Researchers advance drugs that treat pain without addiction
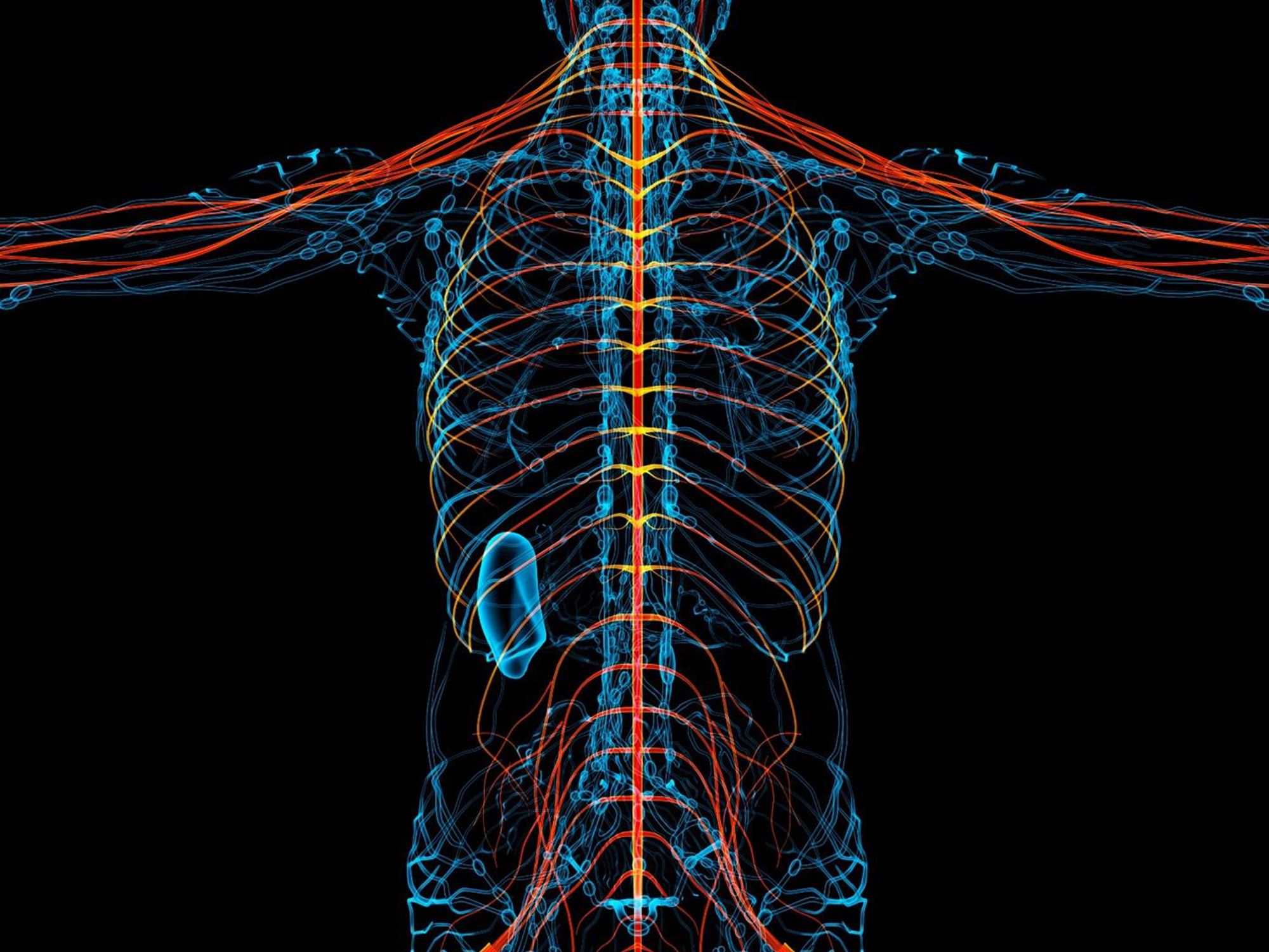
New therapies are using creative approaches that target the body’s sensory neurons, which send pain signals to the brain.
Opioids are one of the most common ways to treat pain. They can be effective but are also highly addictive, an issue that has fueled the ongoing opioid crisis. In 2020, an estimated 2.3 million Americans were dependent on prescription opioids.
Opioids bind to receptors at the end of nerve cells in the brain and body to prevent pain signals. In the process, they trigger endorphins, so the brain constantly craves more. There is a huge risk of addiction in patients using opioids for chronic long-term pain. Even patients using the drugs for acute short-term pain can become dependent on them.
Scientists have been looking for non-addictive drugs to target pain for over 30 years, but their attempts have been largely ineffective. “We desperately need alternatives for pain management,” says Stephen E. Nadeau, a professor of neurology at the University of Florida.
