Eight Big Medical and Science Trends to Watch in 2021

Promising developments underway include advancements in gene and cell therapy, better testing for COVID, and a renewed focus on climate change.
The world as we know it has forever changed. With a greater focus on science and technology than before, experts in the biotech and life sciences spaces are grappling with what comes next as SARS-CoV-2, the coronavirus that causes the COVID-19 illness, has spread and mutated across the world.
Even with vaccines being distributed, so much still remains unknown.
Jared Auclair, Technical Supervisor for the Northeastern University's Life Science Testing Center in Burlington, Massachusetts, guides a COVID testing lab that cranks out thousands of coronavirus test results per day. His lab is also focused on monitoring the quality of new cell and gene therapy products coming to the market.
Here are trends Auclair and other experts are watching in 2021.
Better Diagnostic Testing for COVID
Expect improvements in COVID diagnostic testing and the ability to test at home.
There are currently three types of coronavirus tests. The molecular test—also known as the RT-PCR test, detects the virus's genetic material, and is highly accurate, but it can take days to receive results. There are also antibody tests, done through a blood draw, designed to test whether you've had COVID in the past. Finally, there's the quick antigen test that isn't as accurate as the PCR test, but can identify if people are going to infect others.
Last month, Lucira Health secured the U.S. FDA Emergency Use Authorization for the first prescription molecular diagnostic test for COVID-19 that can be performed at home. On December 15th, the Ellume Covid-19 Home Test received authorization as the first over-the-counter COVID-19 diagnostic antigen test that can be done at home without a prescription. The test uses a nasal swab that is connected to a smartphone app and returns results in 15-20 minutes. Similarly, the BinaxNOW COVID-19 Ag Card Home Test received authorization on Dec. 16 for its 15-minute antigen test that can be used within the first seven days of onset of COIVD-19 symptoms.
Home testing has the possibility to impact the pandemic pretty drastically, Auclair says, but there are other considerations: the type and timing of test that is administered, how expensive is the test (and if it is financially feasible for the general public) and the ability of a home test taker to accurately administer the test.
"The vaccine roll-out will not eliminate the need for testing until late 2021 or early 2022."
Ideally, everyone would frequently get tested, but that would mean the cost of a single home test—which is expected to be around $30 or more—would need to be much cheaper, more in the $5 range.
Auclair expects "innovations in the diagnostic space to explode" with the need for more accurate, inexpensive, quicker COVID tests. Auclair foresees innovations to be at first focused on COVID point-of-care testing, but he expects improvements within diagnostic testing for other types of viruses and diseases too.
"We still need more testing to get the pandemic under control, likely over the next 12 months," Auclair says. "The vaccine roll-out will not eliminate the need for testing until late 2021 or early 2022."
Rise of mRNA-based Vaccines and Therapies
A year ago, vaccines weren't being talked about like they are today.
"But clearly vaccines are the talk of the town," Auclair says. "The reason we got a vaccine so fast was there was so much money thrown at it."
A vaccine can take more than 10 years to fully develop, according to the World Economic Forum. Prior to the new COVID vaccines, which were remarkably developed and tested in under a year, the fastest vaccine ever made was for mumps -- and it took four years.
"Normally you have to produce a protein. This is typically done in eggs. It takes forever," says Catherine Dulac, a neuroscientist and developmental biologist at Harvard University who won the 2021 Breakthrough Prize in Life Sciences. "But an mRNA vaccine just enabled [us] to skip all sorts of steps [compared with burdensome conventional manufacturing] and go directly to a product that can be injected into people."
Non-traditional medicines based on genetic research are in their infancy. With mRNA-based vaccines hitting the market for the first time, look for more vaccines to be developed for whatever viruses we don't currently have vaccines for, like dengue virus and Ebola, Auclair says.
"There's a whole bunch of things that could be explored now that haven't been thought about in the past," Auclair says. "It could really be a game changer."
