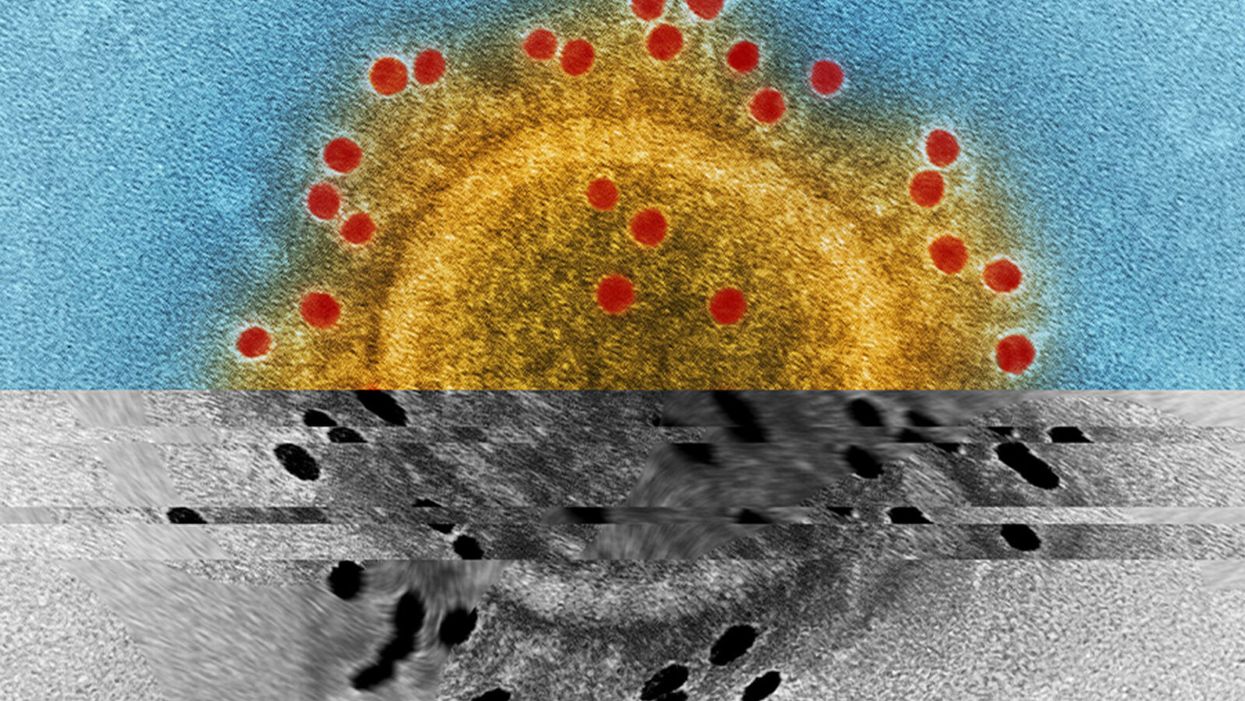
Study Shows “Living Drug” Can Provide a Lasting Cure for Cancer

A recent study by researchers at the University of Pennsylvania examined how CAR-T therapy helped Doug Olson beat a cancer death sentence for over a decade - and how it could work for more people.
Doug Olson was 49 when he was diagnosed with chronic lymphocytic leukemia, a blood cancer that strikes 21,000 Americans annually. Although the disease kills most patients within a decade, Olson’s case progressed more slowly, and courses of mild chemotherapy kept him healthy for 13 years. Then, when he was 62, the medication stopped working. The cancer had mutated, his doctor explained, becoming resistant to standard remedies. Harsher forms of chemo might buy him a few months, but their side effects would be debilitating. It was time to consider the treatment of last resort: a bone-marrow transplant.
Olson, a scientist who developed blood-testing instruments, knew the odds. There was only a 50 percent chance that a transplant would cure him. There was a 20 percent chance that the agonizing procedure—which involves destroying the patient’s marrow with chemo and radiation, then infusing his blood with donated stem cells—would kill him. If he survived, he would face the danger of graft-versus-host disease, in which the donor’s cells attack the recipient’s tissues. To prevent it, he would have to take immunosuppressant drugs, increasing the risk of infections. He could end up with pneumonia if one of his three grandchildren caught a sniffle. “I was being pushed into a corner,” Olson recalls, “with very little room to move.”
