Study Shows “Living Drug” Can Provide a Lasting Cure for Cancer

A recent study by researchers at the University of Pennsylvania examined how CAR-T therapy helped Doug Olson beat a cancer death sentence for over a decade - and how it could work for more people.
Doug Olson was 49 when he was diagnosed with chronic lymphocytic leukemia, a blood cancer that strikes 21,000 Americans annually. Although the disease kills most patients within a decade, Olson’s case progressed more slowly, and courses of mild chemotherapy kept him healthy for 13 years. Then, when he was 62, the medication stopped working. The cancer had mutated, his doctor explained, becoming resistant to standard remedies. Harsher forms of chemo might buy him a few months, but their side effects would be debilitating. It was time to consider the treatment of last resort: a bone-marrow transplant.
Olson, a scientist who developed blood-testing instruments, knew the odds. There was only a 50 percent chance that a transplant would cure him. There was a 20 percent chance that the agonizing procedure—which involves destroying the patient’s marrow with chemo and radiation, then infusing his blood with donated stem cells—would kill him. If he survived, he would face the danger of graft-versus-host disease, in which the donor’s cells attack the recipient’s tissues. To prevent it, he would have to take immunosuppressant drugs, increasing the risk of infections. He could end up with pneumonia if one of his three grandchildren caught a sniffle. “I was being pushed into a corner,” Olson recalls, “with very little room to move.”
Soon afterward, however, his doctor revealed a possible escape route. He and some colleagues at the University of Pennsylvania’s Abramson Cancer Center were starting a clinical trial, he said, and Olson—still mostly symptom-free—might be a good candidate. The experimental treatment, known as CAR-T therapy, would use genetic engineering to turn his T lymphocytes (immune cells that guard against viruses and other pathogens) into a weapon against cancer.
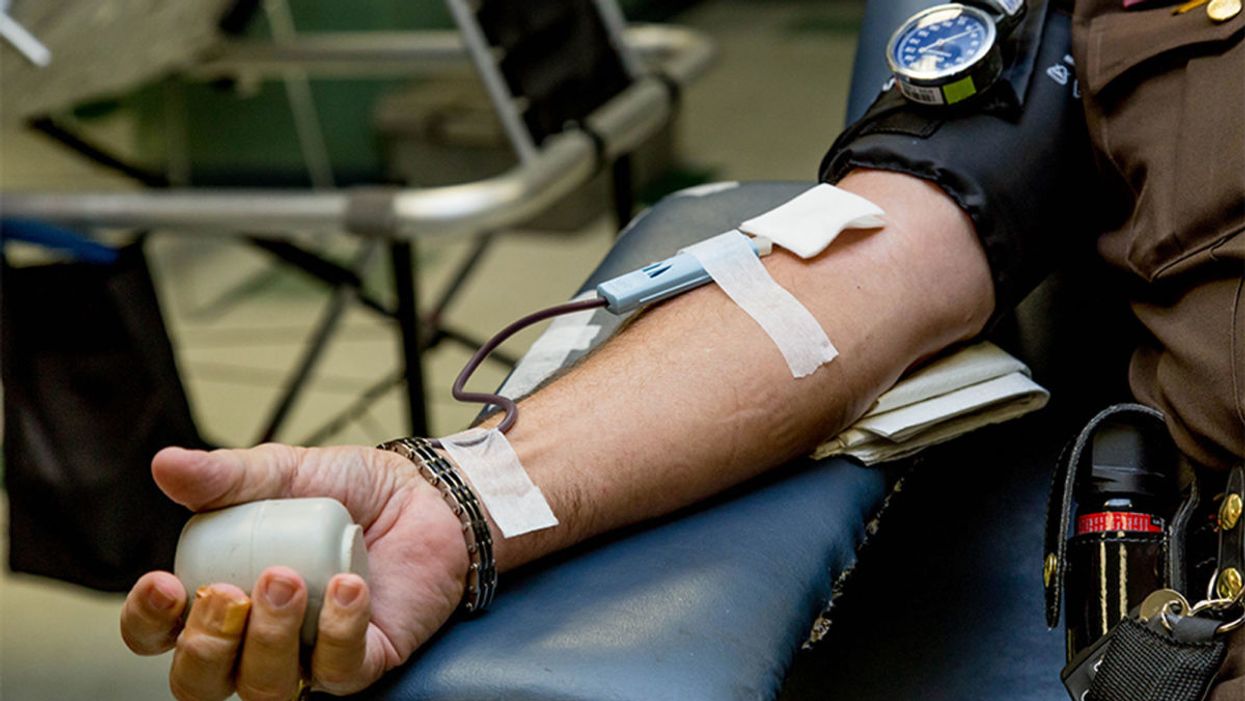
In September 2010, technicians took some of Olson’s T cells to a laboratory, where they were programmed with new molecular marching orders and coaxed to multiply into an army of millions. When they were ready, a nurse inserted a catheter into his neck. At the turn of a valve, his soldiers returned home, ready to do battle.