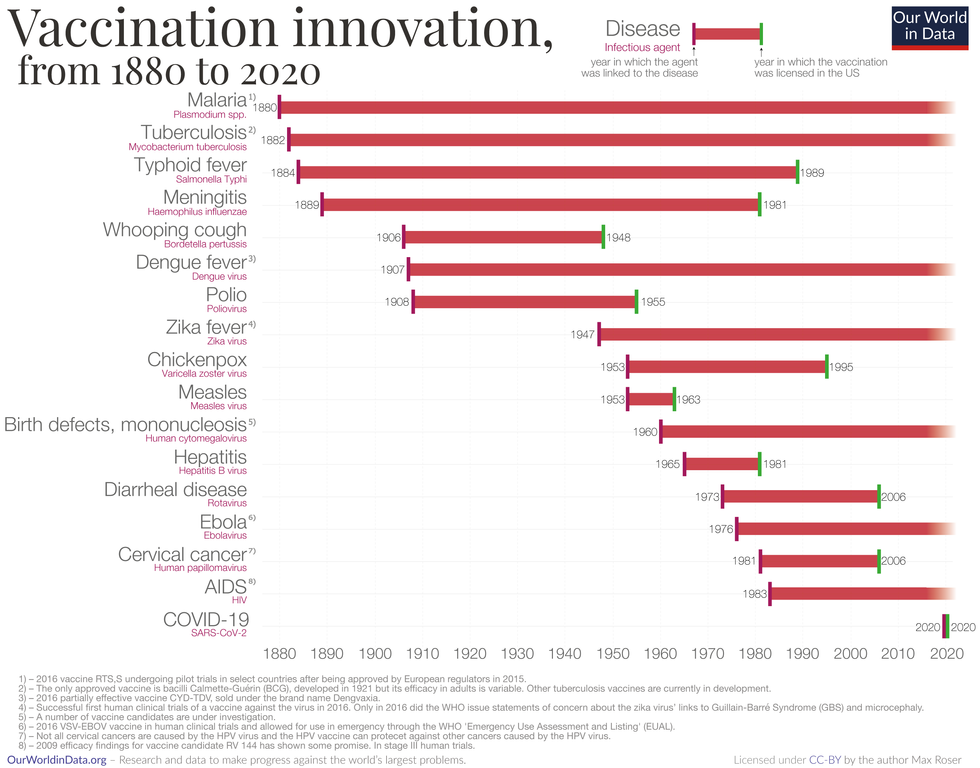
Vaccine Innovation over the last 140 years.
Max Roser/Our World in Data (Creative Commons license)
Advancements in Cell and Gene Therapies
CRISPR, a type of gene editing, is going to be huge in 2021, especially after the Nobel Prize in Chemistry was awarded to Emmanuelle Charpentier and Jennifer Doudna in October for pioneering the technology.
Right now, CRISPR isn't completely precise and can cause deletions or rearrangements of DNA.
"It's definitely not there yet, but over the next year it's going to get a lot closer and you're going to have a lot of momentum in this space," Auclair says. "CRISPR is one of the technologies I'm most excited about and 2021 is the year for it."
Gene therapies are typically used on rare genetic diseases. They work by replacing the faulty dysfunctional genes with corrected DNA codes.
"Cell and gene therapies are really where the field is going," Auclair says. "There is so much opportunity....For the first time in our life, in our existence as a species, we may actually be able to cure disease by using [techniques] like gene editing, where you cut in and out of pieces of DNA that caused a disease and put in healthy DNA," Auclair says.
For example, Spinal Muscular Atrophy is a rare genetic disorder that leads to muscle weakness, paralysis and death in children by age two. As of last year, afflicted children can take a gene therapy drug called Zolgensma that targets the missing or nonworking SMN1 gene with a new copy.
Another recent breakthrough uses gene editing for sickle cell disease. Victoria Gray, a mom from Mississippi who was exclusively followed by NPR, was the first person in the United States to be successfully treated for the genetic disorder with the help of CRISPR. She has continued to improve since her landmark treatment on July 2, 2019 and her once-debilitating pain has greatly eased.
"This is really a life-changer for me," she told NPR. "It's magnificent."
"You are going to see bigger leaps in gene therapies."
Look out also for improvements in cell therapies, but on a much lesser scale.
Cell therapies remove immune cells from a person or use cells from a donor. The cells are modified or cultured in lab, multiplied by the millions and then injected back into patients. These include stem cell therapies as well as CAR-T cell therapies, which are typically therapies of last resort and used in cancers like leukemia, Auclair says.