Soon afterward, however, his doctor revealed a possible escape route. He and some colleagues at the University of Pennsylvania’s Abramson Cancer Center were starting a clinical trial, he said, and Olson—still mostly symptom-free—might be a good candidate. The experimental treatment, known as CAR-T therapy, would use genetic engineering to turn his T lymphocytes (immune cells that guard against viruses and other pathogens) into a weapon against cancer.
In September 2010, technicians took some of Olson’s T cells to a laboratory, where they were programmed with new molecular marching orders and coaxed to multiply into an army of millions. When they were ready, a nurse inserted a catheter into his neck. At the turn of a valve, his soldiers returned home, ready to do battle.
“I felt like I’d won the lottery,” Olson says. But he was only the second person in the world to receive this “living drug,” as the University of Pennsylvania investigators called it. No one knew how long his remission would last.
Three weeks later, Olson was slammed with a 102-degree fever, nausea, and chills. The treatment had triggered two dangerous complications: cytokine release syndrome, in which immune chemicals inflame the patient’s tissues, and tumor lysis syndrome, in which toxins from dying cancer cells overwhelm the kidneys. But the crisis passed quickly, and the CAR-T cells fought on. A month after the infusion, the doctor delivered astounding news: “We can’t find any cancer in your body.”
“I felt like I’d won the lottery,” Olson says. But he was only the second person in the world to receive this “living drug,” as the University of Pennsylvania investigators called it. No one knew how long his remission would last.
An Unexpected Cure
In February 2022, the same cancer researchers reported a remarkable milestone: the trial’s first two patients had survived for more than a decade. Although Olson’s predecessor—a retired corrections officer named Bill Ludwig—died of COVID-19 complications in early 2021, both men had remained cancer-free. And the modified immune cells continued to patrol their territory, ready to kill suspected tumor cells the moment they arose.
“We can now conclude that CAR-T cells can actually cure patients with leukemia,” University of Pennsylvania immunologist Carl June, who spearheaded the development of the technique, told reporters. “We thought the cells would be gone in a month or two. The fact that they’ve survived 10 years is a major surprise.”
Even before the announcement, it was clear that CAR-T therapy could win a lasting reprieve for many patients with cancers that were once a death sentence. Since the Food and Drug Administration approved June’s version (marketed as Kymriah) in 2017, the agency has greenlighted five more such treatments for various types of leukemia, lymphoma, and myeloma. “Every single day, I take care of patients who would previously have been told they had no options,” says Rayne Rouce, a pediatric hematologist/oncologist at Texas Children’s Cancer Center. “Now we not only have a treatment option for those patients, but one that could potentially be the last therapy for their cancer that they’ll ever have to receive.”

Immunologist Carl June, middle, spearheaded development of the CAR-T therapy that gave patients Bill Ludwig, left, and Doug Olson, right, a lengthy reprieve on their terminal cancer diagnoses.
Penn Medicine
Yet the CAR-T approach doesn’t help everyone. So far, it has only shown success for blood cancers—and for those, the overall remission rate is 30 to 40 percent. “When it works, it works extraordinarily well,” says Olson’s former doctor, David Porter, director of Penn’s blood and bone marrow transplant program. “It’s important to know why it works, but it’s equally important to know why it doesn’t—and how we can fix that.”
The team’s study, published in the journal Nature, offers a wealth of data on what worked for these two patients. It may also hold clues for how to make the therapy effective for more people.
Building a Better T Cell
Carl June didn’t set out to cure cancer, but his serendipitous career path—and a personal tragedy—helped him achieve insights that had eluded other researchers. In 1971, hoping to avoid combat in Vietnam, he applied to the U.S. Naval Academy in Annapolis, Maryland. June showed a knack for biology, so the Navy sent him on to Baylor College of Medicine. He fell in love with immunology during a fellowship researching malaria vaccines in Switzerland. Later, the Navy deployed him to the Fred Hutchinson Cancer Research Center in Seattle to study bone marrow transplantation.