A “dimmer switch” for pain
Paul Blum is a professor of biological sciences at the University of Nebraska. He and his team at Neurocarrus have created a drug called N-001 for acute short-term pain. N-001 is made up of specially engineered bacterial proteins that target the body’s sensory neurons, which send pain signals to the brain. The proteins in N-001 turn down pain signals, but they’re too large to cross the blood-brain barrier, so they don’t trigger the release of endorphins. There is no chance of addiction.
When sensory neurons detect pain, they become overactive and send pain signals to the brain. “We wanted a way to tone down sensory neurons but not turn them off completely,” Blum reveals. The proteins in N-001 act “like a dimmer switch, and that's key because pain is sensation overstimulated.”
Blum spent six years developing the drug. He finally managed to identify two proteins that form what’s called a C2C complex that changes the structure of a subunit of axons, the parts of neurons that transmit electrical signals of pain. Changing the structure reduces pain signaling.
“It will be a long path to get to a successful clinical trial in humans," says Stephen E. Nadeau, professor of neurology at the University of Florida. "But it presents a very novel approach to pain reduction.”
Blum is currently focusing on pain after knee and ankle surgery. Typically, patients are treated with anesthetics for a short time after surgery. But anesthetics usually only last for 4 to 6 hours, and long-term use is toxic. For some, the pain subsides. Others continue to suffer after the anesthetics have worn off and start taking opioids.
N-001 numbs sensation. It lasts for up to 7 days, much longer than any anesthetic. “Our goal is to prolong the time before patients have to start opioids,” Blum says. “The hope is that they can switch from an anesthetic to our drug and thereby decrease the likelihood they're going to take the opioid in the first place.”
Their latest animal trial showed promising results. In mice, N-001 reduced pain-like behaviour by 90 percent compared to the control group. One dose became effective in two hours and lasted a week. A high dose had pain-relieving effects similar to an opioid.
Professor Stephen P. Cohen, director of pain operations at John Hopkins, believes the Neurocarrus approach has potential but highlights the need to go beyond animal testing. “While I think it's promising, it's an uphill battle,” he says. “They have shown some efficacy comparable to opioids, but animal studies don't translate well to people.”
Nadeau, the University of Florida neurologist, agrees. “It will be a long path to get to a successful clinical trial in humans. But it presents a very novel approach to pain reduction.”
Blum is now awaiting approval for phase I clinical trials for acute pain. He also hopes to start testing the drug's effect on chronic pain.
Learning from people who feel no pain
Like Blum, a pharmaceutical company called Vertex is focusing on treating acute pain after surgery. But they’re doing this in a different way, by targeting a sodium channel that plays a critical role in transmitting pain signals.
In 2004, Stephen Waxman, a neurology professor at Yale, led a search for genetic pain anomalies and found that biologically related people who felt no pain despite fractures, burns and even childbirth had mutations in the Nav1.7 sodium channel. Further studies in other families who experienced no pain showed similar mutations in the Nav1.8 sodium channel.
Scientists set out to modify these channels. Many unsuccessful efforts followed, but Vertex has now developed VX-548, a medicine to inhibit Nav1.8. Typically, sodium ions flow through sodium channels to generate rapid changes in voltage which create electrical pulses. When pain is detected, these pulses in the Nav1.8 channel transmit pain signals. VX-548 uses small molecules to inhibit the channel from opening. This blocks the flow of sodium ions and the pain signal. Because Nav1.8 operates only in peripheral nerves, located outside the brain, VX-548 can relieve pain without any risk of addiction.
"Frankly we need drugs for chronic pain more than acute pain," says Waxman.
The team just finished phase II clinical trials for patients following abdominoplasty surgery and bunionectomy surgery.
After abdominoplasty surgery, 76 patients were treated with a high dose of VX-548. Researchers then measured its effectiveness in reducing pain over 48 hours, using the SPID48 scale, in which higher scores are desirable. The score for Vertex’s drug was 110.5 compared to 72.7 in the placebo group, whereas the score for patients taking an opioid was 85.2. The study involving bunionectomy surgery showed positive results as well.
Waxman, who has been at the forefront of studies into Nav1.7 and Nav1.8, believes that Vertex's results are promising, though he highlights the need for further clinical trials.
“Blocking Nav1.8 is an attractive target,” he says. “[Vertex is] studying pain that is relatively simple and uniform, and that's key to having a drug trial that is informative. But the study needs to be replicated and frankly we need drugs for chronic pain more than acute pain. If this is borne out by additional studies, it's one important step in a journey.”
Vertex will be launching phase III trials later this year.