“I felt like I’d won the lottery,” Olson says. But he was only the second person in the world to receive this “living drug,” as the University of Pennsylvania investigators called it. No one knew how long his remission would last.
Three weeks later, Olson was slammed with a 102-degree fever, nausea, and chills. The treatment had triggered two dangerous complications: cytokine release syndrome, in which immune chemicals inflame the patient’s tissues, and tumor lysis syndrome, in which toxins from dying cancer cells overwhelm the kidneys. But the crisis passed quickly, and the CAR-T cells fought on. A month after the infusion, the doctor delivered astounding news: “We can’t find any cancer in your body.”
“I felt like I’d won the lottery,” Olson says. But he was only the second person in the world to receive this “living drug,” as the University of Pennsylvania investigators called it. No one knew how long his remission would last.
An Unexpected Cure
In February 2022, the same cancer researchers reported a remarkable milestone: the trial’s first two patients had survived for more than a decade. Although Olson’s predecessor—a retired corrections officer named Bill Ludwig—died of COVID-19 complications in early 2021, both men had remained cancer-free. And the modified immune cells continued to patrol their territory, ready to kill suspected tumor cells the moment they arose.
“We can now conclude that CAR-T cells can actually cure patients with leukemia,” University of Pennsylvania immunologist Carl June, who spearheaded the development of the technique, told reporters. “We thought the cells would be gone in a month or two. The fact that they’ve survived 10 years is a major surprise.”
Even before the announcement, it was clear that CAR-T therapy could win a lasting reprieve for many patients with cancers that were once a death sentence. Since the Food and Drug Administration approved June’s version (marketed as Kymriah) in 2017, the agency has greenlighted five more such treatments for various types of leukemia, lymphoma, and myeloma. “Every single day, I take care of patients who would previously have been told they had no options,” says Rayne Rouce, a pediatric hematologist/oncologist at Texas Children’s Cancer Center. “Now we not only have a treatment option for those patients, but one that could potentially be the last therapy for their cancer that they’ll ever have to receive.”

Immunologist Carl June, middle, spearheaded development of the CAR-T therapy that gave patients Bill Ludwig, left, and Doug Olson, right, a lengthy reprieve on their terminal cancer diagnoses.
Penn Medicine
Yet the CAR-T approach doesn’t help everyone. So far, it has only shown success for blood cancers—and for those, the overall remission rate is 30 to 40 percent. “When it works, it works extraordinarily well,” says Olson’s former doctor, David Porter, director of Penn’s blood and bone marrow transplant program. “It’s important to know why it works, but it’s equally important to know why it doesn’t—and how we can fix that.”
The team’s study, published in the journal Nature, offers a wealth of data on what worked for these two patients. It may also hold clues for how to make the therapy effective for more people.
Building a Better T Cell
Carl June didn’t set out to cure cancer, but his serendipitous career path—and a personal tragedy—helped him achieve insights that had eluded other researchers. In 1971, hoping to avoid combat in Vietnam, he applied to the U.S. Naval Academy in Annapolis, Maryland. June showed a knack for biology, so the Navy sent him on to Baylor College of Medicine. He fell in love with immunology during a fellowship researching malaria vaccines in Switzerland. Later, the Navy deployed him to the Fred Hutchinson Cancer Research Center in Seattle to study bone marrow transplantation.
There, June became part of the first research team to learn how to culture T cells efficiently in a lab. After moving on to the National Naval Medical Center in the ’80s, he used that knowledge to combat the newly emerging AIDS epidemic. HIV, the virus that causes the disease, invades T cells and eventually destroys them. June and his post-doc Bruce Levine developed a method to restore patients’ depleted cell populations, using tiny magnetic beads to deliver growth-stimulating proteins. Infused into the body, the new T cells effectively boosted immune function.