There, June became part of the first research team to learn how to culture T cells efficiently in a lab. After moving on to the National Naval Medical Center in the ’80s, he used that knowledge to combat the newly emerging AIDS epidemic. HIV, the virus that causes the disease, invades T cells and eventually destroys them. June and his post-doc Bruce Levine developed a method to restore patients’ depleted cell populations, using tiny magnetic beads to deliver growth-stimulating proteins. Infused into the body, the new T cells effectively boosted immune function.
In 1999, after leaving the Navy, June joined the University of Pennsylvania. His wife, who’d been diagnosed with ovarian cancer, died two years later, leaving three young children. “I had not known what it was like to be on the other side of the bed,” he recalls. Watching her suffer through grueling but futile chemotherapy, followed by an unsuccessful bone-marrow transplant, he resolved to focus on finding better cancer treatments. He started with leukemia—a family of diseases in which mutant white blood cells proliferate in the marrow.
Cancer is highly skilled at slipping through the immune system’s defenses. T cells, for example, detect pathogens by latching onto them with receptors designed to recognize foreign proteins. Leukemia cells evade detection, in part, by masquerading as normal white blood cells—that is, as part of the immune system itself.
June planned to use a viral vector no one had tried before: HIV.
To June, chimeric antigen receptor (CAR) T cells looked like a promising tool for unmasking and destroying the impostors. Developed in the early ’90s, these cells could be programmed to identify a target protein, and to kill any pathogen that displayed it. To do the programming, you spliced together snippets of DNA and inserted them into a disabled virus. Next, you removed some of the patient’s T cells and infected them with the virus, which genetically hijacked its new hosts—instructing them to find and slay the patient’s particular type of cancer cells. When the T cells multiplied, their descendants carried the new genetic code. You then infused those modified cells into the patient, where they went to war against their designated enemy.
Or that’s what happened in theory. Many scientists had tried to develop therapies using CAR-T cells, but none had succeeded. Although the technique worked in lab animals, the cells either died out or lost their potency in humans.
But June had the advantage of his years nurturing T cells for AIDS patients, as well as the technology he’d developed with Levine (who’d followed him to Penn with other team members). He also planned to use a viral vector no one had tried before: HIV, which had evolved to thrive in human T cells and could be altered to avoid causing disease. By the summer of 2010, he was ready to test CAR-T therapy against chronic lymphocytic leukemia (CLL), the most common form of the disease in adults.
Three patients signed up for the trial, including Doug Olson and Bill Ludwig. A portion of each man’s T cells were reprogrammed to detect a protein found only on B lymphocytes, the type of white blood cells affected by CLL. Their genetic instructions ordered them to destroy any cell carrying the protein, known as CD19, and to multiply whenever they encountered one. This meant the patients would forfeit all their B cells, not just cancerous ones—but regular injections of gamma globulins (a cocktail of antibodies) would make up for the loss.
After being infused with the CAR-T cells, all three men suffered high fevers and potentially life-threatening inflammation, but all pulled through without lasting damage. The third patient experienced a partial remission and survived for eight months. Olson and Ludwig were cured.
Learning What Works
Since those first infusions, researchers have developed reliable ways to prevent or treat the side effects of CAR-T therapy, greatly reducing its risks. They’ve also been experimenting with combination therapies—pairing CAR-T with chemo, cancer vaccines, and immunotherapy drugs called checkpoint inhibitors—to improve its success rate. But CAR-T cells are still ineffective for at least 60 percent of blood cancer patients. And they remain in the experimental stage for solid tumors (including pancreatic cancer, mesothelioma, and glioblastoma), whose greater complexity make them harder to attack.
The new Nature study offers clues that could fuel further advances. The Penn team “profiled these cells at a level where we can almost say, ‘These are the characteristics that a T cell would need to survive 10 years,’” says Rouce, the physician at Texas Children’s Cancer Center.