Finding just the right amount of Nerve Growth Factor
Whereas Neurocarrus and Vertex are targeting short-term pain, a company called Levicept is concentrating on relieving chronic osteoarthritis pain. Around 32.5 million Americans suffer from osteoarthritis. Patients commonly take NSAIDs, or non-steroidal anti-inflammatory drugs, but they cannot be taken long-term. Some take opioids but they aren't very effective.
Levicept’s drug, Levi-04, is designed to modify a signaling pathway associated with pain. Nerve Growth Factor (NGF) is a neurotrophin: it’s involved in nerve growth and function. NGF signals by attaching to receptors. In pain there are excess neurotrophins attaching to receptors and activating pain signals.
“What Levi-04 does is it returns the natural equilibrium of neurotrophins,” says Simon Westbrook, the CEO and founder of Levicept. It stabilizes excess neurotrophins so that the NGF pathway does not signal pain. Levi-04 isn't addictive since it works within joints and in nerves outside the brain.
Westbrook was initially involved in creating an anti-NGF molecule for Pfizer called Tanezumab. At first, Tanezumab seemed effective in clinical trials and other companies even started developing their own versions. However, a problem emerged. Tanezumab caused rapidly progressive osteoarthritis, or RPOA, in some patients because it completely removed NGF from the system. NGF is not just involved in pain signalling, it’s also involved in bone growth and maintenance.
Levicept has found a way to modify the NGF pathway without completely removing NGF. They have now finished a small-scale phase I trial mainly designed to test safety rather than efficacy. “We demonstrated that Levi-04 is safe and that it bound to its target, NGF,” says Westbrook. It has not caused RPOA.
Professor Philip Conaghan, director of the Leeds Institute of Rheumatic and Musculoskeletal Medicine, believes that Levi-04 has potential but urges the need for caution. “At this early stage of development, their molecule looks promising for osteoarthritis pain,” he says. “They will have to watch out for RPOA which is a potential problem.”
Westbrook starts phase II trials with 500 patients this summer to check for potential side effects and test the drug’s efficacy.
There is a real push to find an effective alternative to opioids. “We have a lot of work to do,” says Professor Waxman. “But I am confident that we will be able to develop new, much more effective pain therapies.”
What to Know about the Fast-Spreading Delta Variant
A highly contagious form of the coronavirus known as the Delta variant is spreading rapidly and becoming increasingly prevalent around the world. First identified in India in December, Delta has now been identified in 111 countries.
In the United States, the variant now accounts for 83% of sequenced COVID-19 cases, said Rochelle Walensky, director of the Centers for Disease Control and Prevention, at a July 20 Senate hearing. In May, Delta was responsible for just 3% of U.S. cases. The World Health Organization projects that Delta will become the dominant variant globally over the coming months.
So, how worried should you be about the Delta variant? We asked experts some common questions about Delta.
What is a variant?
To understand Delta, it's helpful to first understand what a variant is. When a virus infects a person, it gets into your cells and makes a copy of its genome so it can replicate and spread throughout your body.
In the process of making new copies of itself, the virus can make a mistake in its genetic code. Because viruses are replicating all the time, these mistakes — also called mutations — happen pretty often. A new variant emerges when a virus acquires one or more new mutations and starts spreading within a population.
There are thousands of SARS-CoV-2 variants, but most of them don't substantially change the way the virus behaves. The variants that scientists are most interested in are known as variants of concern. These are versions of the virus with mutations that allow the virus to spread more easily, evade vaccines, or cause more severe disease.
"The vast majority of the mutations that have accumulated in SARS-CoV-2 don't change the biology as far as we're concerned," said Jennifer Surtees, a biochemist at the University of Buffalo who's studying the coronavirus. "But there have been a handful of key mutations and combinations of mutations that have led to what we're now calling variants of concern."
One of those variants of concern is Delta, which is now driving many new COVID-19 infections.
Why is the Delta variant so concerning?
"The reason why the Delta variant is concerning is because it's causing an increase in transmission," said Alba Grifoni, an infectious disease researcher at the La Jolla Institute for Immunology. "The virus is spreading faster and people — particularly those who are not vaccinated yet — are more prone to exposure."
The Delta variant has a few key mutations that make it better at attaching to our cells and evading the neutralizing antibodies in our immune system. These mutations have changed the virus enough to make it more than twice as contagious as the original SARS-CoV-2 virus that emerged in Wuhan and about 50% more contagious than the Alpha variant, previously known as B.1.1.7, or the U.K. variant.
These mutations were previously seen in other variants on their own, but it's their combination that makes Delta so much more infectious.
Do vaccines work against the Delta variant?
The good news is, the COVID-19 vaccines made by AstraZeneca, Johnson & Johnson, Moderna, and Pfizer still work against the Delta variant. They remain more than 90% effective at preventing hospitalizations and death due to Delta. While they're slightly less protective against disease symptoms, they're still very effective at preventing severe illness caused by the Delta variant.