In 1999, after leaving the Navy, June joined the University of Pennsylvania. His wife, who’d been diagnosed with ovarian cancer, died two years later, leaving three young children. “I had not known what it was like to be on the other side of the bed,” he recalls. Watching her suffer through grueling but futile chemotherapy, followed by an unsuccessful bone-marrow transplant, he resolved to focus on finding better cancer treatments. He started with leukemia—a family of diseases in which mutant white blood cells proliferate in the marrow.
Cancer is highly skilled at slipping through the immune system’s defenses. T cells, for example, detect pathogens by latching onto them with receptors designed to recognize foreign proteins. Leukemia cells evade detection, in part, by masquerading as normal white blood cells—that is, as part of the immune system itself.
June planned to use a viral vector no one had tried before: HIV.
To June, chimeric antigen receptor (CAR) T cells looked like a promising tool for unmasking and destroying the impostors. Developed in the early ’90s, these cells could be programmed to identify a target protein, and to kill any pathogen that displayed it. To do the programming, you spliced together snippets of DNA and inserted them into a disabled virus. Next, you removed some of the patient’s T cells and infected them with the virus, which genetically hijacked its new hosts—instructing them to find and slay the patient’s particular type of cancer cells. When the T cells multiplied, their descendants carried the new genetic code. You then infused those modified cells into the patient, where they went to war against their designated enemy.
Or that’s what happened in theory. Many scientists had tried to develop therapies using CAR-T cells, but none had succeeded. Although the technique worked in lab animals, the cells either died out or lost their potency in humans.
But June had the advantage of his years nurturing T cells for AIDS patients, as well as the technology he’d developed with Levine (who’d followed him to Penn with other team members). He also planned to use a viral vector no one had tried before: HIV, which had evolved to thrive in human T cells and could be altered to avoid causing disease. By the summer of 2010, he was ready to test CAR-T therapy against chronic lymphocytic leukemia (CLL), the most common form of the disease in adults.
Three patients signed up for the trial, including Doug Olson and Bill Ludwig. A portion of each man’s T cells were reprogrammed to detect a protein found only on B lymphocytes, the type of white blood cells affected by CLL. Their genetic instructions ordered them to destroy any cell carrying the protein, known as CD19, and to multiply whenever they encountered one. This meant the patients would forfeit all their B cells, not just cancerous ones—but regular injections of gamma globulins (a cocktail of antibodies) would make up for the loss.
After being infused with the CAR-T cells, all three men suffered high fevers and potentially life-threatening inflammation, but all pulled through without lasting damage. The third patient experienced a partial remission and survived for eight months. Olson and Ludwig were cured.
Learning What Works
Since those first infusions, researchers have developed reliable ways to prevent or treat the side effects of CAR-T therapy, greatly reducing its risks. They’ve also been experimenting with combination therapies—pairing CAR-T with chemo, cancer vaccines, and immunotherapy drugs called checkpoint inhibitors—to improve its success rate. But CAR-T cells are still ineffective for at least 60 percent of blood cancer patients. And they remain in the experimental stage for solid tumors (including pancreatic cancer, mesothelioma, and glioblastoma), whose greater complexity make them harder to attack.
The new Nature study offers clues that could fuel further advances. The Penn team “profiled these cells at a level where we can almost say, ‘These are the characteristics that a T cell would need to survive 10 years,’” says Rouce, the physician at Texas Children’s Cancer Center.
One surprising finding involves how CAR-T cells change in the body over time. At first, those that Olson and Ludwig received showed the hallmarks of “killer” T-cells (also known as CD8 cells)—highly active lymphocytes bent on exterminating every tumor cell in sight. After several months, however, the population shifted toward “helper” T-cells (or CD4s), which aid in forming long-term immune memory but are normally incapable of direct aggression. Over the years, the numbers swung back and forth, until only helper cells remained. Those cells showed markers suggesting they were too exhausted to function—but in the lab, they were able not only to recognize but to destroy cancer cells.
June and his team suspect that those tired-looking helper cells had enough oomph to kill off any B cells Olson and Ludwig made, keeping the pair’s cancers permanently at bay. If so, that could prompt new approaches to selecting cells for CAR-T therapy. Maybe starting with a mix of cell types—not only CD8s, but CD4s and other varieties—would work better than using CD8s alone. Or perhaps inducing changes in cell populations at different times would help.
Another potential avenue for improvement is starting with healthier cells. Evidence from this and other trials hints that patients whose T cells are more robust to begin with respond better when their cells are used in CAR-T therapy. The Penn team recently completed a clinical trial in which CLL patients were treated with ibrutinib—a drug that enhances T-cell function—before their CAR-T cells were manufactured. The response rate, says David Porter, was “very high,” with most patients remaining cancer-free a year after being infused with the souped-up cells.
Such approaches, he adds, are essential to achieving the next phase in CAR-T therapy: “Getting it to work not just in more people, but in everybody.”

Doug Olson enjoys nature - and having a future.
Penn Medicine
To grasp what that could mean, it helps to talk with Doug Olson, who’s now 75. In the years since his infusion, he has watched his four children forge careers, and his grandkids reach their teens. He has built a business and enjoyed the rewards of semi-retirement. He’s done volunteer and advocacy work for cancer patients, run half-marathons, sailed the Caribbean, and ridden his bike along the sun-dappled roads of Silicon Valley, his current home.
And in his spare moments, he has just sat there feeling grateful. “You don’t really appreciate the effect of having a lethal disease until it’s not there anymore,” he says. “The world looks different when you have a future.”
This article was first published on Leaps.org on March 24, 2022.
A Single Blood Test May Soon Replace Your Annual Physical
The measurement of proteins in one blood test could provide a comprehensive snapshot of a person's health in the near future.
For all the excitement over "personalized medicine" in the last two decades, its promise has not fully come to pass. Consider your standard annual physical.
Scientists have measured thousands of proteins from a single blood test to assess many individualized health conditions at once.
Your doctor still does a blood test to check your cholesterol and gauge your risk for heart disease by considering traditional risk factors (like smoking, diabetes, blood pressure) — an evaluation that has not changed in decades.
But a high-risk number alone is not enough to tell accurately whether you will suffer from heart disease. It just reflects your risk compared to population-level averages. In other words, not every person with elevated "bad" cholesterol will have a heart attack, so how can doctors determine who truly needs to give up the cheeseburgers and who doesn't?
Now, an emerging area of research may unlock some real-time answers. For the first time, as reported in the journal Nature Medicine last week, scientists have measured thousands of proteins from a single blood test to assess many individualized health conditions at once, including liver and kidney function, diabetes risk, body fat, cardiopulmonary fitness, and even smoking and alcohol consumption. Proteins can give a clear snapshot of how your body is faring at any given moment, as well as a sneak preview at what diseases may be lurking under the surface.
"Years from now," says study co-author Peter Ganz of UCSF, "we will probably be looking back on this paper as a milestone in personalized medicine."
We spoke to Ganz about the significance of this milestone. Our interview has been edited and condensed.
Is this the first study of its kind?
Yes, it is. This is a study where we measured 5,000 proteins at once to look for patterns that could either predict the risk of future diseases or inform the current state of health. Previous to this, people have measured typically one protein at a time, and some of these individual proteins have made it into clinical practice.
An example would be a protein called C-reactive protein, which is a measure of inflammation and is used sometimes in cardiology to predict the risk of future heart attacks. But what's really new is this scale. We wanted to get away from just focusing on one problem that the patient may have at a time, whether it's heart disease or kidney disease, and by measuring a much greater number of proteins, the hope is that we could inform the health of ultimately just about every organ in the body or every tissue. It's a step forward for what I would call "a one-stop shop."
"I'm very excited about personalized medicine through proteins as opposed to genes because you get both the nature and nurture."
Three things get me excited about this. One is the convenience for the patient of a single test to determine many different diseases. The second thing is the healthcare cost savings. We estimated what the cost would be to get these 11 healthcare measures that we reported on using traditional testing and the cost was upwards of 3,000 British pounds. And even though I don't know for sure what the cost of the protein tests would ultimately be, [it could come down to about $50 to $100].
The last thing is that the measurement of proteins is part of what people have called personalized medicine or precision medicine. If you look at risk factors across the population, it may not apply to individuals. In contrast, proteins are downstream of risk factors. So proteins actually tell us whether the traditional risk factors have set in motion the necessary machinery to cause disease. Proteins are the worker bees that regulate what the human body does, and so if you can find some anomalies in the proteins, that may inform us if a disease is likely to be ongoing even in its earliest stages.
Does protein testing have advantages over genetic testing for predicting future health risks?
The problem with genomics is that genes usually don't take care of the environment. It's a blueprint, but your blueprint has no idea what you will be exposed to during your lifetime in terms of the environment and lifestyle that you may choose and medications that you may be on. These are things that proteins can account for. I'm very excited about personalized medicine through proteins as opposed to genes because you get both the nature and nurture as opposed to genomics, which only gives you nature but doesn't account for anything else.
Proteins can also be tracked over time and that's not something you can do with genes. So if your behavior improves, your genes won't change, but your proteins will.
Could this new test become a regular feature of your annual physical?
That's the idea. This would be basically almost a standalone test that you could have done every year. And hopefully you wouldn't need other tests to complement this. This could be your yearly physical.
How much more does it need to be validated before it can enter the clinic and patients can trust the results?
This was a proof-of concept study. To really make this useful, we need to expand from 11 measures of health to a hundred or more health insights, to cover the whole body. And we need to expand this to all racial groups. Three of the five centers in the study were European – all Caucasian – so it's one of our high priorities to find groups of patients with better representation of minorities.
When do you expect doctors to be routinely giving this test to patients?
Much closer to five years than 20 years. We're scaling up from 11 disease states to 100, and many of those studies are underway. Results should be done within three to five years.
Do you think insurance will cover it?
Good question. I have been approached by an insurance company that wanted to understand the product better – a major insurer, with the possibility that this could actually be cost saving.
I have to ask you a curveball -- do you think that the downfall of Theranos will make consumers hesitant to trust a new technology that relies on using a single blood sample to screen for multiple health risks?
[Laughs] You're not the first person to ask me that today. I actually got a call from Elizabeth Holmes [in 2008 when I was at Harvard]. I met with her for an afternoon and met her team two more times. I gave them advice that they completely disregarded.
In many ways, what we do is diametrically opposite to Theranos. They had a culture of secrecy, and what we do is about openness. We publish, like this paper in Nature Medicine, to show the scientific details. Our supplement is much longer than the typical academic paper. We reveal everything we know. A lot of the research we do is funded by [the National Institutes of Health], and they have strict expectations about data sharing. So we agree to make the data available on a public website. If there is something we haven't done with the data, others can do it.
So you're saying that this is not another Theranos.
No, God forbid. We hope to be the opposite.
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.
We Pioneered a Technology to Save Millions of Poor Children, But a Worldwide Smear Campaign Has Blocked It
On left, a picture of white rice next to Golden Rice, and on right, a girl who lost one eye due to vitamin A deficiency.
In a few weeks it will be 20 years that we three have been working together. Our project has been independently praised as one of the most influential of all projects of the last 50 years.
Two of us figured out how to make rice produce a source of vitamin A, and the rice becomes a golden color instead of white.
The project's objectives have been admired by some and vilified by others. It has directly involved teams of highly motivated people from a handful of nations, from both the private and public sector. A book, dedicated to the three of us, has been written about our work. Nevertheless, success has, so far, eluded us all. The story of our thwarted efforts is a tragedy that we hope will soon – finally – reach a milestone of potentially profound significance for humanity.
So, what have we been working on, and why haven't we succeeded yet?
Food: everybody needs it, and many are fortunate enough to have enough, even too much of it. Food is a highly emotional subject on every continent and in every culture. For a healthy life our food has to provide energy, as well as, in very small amounts, minerals and vitamins. A varied diet, easily achieved and common in industrialised countries, provides everything.
But poor people in countries where rice is grown often eat little else. White rice only provides energy: no minerals or vitamins. And the lack of one of the vitamins, vitamin A, is responsible for killing around 4,500 poor children every day. Lack of vitamin A is the biggest killer of children, and also the main cause of irreversible childhood blindness.
Our project is about fixing this one dietary deficiency – vitamin A – in this one crop – rice – for this one group of people. It is a huge group though: half of the world's population live by eating a lot of rice every day. Two of us (PB & IP) figured out how to make rice produce a source of vitamin A, and the rice becomes a golden color instead of white. The source is beta-carotene, which the human body converts to vitamin A. Beta-carotene is what makes carrots orange. Our rice is called "Golden Rice."
The technology has been donated to assist those rice eaters who suffer from vitamin A deficiency ('VAD') so that Golden Rice will cost no more than white rice, there will be no restrictions on the small farmers who grow it, and nothing extra to pay for the additional nutrition. Very small amounts of beta-carotene will contribute to alleviation of VAD, and even the earliest version of Golden Rice – which had smaller amounts than today's Golden Rice - would have helped. So far, though, no small farmer has been allowed to grow it. What happened?