One surprising finding involves how CAR-T cells change in the body over time. At first, those that Olson and Ludwig received showed the hallmarks of “killer” T-cells (also known as CD8 cells)—highly active lymphocytes bent on exterminating every tumor cell in sight. After several months, however, the population shifted toward “helper” T-cells (or CD4s), which aid in forming long-term immune memory but are normally incapable of direct aggression. Over the years, the numbers swung back and forth, until only helper cells remained. Those cells showed markers suggesting they were too exhausted to function—but in the lab, they were able not only to recognize but to destroy cancer cells.
June and his team suspect that those tired-looking helper cells had enough oomph to kill off any B cells Olson and Ludwig made, keeping the pair’s cancers permanently at bay. If so, that could prompt new approaches to selecting cells for CAR-T therapy. Maybe starting with a mix of cell types—not only CD8s, but CD4s and other varieties—would work better than using CD8s alone. Or perhaps inducing changes in cell populations at different times would help.
Another potential avenue for improvement is starting with healthier cells. Evidence from this and other trials hints that patients whose T cells are more robust to begin with respond better when their cells are used in CAR-T therapy. The Penn team recently completed a clinical trial in which CLL patients were treated with ibrutinib—a drug that enhances T-cell function—before their CAR-T cells were manufactured. The response rate, says David Porter, was “very high,” with most patients remaining cancer-free a year after being infused with the souped-up cells.
Such approaches, he adds, are essential to achieving the next phase in CAR-T therapy: “Getting it to work not just in more people, but in everybody.”

Doug Olson enjoys nature - and having a future.
Penn Medicine
To grasp what that could mean, it helps to talk with Doug Olson, who’s now 75. In the years since his infusion, he has watched his four children forge careers, and his grandkids reach their teens. He has built a business and enjoyed the rewards of semi-retirement. He’s done volunteer and advocacy work for cancer patients, run half-marathons, sailed the Caribbean, and ridden his bike along the sun-dappled roads of Silicon Valley, his current home.
And in his spare moments, he has just sat there feeling grateful. “You don’t really appreciate the effect of having a lethal disease until it’s not there anymore,” he says. “The world looks different when you have a future.”
This article was first published on Leaps.org on March 24, 2022.
Pseudoscience Is Rampant: How Not to Fall for It
A metaphorical rendering of scientific truth gone awry.
Whom to believe?
The relentless and often unpredictable coronavirus (SARS-CoV-2) has, among its many quirky terrors, dredged up once again the issue that will not die: science versus pseudoscience.
How does one learn to spot the con without getting a Ph.D. and spending years in a laboratory?
The scientists, experts who would be the first to admit they are not infallible, are now in danger of being drowned out by the growing chorus of pseudoscientists, conspiracy theorists, and just plain troublemakers that seem to be as symptomatic of the virus as fever and weakness.
How is the average citizen to filter this cacophony of information and misinformation posing as science alongside real science? While all that noise makes it difficult to separate the real stuff from the fakes, there is at least one positive aspect to it all.
A famous aphorism by one Charles Caleb Colton, a popular 19th-century English cleric and writer, says that "imitation is the sincerest form of flattery."
The frauds and the paranoid conspiracy mongers who would perpetrate false science on a susceptible public are at least recognizing the value of science—they imitate it. They imitate the ways in which science works and make claims as if they were scientists, because even they recognize the power of a scientific approach. They are inadvertently showing us how much we value science. Unfortunately they are just shabby counterfeits.
Separating real science from pseudoscience is not a new problem. Philosophers, politicians, scientists, and others have been worrying about this perhaps since science as we know it, a science based entirely on experiment and not opinion, arrived in the 1600s. The original charter of the British Royal Society, the first organized scientific society, stated that at their formal meetings there would be no discussion of politics, religion, or perpetual motion machines.
The first two of those for the obvious purpose of keeping the peace. But the third is interesting because at that time perpetual motion machines were one of the main offerings of the imitators, the bogus scientists who were sure that you could find ways around the universal laws of energy and make a buck on it. The motto adopted by the society was, and remains, Nullius in verba, Latin for "take nobody's word for it." Kind of an early version of Missouri's venerable state motto: "Show me."
You might think that telling phony science from the real thing wouldn't be so difficult, but events, historical and current, tell a very different story—often with tragic outcomes. Just one terrible example is the estimated 350,000 additional HIV deaths in South Africa directly caused by the now-infamous conspiracy theories of their own elected President no less (sound familiar?). It's surprisingly easy to dress up phony science as the real thing by simply adopting, or appearing to adopt, the trappings of science.
Thus, the anti-vaccine movement claims to be based on suspicion of authority, beginning with medical authority in this case, stemming from the fraudulent data published by the now-disgraced Andrew Wakefield, an English gastroenterologist. And it's true that much of science is based on suspicion of authority. Science got its start when the likes of Galileo and Copernicus claimed that the Church, the State, even Aristotle, could no longer be trusted as authoritative sources of knowledge.
But Galileo and those who followed him produced alternative explanations, and those alternatives were based on data that arose independently from many sources and generated a great deal of debate and, most importantly, could be tested by experiments that could prove them wrong. The anti-vaccine movement imitates science, still citing the discredited Wakefield report, but really offers nothing but suspicion—and that is paranoia, not science.
Similarly, there are those who try to cloak their nefarious motives in the trappings of science by claiming that they are taking the scientific posture of doubt. Science after all depends on doubt—every scientist doubts every finding they make. Every scientist knows that they can't possibly foresee all possible instances or situations in which they could be proven wrong, no matter how strong their data. Einstein was doubted for two decades, and cosmologists are still searching for experimental proofs of relativity. Science indeed progresses by doubt. In science revision is a victory.
But the imitators merely use doubt to suggest that science is not dependable and should not be used for informing policy or altering our behavior. They claim to be taking the legitimate scientific stance of doubt. Of course, they don't doubt everything, only what is problematic for their individual enterprises. They don't doubt the value of blood pressure medicine to control their hypertension. But they should, because every medicine has side effects and we don't completely understand how blood pressure is regulated and whether there may not be still better ways of controlling it.
But we use the pills we have because the science is sound even when it is not completely settled. Ask a hypertensive oil executive who would like you to believe that climate science should be ignored because there are too many uncertainties in the data, if he is willing to forgo his blood pressure medicine—because it, too, has its share of uncertainties and unwanted side effects.
The apparent success of pseudoscience is not due to gullibility on the part of the public. The problem is that science is recognized as valuable and that the imitators are unfortunately good at what they do. They take a scientific pose to gain your confidence and then distort the facts to their own purposes. How does one learn to spot the con without getting a Ph.D. and spending years in a laboratory?
"If someone claims to have the ultimate answer or that they know something for certain, the only thing for sure is that they are trying to fool you."
What can be done to make the distinction clearer? Several solutions have been tried—and seem to have failed. Radio and television shows about the latest scientific breakthroughs are a noble attempt to give the public a taste of good science, but they do nothing to help you distinguish between them and the pseudoscience being purveyed on the neighboring channel and its "scientific investigations" of haunted houses.
Similarly, attempts to inculcate what are called "scientific habits of mind" are of little practical help. These habits of mind are not so easy to adopt. They invariably require some amount of statistics and probability and much of that is counterintuitive—one of the great values of science is to help us to counter our normal biases and expectations by showing that the actual measurements may not bear them out.
Additionally, there is math—no matter how much you try to hide it, much of the language of science is math (Galileo said that). And half the audience is gone with each equation (Stephen Hawking said that). It's hard to imagine a successful program of making a non-scientifically trained public interested in adopting the rigors of scientific habits of mind. Indeed, I suspect there are some people, artists for example, who would be rightfully suspicious of changing their thinking to being habitually scientific. Many scientists are frustrated by the public's inability to think like a scientist, but in fact it is neither easy nor always desirable to do so. And it is certainly not practical.
There is a more intuitive and simpler way to tell the difference between the real thing and the cheap knock-off. In fact, it is not so much intuitive as it is counterintuitive, so it takes a little bit of mental work. But the good thing is it works almost all the time by following a simple, if as I say, counterintuitive, rule.
True science, you see, is mostly concerned with the unknown and the uncertain. If someone claims to have the ultimate answer or that they know something for certain, the only thing for sure is that they are trying to fool you. Mystery and uncertainty may not strike you right off as desirable or strong traits, but that is precisely where one finds the creative solutions that science has historically arrived at. Yes, science accumulates factual knowledge, but it is at its best when it generates new and better questions. Uncertainty is not a place of worry, but of opportunity. Progress lives at the border of the unknown.
How much would it take to alter the public perception of science to appreciate unknowns and uncertainties along with facts and conclusions? Less than you might think. In fact, we make decisions based on uncertainty every day—what to wear in case of 60 percent chance of rain—so it should not be so difficult to extend that to science, in spite of what you were taught in school about all the hard facts in those giant textbooks.
You can believe science that says there is clear evidence that takes us this far… and then we have to speculate a bit and it could go one of two or three ways—or maybe even some way we don't see yet. But like your blood pressure medicine, the stuff we know is reliable even if incomplete. It will lower your blood pressure, no matter that better treatments with fewer side effects may await us in the future.
Unsettled science is not unsound science. The honesty and humility of someone who is willing to tell you that they don't have all the answers, but they do have some thoughtful questions to pursue, are easy to distinguish from the charlatans who have ready answers or claim that nothing should be done until we are an impossible 100-percent sure.
Imitation may be the sincerest form of flattery.
The problem, as we all know, is that flattery will get you nowhere.
[Editor's Note: This article was originally published on June 8th, 2020 as part of a standalone magazine called GOOD10: The Pandemic Issue. Produced as a partnership among LeapsMag, The Aspen Institute, and GOOD, the magazine is available for free online.]
Henrietta Lacks' Cells Enabled Medical Breakthroughs. Is It Time to Finally Retire Them?
Henrietta Lacks, (1920-1951) unknowingly had her cells cultured and used in medical research.
For Victoria Tokarz, a third-year PhD student at the University of Toronto, experimenting with cells is just part of a day's work. Tokarz, 26, is studying to be a cell biologist and spends her time inside the lab manipulating muscle cells sourced from rodents to try to figure out how they respond to insulin. She hopes this research could someday lead to a breakthrough in our understanding of diabetes.
"People like to use HeLa cells because they're easy to use."
But in all her research, there is one cell culture that Tokarz refuses to touch. The culture is called HeLa, short for Henrietta Lacks, named after the 31-year-old tobacco farmer the cells were stolen from during a tumor biopsy she underwent in 1951.
"In my opinion, there's no question or experiment I can think of that validates stealing from and profiting off of a black woman's body," Tokarz says. "We're not talking about a reagent we created in a lab, a mixture of some chemicals. We're talking about a human being who suffered indescribably so we could profit off of her misfortune."
Lacks' suffering is something that, until recently, was not widely known. Born to a poor family in Roanoke, VA, Lacks was sent to live with her grandfather on the family tobacco farm at age four, shortly after the death of her mother. She gave birth to her first child at just fourteen, and two years later had another child with profound developmental disabilities. Lacks married her first cousin, David, in 1941 and the family moved to Maryland where they had three additional children.
But the real misfortune came in 1951, when Lacks told her cousins that she felt a hard "knot" in her womb. When Lacks went to Johns Hopkins hospital to have the knot examined, doctors discovered that the hard lump Henrietta felt was a rapidly-growing cervical tumor.
Before the doctors treated the tumor – inserting radium tubes into her vagina, in the hopes they could kill the cancer, Lacks' doctors clipped two tissue samples from her cervix, without Lacks' knowledge or consent. While it's considered widely unethical today, taking tissue samples from patients was commonplace at the time. The samples were sent to a cancer researcher at Johns Hopkins and Lacks continued treatment unsuccessfully until she died a few months later of metastatic cancer.
Lacks' story was not over, however: When her tissue sample arrived at the lab of George Otto Gey, the Johns Hopkins cancer researcher, he noticed that the cancerous cells grew at a shocking pace. Unlike other cell cultures that would die within a day or two of arriving at the lab, Lacks' cells kept multiplying. They doubled every 24 hours, and to this day, have never stopped.