"They're not as good as they were against the prior strains, but they're holding up pretty well," said Eric Topol, a physician and director of the Scripps Translational Research Institute, during a July 19 briefing for journalists.
Because Delta is better at evading our immune systems, it's likely causing more breakthrough infections — COVID-19 cases in people who are vaccinated. However, breakthrough infections were expected before the Delta variant became widespread. No vaccine is 100% effective, so breakthrough infections can happen with other vaccines as well. Experts say the COVID-19 vaccines are still working as expected, even if breakthrough infections occur. The majority of these infections are asymptomatic or cause only mild symptoms.
Should vaccinated people worry about the Delta variant?
Vaccines train our immune systems to protect us against infection. They do this by spurring the production of antibodies, which stick around in our bodies to help fight off a particular pathogen in case we ever come into contact with it.
But even if the new Delta variant slips past our neutralizing antibodies, there's another component of our immune system that can help overtake the virus: T cells. Studies are showing that the COVID-19 vaccines also galvanize T cells, which help limit disease severity in people who have been vaccinated.
"While antibodies block the virus and prevent the virus from infecting cells, T cells are able to attack cells that have already been infected," Grifoni said. In other words, T cells can prevent the infection from spreading to more places in the body. A study published July 1 by Grifoni and her colleagues found that T cells were still able to recognize mutated forms of the virus — further evidence that our current vaccines are effective against Delta.
Can fully vaccinated people spread the Delta variant?
Previously, scientists believed it was unlikely for fully vaccinated individuals with asymptomatic infections to spread Covid-19. But the Delta variant causes the virus to make so many more copies of itself inside the body, and high viral loads have been found in the respiratory tracts of people who are fully vaccinated. This suggests that vaccinated people may be able to spread the Delta variant to some degree.
If you have COVID-19 symptoms, even if you're fully vaccinated, you should get tested and isolate from friends and family because you could spread the virus.
What risk does Delta pose to unvaccinated people?
The Delta variant is behind a surge in cases in communities with low vaccination rates, and unvaccinated Americans currently account for 97% of hospitalizations due to COVID-19, according to Walensky. The best thing you can do right now to prevent yourself from getting sick is to get vaccinated.
Gigi Gronvall, an immunologist and senior scholar at the Johns Hopkins Center for Health Security, said in this week's "Making Sense of Science" podcast that it's especially important to get all required doses of the vaccine in order to have the best protection against the Delta variant. "Even if it's been more than the allotted time that you were told to come back and get the second, there's no time like the present," she said.
With more than 3.6 billion COVID-19 doses administered globally, the vaccines have been shown to be incredibly safe. Serious adverse effects are rare, although scientists continue to monitor for them.
Being vaccinated also helps prevent the emergence of new and potentially more dangerous variants. Viruses need to infect people in order to replicate, and variants emerge because the virus continues to infect more people. More infections create more opportunities for the virus to acquire new mutations.
Surtees and others worry about a scenario in which a new variant emerges that's even more transmissible or resistant to vaccines. "This is our window of opportunity to try to get as many people vaccinated as possible and get people protected so that so that the virus doesn't evolve to be even better at infecting people," she said.
Does Delta cause more severe disease?
While hospitalizations and deaths from COVID-19 are increasing again, it's not yet clear whether Delta causes more severe illness than previous strains.
How can we protect unvaccinated children from the Delta variant?
With children 12 and under not yet eligible for the COVID-19 vaccine, kids are especially vulnerable to the Delta variant. One way to protect unvaccinated children is for parents and other close family members to get vaccinated.
It's also a good idea to keep masks handy when going out in public places. Due to risk Delta poses, the American Academy of Pediatrics issued new guidelines July 19 recommending that all staff and students over age 2 wear face masks in school this fall, even if they have been vaccinated.
Parents should also avoid taking their unvaccinated children to crowded, indoor locations and make sure their kids are practicing good hand-washing hygiene. For children younger than 2, limit visits with friends and family members who are unvaccinated or whose vaccination status is unknown and keep up social distancing practices while in public.
While there's no evidence yet that Delta increases disease severity in children, parents should be mindful that in some rare cases, kids can get a severe form of the disease.
"We're seeing more children getting sick and we're seeing some of them get very sick," Surtees said. "Those children can then pass on the virus to other individuals, including people who are immunocompromised or unvaccinated."
New Podcast: Dr. Gigi Gronvall Addresses the Risk of Delta and COVID-19’s Possible Origins
"Making Sense of Science" is a monthly podcast that features interviews with leading medical and scientific experts about the latest developments and the big ethical and societal questions they raise. This episode is hosted by science and biotech journalist Emily Mullin, summer editor of the award-winning science outlet Leaps.org.

