnered with the son of another surgeon who was implanting the devices, and he was referred.
Doerr had no reason to believe the shower of fairy dust wouldn’t continue. Second Sight held out promises that the Argus II recipients’ vision would gradually improve through upgrades to much higher pixel densities. The ability to recognize individual faces was even touted as a possibility. In the winter of 2020, Doerr was preparing to travel across the U.S. to Second Sight’s headquarters to receive an upgrade. But then COVID-19 descended, and the trip was canceled.
The pandemic also hit Second Sight’s bottom line. Doerr found out about its tribulations only from one of the company’s vision therapists, who told him the entire department was being laid off. Second Sight cut nearly 80% of its workforce in March 2020 and announced it would wind down operations.

Ross Doerr has mostly stopped using his Argus II, the result of combination of fear of losing its assistance from wear and tear and disdain for the company that brought it to market.
Jan Doerr
Second Sight’s implosion left some 350 Argus recipients in the metaphorical dark about what to do if their implants failed. Skeleton staff seem to have rarely responded to queries from their customers, at least based on the experiences of Perk and Doerr. And some recipients have unfortunately returned to the actual dark as well, as reports have surfaced of Argus II failures due to aging or worn-down parts.
Product support for complex products is remarkably uneven. Although the iconic Ford Mustang ceased production in the late 1960s, its parts market is so robust that it’s theoretically possible to assemble a new vehicle from recently crafted components. Conversely, smartphone and computer manufacturers typically stop issuing software upgrades to their devices after two or three years, eventually rendering them bricks. Consumers have accepted both extremes.
But is the smartphone approach acceptable for a device that helps restore the most crucial sense a human being possesses?
Margaret McLean, a senior fellow at the Markkula Center for Applied Ethics at Santa Clara University in California, notes companies like Second Sight have a greater obligation for product support than other consumer product ventures.
“In this particular case, you have a great deal of risk that is involved in using this device, the implant, and the after care of this device,” she says. “You cannot, like with your car, decide that ‘I don’t like my Mustang anymore,’ and go out and buy a Corvette.”
And, whether the Argus II implant works or not, its physical presence can impact critical medical decisions. Doerr’s doctor wanted him to undergo an MRI to assist in diagnosing attacks of vertigo. But the physician was concerned his implant might interfere. With the latest available manufacturer advisories on his implant nearly a decade old, the procedure was held up. Doerr spent months importuning Second Sight through phone calls, emails and Facebook postings to learn if his implant was contraindicated with MRIs, which he never received. Although the cause of his vertigo was found without an MRI, Doerr was hardly assured.
“Put that into context for a minute. I get into a serious car accident. I end up in the emergency room, and I have a tag saying I have an implanted medical device,” he says. “You can’t do an MRI until you get the proper information from the company. Who’s going to answer the phone?”
Second Sight’s management did answer the call to revamp its business. It netted nearly $78 million through a private stock placement and an initial public offering last year. At the end of 2021, Second Sight had nearly $70 million in cash on hand, according to a recent filing with the Securities and Exchange Commission.
And while the Argus II is still touted at length on Second Sight’s home page, it appears little of its corporate coffers are earmarked toward its support. These days, the company is focused on obtaining federal approvals for Orion, a new implant that would go directly into the recipient’s brain and could be used to remedy blindness from a variety of causes. It obtained a $6.4 million grant from the National Institutes of Health in May 2021 to help develop Orion.
Presented with a list of written questions by email, Second Sight’s spokesperson, Dave Gentry of the investor relations firm Red Chip Companies, copied a subordinate with an abrupt message to “please handle.” That was the only response from a company representative. A call to Second Sight acting chief executive officer Scott Dunbar went unreturned.
Whether or not the Orion succeeds remains to be seen. The company’s SEC filings suggest a viable and FDA-approved device is years away, and that operational losses are expected for the “foreseeable future.” Second Sight reported zero revenue in 2020 or 2021.
Moreover, the experiences of the Argus II recipients could color the reception of future Second Sight products. Doerr notes that his insurer paid nearly $500,000 to implant his device and for training on how to use it.
“What’s the insurance industry going to say the next time this crops up?” Doerr asks, noting that the company’s reputation is “completely shot” with the recipients of its implants.
Perk, who made speeches to praise the Argus II and is still featured in a video on the Second Sight website, says he also no longer supports the company.

Jeroen Perk, an investigator for the Dutch customs service, cried for joy after partially regaining his sight, but he no longer trusts Second Sight, the company that provided his implant.
Nanda Perk
Nevertheless, Perk remains highly reliant on the technology. When he dropped an external component of his device in late 2020 and it broke, Perk briefly debated whether to remain blind or find a way to get his Argus II working again. Three months later, he was able to revive it by crowdsourcing parts, primarily from surgeons with spare components or other Argus II recipients who no longer use their devices. Perk now has several spare parts in reserve in case of future breakdowns.
Despite the frantic efforts to retain what little sight he has, Perk has no regrets about having the device implanted. And while he no longer trusts Second Sight, he is looking forward to possibly obtaining more advanced implants from companies in the Netherlands and Australia working on their own products.
Doerr suggests that biotech firms whose implants are distributed globally be bound to some sort of international treaty requiring them to service their products in perpetuity. Such treaties are still applied to the salvage rights for ships that sunk centuries ago, he notes.
“I think that in a global tech economy, that would be a good thing,” says McLean, the fellow at Santa Clara, “but I am not optimistic about it in the near term. Business incentives push toward return on share to stockholders, not to patients and other stakeholders. We likely need to rely on some combination of corporately responsibility…and [international] government regulation. It’s tough—the Paris Climate Accord implementation at a slow walk comes to mind.”
Unlike Perk, Doerr has mostly stopped using his Argus II, the result of combination of fear of losing its assistance from wear and tear and disdain for the company that brought it to market. At 70, Doerr says he does not have the time or energy to hold the company more accountable. And with Second Sight having gone through a considerable corporate reorganization, Doerr believes a lawsuit to compel it to better serve its Argus recipients would be nothing but an extremely costly longshot.
“It’s corporate America at its best,” he observes.
Rehabilitating psychedelic drugs: Another key to treating severe mental health disorders
A recent review paper found evidence that using psychedelics such as MDMA can help with treating a variety of common mental illnesses, but experts fear that research might easily be shut down in the future.
Lori Tipton's life was a cascade of trauma that even a soap opera would not dare inflict upon a character: a mentally unstable family; a brother who died of a drug overdose; the shocking discovery of the bodies of two persons her mother had killed before turning the gun on herself; the devastation of Hurricane Katrina that savaged her hometown of New Orleans; being raped by someone she trusted; and having an abortion. She suffered from severe PTSD.
“My life was filled with anxiety and hypervigilance,” she says. “I was constantly afraid and had mood swings, panic attacks, insomnia, intrusive thoughts and suicidal ideation. I tried to take my life more than once.” She was fortunate to be able to access multiple mental health services, “And while at times some of these modalities would relieve the symptoms, nothing really lasted and nothing really address the core trauma.”
Then in 2018 Tipton enrolled in a clinical trial that combined intense sessions of psychotherapy with limited use of Methylenedioxymethamphetamine, or MDMA, a drug classified as a psychedelic and commonly known as ecstasy or Molly. The regimen was arduous; 1-2 hour preparation sessions, three sessions where MDMA was used, which lasted 6-8 hours, and lengthy sessions afterward to process and integrate the experiences. Two therapists were with her every moment of the three-month program that totaled more than 40 hours.
“It was clear to me that [the therapists] weren't going to heal me, that I was going to have to do the work for myself, but that they were there to completely support my process,” she says. “But the effects of MDMA were really undeniable for me. I felt embodied in a way that I hadn't in years. PTSD had robbed me of the ability to feel safe in my own body.”
Tipton doesn’t think the therapy completely cured her PTSD. “But when I completed the trial in 2018, I no longer qualified for the diagnosis, and I still don't qualify for the diagnosis today,” she told an April workshop on psychedelics as mental health treatment by the National Academies of Sciences, Engineering and Medicine, or NASEM.