"You are going to see bigger leaps in gene therapies," Auclair says. "It's being heavily researched and we understand more about how to do gene therapies. Cell therapies will lie behind it a bit because they are so much more difficult to work with right now."
More Monoclonal Antibody Therapies
Look for more customized drugs to personalize medicine even more in the biotechnology space.
In 2019, the FDA anticipated receiving more than 200 Investigational New Drug (IND) applications in 2020. But with COVID, the number of INDs skyrocketed to 6,954 applications for the 2020 fiscal year, which ended September 30, 2020, according to the FDA's online tracker. Look for antibody therapies to play a bigger role.
Monoclonal antibodies are lab-grown proteins that mimic or enhance the immune system's response to fight off pathogens, like viruses, and they've been used to treat cancer. Now they are being used to treat patients with COVID-19.
President Donald Trump received a monoclonal antibody cocktail, called REGEN-COV2, which later received FDA emergency use authorization.
A newer type of monoclonal antibody therapy is Antibody-Drug Conjugates, also called ADCs. It's something we're going to be hearing a lot about in 2021, Auclair says.
"Antibody-Drug Conjugates is a monoclonal antibody with a chemical, we consider it a chemical warhead on it," Auclair says. "The monoclonal antibody binds to a specific antigen in your body or protein and delivers a chemical to that location and kills the infected cell."
Moving Beyond Male-Centric Lab Testing
Scientific testing for biology has, until recently, focused on testing males. Dulac, a Howard Hughes Medical Investigator and professor of molecular and cellular biology at Harvard University, challenged that idea to find brain circuitry behind sex-specific behaviors.
"For the longest time, until now, all the model systems in biology, are male," Dulac says. "The idea is if you do testing on males, you don't need to do testing on females."
Clinical models are done in male animals, as well as fundamental research. Because biological research is always done on male models, Dulac says the outcomes and understanding in biology is geared towards understanding male biology.
"All the drugs currently on the market and diagnoses of diseases are biased towards the understanding of male biology," Dulac says. "The diagnostics of diseases is way weaker in women than men."
That means the treatment isn't necessarily as good for women as men, she says, including what is known and understood about pain medication.
"So pain medication doesn't work well in women," Dulac says. "It works way better in men. It's true for almost all diseases that I know. Why? because you have a science that is dominated by males."
Although some in the scientific community challenge that females are not interesting or too complicated with their hormonal variations, Dulac says that's simply not true.
"There's absolutely no reason to decide 50% of life forms are interesting and the other 50% are not interesting. What about looking at both?" says Dulac, who was awarded the $3 million Breakthrough Prize in Life Sciences in September for connecting specific neural mechanisms to male and female parenting behaviors.
Disease Research on Single Cells
To better understand how diseases manifest in the body's cell and tissues, many researchers are looking at single-cell biology. Cells are the most fundamental building blocks of life. Much still needs to be learned.
"A remarkable development this year is the massive use of analysis of gene expression and chromosomal regulation at the single-cell level," Dulac says.
Much is focused on the Human Cell Atlas (HCA), a global initiative to map all cells in healthy humans and to better identify which genes associated with diseases are active in a person's body. Most estimates put the number of cells around 30 trillion.
Dulac points to work being conducted by the Cell Census Network (BICCN) Brain Initiative, an initiative by the National Institutes of Health to come up with an atlas of cell types in mouse, human and non-human primate brains, and the Chan Zuckerberg Initiative's funding of single-cell biology projects, including those focused on single-cell analysis of inflammation.
"Our body and our brain are made of a large number of cell types," Dulac says. "The ability to explore and identify differences in gene expression and regulation in massively multiplex ways by analyzing millions of cells is extraordinarily important."
Converting Plastics into Food
Yep, you heard it right, plastics may eventually be turned into food. The Defense Advanced Research Projects Agency, better known as DARPA, is funding a project—formally titled "Production of Macronutrients from Thermally Oxo-Degraded Wastes"—and asking researchers how to do this.
"When I first heard about this challenge, I thought it was absolutely absurd," says Dr. