Carl Zimmer: Genetically Editing Humans Should Not Be Our Biggest Worry
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.

The award-winning New York Times science writer Carl Zimmer.
Carl Zimmer, the award-winning New York Times science writer, recently published a stellar book about human heredity called "She Has Her Mother's Laugh." Truly a magnum opus, the book delves into the cultural and scientific evolution of genetics, the field's outsize impact on society, and the new ways we might fundamentally alter our species and our planet.
"I was only prepared to write about how someday we would cross this line, and actually, we've already crossed it."
Zimmer spoke last week with editor-in-chief Kira Peikoff about the international race to edit the genes of human embryos, the biggest danger he sees for society (hint: it's not super geniuses created by CRISPR), and some outlandish possibilities for how we might reproduce in the future. This interview has been edited and condensed for clarity.
I was struck by the number of surprises you uncovered while researching human heredity, like how fetal cells can endure for a lifetime in a mother's body and brain. What was one of the biggest surprises for you?
Something that really jumped out for me was for the section on genetically modifying people. It does seem incredibly hypothetical. But then I started looking into mitochondrial replacement therapy, so-called "three parent babies." I was really surprised to discover that almost by accident, a number of genetically modified people were created this way [in the late 90s and early 2000s]. They walk among us, and they're actually fine as far as anyone can tell. I was only prepared to write about how someday we would cross this line, and actually, we've already crossed it.
And now we have the current arms race between the U.S. and China to edit diseases out of human embryos, with China being much more willing and the U.S. more reluctant. Do you think it's more important to get ahead or to proceed as ethically as possible?
I would prefer a middle road. I think that rushing into tinkering with the features of human heredity could be a disastrous mistake for a lot of reasons. On the other hand, if we completely retreat from it out of some vague fear, I think that we won't take advantage of the actual benefits that this technology might have that are totally ethically sound.
I think the United Kingdom is actually showing how you can go the middle route with mitochondrial replacement therapy. The United States has just said nope, you can't do it at all, and you have Congressmen talking about how it's just playing God or Frankenstein. And then there are countries like Mexico or the Ukraine where people are doing mitochondrial replacement therapy because there are no regulations at all. It's a wild west situation, and that's not a good idea either.
But in the UK, they said alright, well let's talk about this, let's have a debate in Parliament, and they did, and then the government came up with a well thought-through policy. They decided that they were going to allow for this, but only in places that applied for a license, and would be monitored, and would keep track of the procedure and the health of these children and actually have real data going forward. I would imagine that they're going to very soon have their first patients.
As you mentioned, one researcher recently traveled to Mexico from New York to carry out the so-called "three-parent baby" procedure in order to escape the FDA's rules. What's your take on scientists having to leave their own jurisdictions to advance their research programs under less scrutiny?
I think it's a problem when people who have a real medical need have to leave their own country to get truly effective treatment for it. On the other hand, we're seeing lots of people going abroad to countries that don't monitor all the claims that clinics are making about their treatments. So you have stem cell clinics in all sorts of places that are making all sorts of ridiculous promises. They're not delivering those results, and in some cases, they're doing harm.
"Advances in stem cell biology and reproductive biology are a much bigger challenge to our conventional ideas about heredity than CRISPR is."
It's a tricky tension for sure. Speaking of gene editing humans, you mention in the book that one of the CRISPR pioneers, Jennifer Doudna, now has recurring nightmares about Hitler. Do you think that her fears about eugenics being revived with gene editing are justified?
The word "eugenics" has a long history and it's meant different things to different people. So we have to do a better job of talking about it in the future if we really want to talk about the risks and the promises of technology like CRISPR. Eugenics in its most toxic form was an ideology that let governments, including the United States, sterilize their own citizens by the tens of thousands. Then Nazi Germany also used eugenics as a justification to exterminate many more people.
Nobody's talking about that with CRISPR. Now, are people concerned that we are going to wipe out lots of human genetic diversity with it? That would be a bad thing, but I'm skeptical that would actually ever happen. You would have to have some sort of science fiction one-world government that required every new child to be born with IVF. It's not something that keeps me up at night. Honestly, I think we have much bigger problems to worry about.
What is the biggest danger relating to genetics that we should be aware of?
Part of what made eugenics such a toxic ideology was that it was used as a justification for indifference. In other words, if there are problems in society, like a large swath of people who are living in poverty, well, there's nothing you can do about it because it must be due to genetics.
If you look at genetics as being the sole place where you can solve humanity's problems, then you're going to say well, there's no point in trying to clean up the environment or trying to improve human welfare.
A major theme in your book is that we should not narrow our focus on genes as the only type of heredity. We also may inherit some epigenetic marks, some of our mother's microbiome and mitochondria, and importantly, our culture and our environment. Why does an expanded view of heredity matter?
We should think about the world that our children are going to inherit, and their children, and their children. They're going to inherit our genes, but they're also going to inherit this planet and we're doing things that are going to have an incredibly long-lasting impact on it. I think global warming is one of the biggest. When you put carbon dioxide into the air, it stays there for a very, very long time. If we stopped emitting carbon dioxide now, the Earth would stay warm for many centuries. We should think about tinkering with the future of genetic heredity, but I think we should also be doing that with our environmental heredity and our cultural heredity.
At the end of the book, you discuss some very bizarre possibilities for inheritance that could be made possible through induced pluripotent stem cell technology and IVF -- like four-parent babies, men producing eggs, and children with 8-celled embryos as their parents. If this is where reproductive medicine is headed, how can ethics keep up?
I'm not sure actually. I think that these advances in stem cell biology and reproductive biology are a much bigger challenge to our conventional ideas about heredity than CRISPR is. With CRISPR, you might be tweaking a gene here and there, but they're still genes in an embryo which then becomes a person, who would then have children -- the process our species has been familiar with for a long time.
"We have to recognize that we need a new language that fits with the science of heredity in the 21st century."
We all assume that there's no way to find a fundamentally different way of passing down genes, but it turns out that it's not really that hard to turn a skin cell from a cheek scraping into an egg or sperm. There are some challenges that still have to be worked out to make this something that could be carried out a lot in labs, but I don't see any huge barriers to it. Ethics doesn't even have the language to discuss the possibilities. Like for example, one person producing both male and female sex cells, which are then fertilized to produce embryos so that you have a child who only has one parent. How do we even talk about that? I don't know. But that's coming up fast.
We haven't developed our language as quickly as the technology itself. So how do we move forward?
We have to recognize that we need a new language that fits with the science of heredity in the 21st century. I think one of the biggest problems we have as a society is that most of our understanding about these issues largely comes from what we learned in grade school and high school in biology class. A high school biology class, even now, gets up to Mendel and then stops. Gregor Mendel is a great place to start, but it's a really bad place to stop talking about heredity.
[Ed. Note: Zimmer's book can be purchased through your retailer of choice here.]
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.
Thanks to safety cautions from the COVID-19 pandemic, a strain of influenza has been completely eliminated.
If you were one of the millions who masked up, washed your hands thoroughly and socially distanced, pat yourself on the back—you may have helped change the course of human history.
Scientists say that thanks to these safety precautions, which were introduced in early 2020 as a way to stop transmission of the novel COVID-19 virus, a strain of influenza has been completely eliminated. This marks the first time in human history that a virus has been wiped out through non-pharmaceutical interventions, such as vaccines.
The flu shot, explained
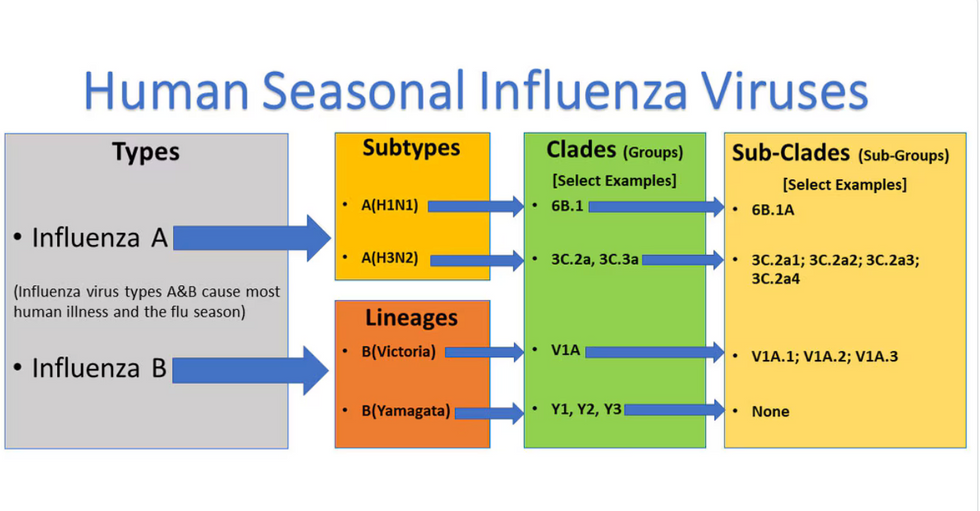
Influenza viruses type A and B are responsible for the majority of human illnesses and the flu season.
Centers for Disease Control
For more than a decade, flu shots have protected against two types of the influenza virus–type A and type B. While there are four different strains of influenza in existence (A, B, C, and D), only strains A, B, and C are capable of infecting humans, and only A and B cause pandemics. In other words, if you catch the flu during flu season, you’re most likely sick with flu type A or B.
Flu vaccines contain inactivated—or dead—influenza virus. These inactivated viruses can’t cause sickness in humans, but when administered as part of a vaccine, they teach a person’s immune system to recognize and kill those viruses when they’re encountered in the wild.
Each spring, a panel of experts gives a recommendation to the US Food and Drug Administration on which strains of each flu type to include in that year’s flu vaccine, depending on what surveillance data says is circulating and what they believe is likely to cause the most illness during the upcoming flu season. For the past decade, Americans have had access to vaccines that provide protection against two strains of influenza A and two lineages of influenza B, known as the Victoria lineage and the Yamagata lineage. But this year, the seasonal flu shot won’t include the Yamagata strain, because the Yamagata strain is no longer circulating among humans.
How Yamagata Disappeared

Flu surveillance data from the Global Initiative on Sharing All Influenza Data (GISAID) shows that the Yamagata lineage of flu type B has not been sequenced since April 2020.
Nature
Experts believe that the Yamagata lineage had already been in decline before the pandemic hit, likely because the strain was naturally less capable of infecting large numbers of people compared to the other strains. When the COVID-19 pandemic hit, the resulting safety precautions such as social distancing, isolating, hand-washing, and masking were enough to drive the virus into extinction completely.
Because the strain hasn’t been circulating since 2020, the FDA elected to remove the Yamagata strain from the seasonal flu vaccine. This will mark the first time since 2012 that the annual flu shot will be trivalent (three-component) rather than quadrivalent (four-component).
Should I still get the flu shot?
The flu shot will protect against fewer strains this year—but that doesn’t mean we should skip it. Influenza places a substantial health burden on the United States every year, responsible for hundreds of thousands of hospitalizations and tens of thousands of deaths. The flu shot has been shown to prevent millions of illnesses each year (more than six million during the 2022-2023 season). And while it’s still possible to catch the flu after getting the flu shot, studies show that people are far less likely to be hospitalized or die when they’re vaccinated.
Another unexpected benefit of dropping the Yamagata strain from the seasonal vaccine? This will possibly make production of the flu vaccine faster, and enable manufacturers to make more vaccines, helping countries who have a flu vaccine shortage and potentially saving millions more lives.
After his grandmother’s dementia diagnosis, one man invented a snack to keep her healthy and hydrated.
Founder Lewis Hornby and his grandmother Pat, sampling Jelly Drops—an edible gummy containing water and life-saving electrolytes.
On a visit to his grandmother’s nursing home in 2016, college student Lewis Hornby made a shocking discovery: Dehydration is a common (and dangerous) problem among seniors—especially those that are diagnosed with dementia.
Hornby’s grandmother, Pat, had always had difficulty keeping up her water intake as she got older, a common issue with seniors. As we age, our body composition changes, and we naturally hold less water than younger adults or children, so it’s easier to become dehydrated quickly if those fluids aren’t replenished. What’s more, our thirst signals diminish naturally as we age as well—meaning our body is not as good as it once was in letting us know that we need to rehydrate. This often creates a perfect storm that commonly leads to dehydration. In Pat’s case, her dehydration was so severe she nearly died.
When Lewis Hornby visited his grandmother at her nursing home afterward, he learned that dehydration especially affects people with dementia, as they often don’t feel thirst cues at all, or may not recognize how to use cups correctly. But while dementia patients often don’t remember to drink water, it seemed to Hornby that they had less problem remembering to eat, particularly candy.

Hornby wanted to create a solution for elderly people who struggled keeping their fluid intake up. He spent the next eighteen months researching and designing a solution and securing funding for his project. In 2019, Hornby won a sizable grant from the Alzheimer’s Society, a UK-based care and research charity for people with dementia and their caregivers. Together, through the charity’s Accelerator Program, they created a bite-sized, sugar-free, edible jelly drop that looked and tasted like candy. The candy, called Jelly Drops, contained 95% water and electrolytes—important minerals that are often lost during dehydration. The final product launched in 2020—and was an immediate success. The drops were able to provide extra hydration to the elderly, as well as help keep dementia patients safe, since dehydration commonly leads to confusion, hospitalization, and sometimes even death.
Not only did Jelly Drops quickly become a favorite snack among dementia patients in the UK, but they were able to provide an additional boost of hydration to hospital workers during the pandemic. In NHS coronavirus hospital wards, patients infected with the virus were regularly given Jelly Drops to keep their fluid levels normal—and staff members snacked on them as well, since long shifts and personal protective equipment (PPE) they were required to wear often left them feeling parched.
In April 2022, Jelly Drops launched in the United States. The company continues to donate 1% of its profits to help fund Alzheimer’s research.