To create Golden Rice, it was necessary to precisely add two genes to the 30,000 genes normally present in rice plants. One of the genes is from maize, also known as corn, and the other from a commonly eaten soil bacterium. The only difference from white rice is that Golden Rice contains beta-carotene.
It has been proven to be safe to man and the environment, and consumption of only small quantities of Golden Rice will combat VAD, with no chance of overdosing. All current Golden Rice results from one introduction of these two genes in 2004. But the use of that method – once, 15 years ago - means that Golden Rice is a 'GMO' ('genetically modified organism'). The enzymes used in the manufacture of bread, cheese, beer and wine, and the insulin which diabetics take to keep them alive, are all made from GMOs too.
The first GMO crops were created by agri-business companies. Suspicion of the technology and suspicion of commercial motivations merged, only for crop (but not enzymes or pharmaceutical) applications of GMO technology. Activists motivated by these suspicions were successful in getting the 'precautionary principle' incorporated in an international treaty which has been ratified by 166 countries and the European Union – The Cartagena Protocol.
The equivalent of 13 jumbo jets full of children crashes into the ground every day and kills them all, because of vitamin A deficiency.
This protocol is the basis of national rules governing the introduction of GMO crops in every signatory country. Government regulators in, and for, each country must agree before a GMO crop can be 'registered' to be allowed to be used by the public in that country. Currently regulatory decisions to allow Golden Rice release are being considered in Bangladesh and the Philippines.
The Cartagena Protocol obliges the regulators in each country to consider all possible risks, and to take no account of any possible benefits. Because the anti-gmo-activists' initial concerns were principally about the environment, the responsibility for governments' regulation for GMO crops – even for Golden Rice, a public health project delivered through agriculture – usually rests with the Ministry of the Environment, not the Ministry of Health or the Ministry of Agriculture.
Activists discovered, before Golden Rice was created, that inducing fear of GMO food crops from 'multinational agribusinesses' was very good for generating donations from a public that was largely illiterate about food technology and production. And this source of emotionally charged donations would cease if Golden Rice was proven to save sight and lives, because Golden Rice represented the opposite of all the tropes used in anti-GMO campaigns.
Golden Rice is created to deliver a consumer benefit, it is not for profit – to multinational agribusiness or anyone else; the technology originated in the public sector and is being delivered through the public sector. It is entirely altruistic in its motivations; which activists find impossible to accept. So, the activists believed, suspicion against Golden Rice had to be amplified, Golden Rice had to be stopped: "If we lose the Golden Rice battle, we lose the GMO war."
Activism continues to this day. And any Environment Ministry, with no responsibility for public health or agriculture, and of course an interest in avoiding controversy about its regulatory decisions, is vulnerable to such activism.
The anti-GMO crop campaigns, and especially anti-Golden Rice campaigns, have been extraordinarily effective. If so much regulation by governments is required, surely there must be something to be suspicious about: 'There is no smoke without fire'. The suspicion pervades research institutions and universities, the publishers of scientific journals and The World Health Organisation, and UNICEF: even the most scientifically literate are fearful of entanglement in activist-stoked public controversy.
The equivalent of 13 jumbo jets full of children crashes into the ground every day and kills them all, because of VAD. Yet the solution of Golden Rice, developed by national scientists in the counties where VAD is endemic, is ignored because of fear of controversy, and because poor children's deaths can be ignored without controversy.
Perhaps more controversy lies in not taking scientifically based regulatory decisions than in taking them.
The tide is turning, however. 151 Nobel Laureates, a very significant proportion of all Nobel Laureates, have called on the UN, governments of the world, and Greenpeace to cease their unfounded vilification of GMO crops in general and Golden Rice in particular. A recent Golden Rice article commented, "What shocks me is that some activists continue to misrepresent the truth about the rice. The cynic in me expects profit-driven multinationals to behave unethically, but I want to think that those voluntarily campaigning on issues they care about have higher standards."
The recently published book has exposed the frustrating saga in simple detail. And the publicity from all the above is perhaps starting to change the balance of where controversy lies. Perhaps more controversy lies in not taking scientifically based regulatory decisions than in taking them.
But until they are taken, while there continues a chance of frustrating the objectives of the Golden Rice project, the antagonism will continue. And despite a solution so close at hand, VAD-induced death and blindness, and the misery of affected families, will continue also.
© The Authors 2019. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License, which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver applies to the data made available in this article, unless otherwise stated.