Scientists would later find out that this growth was due to an infection of Human Papilloma Virus, or HPV, which is known for causing aggressive cancers. Lacks' cells became the world's first-ever "immortalized" human cell line, meaning that as long as certain environmental conditions are met, the cells can replicate indefinitely. Although scientists have cultivated other immortalized cell lines since then, HeLa cells remain a favorite among scientists due to their resilience, Tokarz says.
"People like to use HeLa cells because they're easy to use," Tokarz says. "They're easy to manipulate, because they're very hardy, and they allow for transection, which means expressing a protein in a cell that's not normally there. Other cells, like endothelial cells, don't handle those manipulations well."
Once the doctors at Johns Hopkins discovered that Lacks' cells could replicate indefinitely, they started shipping them to labs around the world to promote medical research. As they were the only immortalized cell line available at the time, researchers used them for thousands of experiments — some of which resulted in life-saving treatments. Jonas Salk's polio vaccine, for example, was manufactured using HeLa cells. HeLa cell research was also used to develop a vaccine for HPV, and for the development of in vitro fertilization and gene mapping. Between 1951 and 2018, HeLa cells have been cited in over 110,000 publications, according to a review from the National Institutes of Health.
But while some scientists like Tokarz are thankful for the advances brought about by HeLa cells, they still believe it's well past time to stop using them in research.
"Am I thankful we have a polio vaccine? Absolutely. Do I resent the way we came to have that vaccine? Absolutely," Tokarz says. "We could have still arrived at those same advances by treating her as the human being she is, not just a specimen."
Ethical considerations aside, HeLa is no longer the world's only available cell line – nor, Tokarz argues, are her cells the most suitable for every type of research. "The closer you can get to the physiology of the thing you're studying, the better," she says. "Now we have the ability to use primary cells, which are isolated from a person and put right into the culture dish, and those don't have the same mutations as cells that have been growing for 20 years. We didn't have the expertise to do that initially, but now we do."
Raphael Valdivia, a professor of molecular genetics and microbiology at Duke University School of Medicine, agrees that HeLa cells are no longer optimal for most research. "A lot of scientists are moving away from HeLa cells because they're so unstable," he says. "They mutate, they rearrange chromosomes to become adaptive, and different batches of cells evolve separately from each other. The HeLa cells in my lab are very different than the ones down the hall, and that means sometimes we can't replicate our results. We have to go back to an earlier batch of cells in the freezer and re-test."
Still, the idea of retiring the cells completely doesn't make sense, Valdivia says: "To some extent, you're beholden to previous research. You need to be able to confirm findings that happen in earlier studies, and to do that you need to use the same cell line that other researchers have used."
"Ethics is not black and white, and sometimes there's no such thing as a straightforward ethical or unethical choice."
"The way in which the cells were taken – without patient consent – is completely inappropriate," says Yann Joly, associate professor at the Faculty of Medicine in Toronto and Research Director at the Centre of Genomics and Policy. "The question now becomes, what can we do about it now? What are our options?"
While scientists are not able to erase what was done to Henrietta Lacks, Joly argues that retiring her cells is also non-consensual, assuming – maybe incorrectly – what Henrietta would have wanted, without her input. Additionally, Joly points out that other immortalized human cell lines are fraught with what some people consider to be ethical concerns as well, such as the human embryonic kidney cell line, commonly referred to as HEK-293, that was derived from an aborted female fetus. "Just because you're using another kind of cell doesn't mean it's devoid of ethical issue," he says.
Seemingly, the one thing scientists can agree on is that Henrietta Lacks was mistreated by the medical community. But even so, retiring her cells from medical research is not an obvious solution. Scientists are now using HeLa cells to better understand how the novel coronavirus affects humans, and this knowledge will inform how researchers develop a COVID-19 vaccine.
"Ethics is not black and white, and sometimes there's no such thing as a straightforward ethical or unethical choice," Joly says. "If [ethics] were that easy, nobody would need to teach it."