A Champion
Rick Doblin has been a catalyst behind much of the contemporary research into psychedelics. Prior to the DEA clamp down, the Boston psychotherapist had seen that MDMA and other psychedelics could benefit some of his patients where other measures had failed. He immediately organized efforts to question the drug rescheduling but to little avail. In 1986, he created the nonprofit Multidisciplinary Association for Psychedelic Studies (MAPS), which slowly laid the scientific foundation for clinical trials, including the one that Tipton joined, using psychedelics to treat mental health conditions.
Now, only slowly, have researchers been able to explore the power of these drugs to treat a broad spectrum of severely debilitating mental health conditions, including trauma, depression, and PTSD, where other available treatments proved inadequate.
“Psychedelic psychotherapy is an attempt to go after the root causes of the problems with just a relatively few administrations, as contrasted to most of the psychiatric drugs used today that are mostly just reducing symptoms and are meant to be taken on a daily basis,” Doblin said in a 2019 TED Talk. Most of these drugs can have broad effect but “some are probably more effective than others for certain conditions,” he added in a recent interview with Leaps.org. Comparative head-to-head studies of psychedelic therapies simply have not been conducted.
Their mechanisms of action are poorly understood and can vary between drugs, but it is generally believed that psychedelics change the activity of neurons so that the brain processes information differently, says Katrin Preller, a neuropsychologist at the University of Zurich. A recent important study in Nature Medicine by Richard Daws and colleagues used functional magnetic resonance imaging (fMRI) of the brain and found that “functional networks became more functionally interconnected and flexible after psilocybin treatment…implying that psilocybin's antidepressant action may depend on a global increase in brain network integration.”
Rosalind Watts, a clinical investigator at the Imperial College in London, believes there is “an overestimation of the importance of the drug and an underestimation of the importance of the [therapeutic] context” in psychedelic research. “It is unethical to provide the drug without the other,” she says. Doblin notes that “psychotherapy outcomes research demonstrates that the therapeutic alliance between the therapist and the patients is the single most predictive factor of outcomes. [It is] trust and the sense of safety, the willingness to go into difficult spaces” that makes clinical breakthroughs possible with the drug.
Excitement and Challenges
Recurrent themes expressed at the NASEM workshop were exciting glimpses of the potential for psychedelics to treat mental health conditions combined with the challenges of realizing those potentials. A recent review paper found evidence that using psychedelics can help with treating a variety of common mental illnesses, but the paper could identify only 14 clinical trials of classic psychedelics published since 1991. Much of the reason is that the drugs are not patentable and so the pharmaceutical industry has no interest in investing in expensive clinical trials to bring them to market. MAPS has raised about $135 million over its 36-year history to conduct such research, says Doblin, the vast majority of it from individual donors and none from foundations.
The workshop participants’ views also were colored by the history of drug crackdowns and a fear that research might easily be shut down in the future. There was great concern that use of psychedelics should be confined to clinical trials with high safety and ethical standards, instead of doctors and patients experimenting on their own. “We need to get it right this time,” says Charles Grob, a psychiatrist at the UCLA School of Medicine. But restricting access to psychedelics will become even more difficult now that Oregon and several cities have acted to decriminalize possession and use of many of these drugs.
The experience with ketamine also troubled Grob. He is hoping to “mitigate the rush of rapid commercialization” that occurred with that drug. Ketamine technically is not a psychedelic though it does share some of their potentially euphoric properties. In 2019, soon after the FDA approved a form of ketamine with a limited label indication to treat depression, for profit clinics sprang up promoting off label use of the drug for psychiatric conditions where there was little clinical evidence of efficacy. He fears the same thing will happen when true psychedelics are made available.
If these therapies are approved, access to them is likely to be a problem. The drugs themselves are cheap but the accompanying therapy is not, and there is a shortage of trained psychotherapists. Mental health services often are not adequately covered by health insurance, while the poor and people of color suffer additional burdens of inadequate access. Doblin is committed to health care equity by training additional providers and by investigating whether some of the preparatory and integration sessions might be handled in a group setting. He says it is important that the legal aspects of psychedelics also be addressed so that patients “don't have to go underground” in order to receive this care.

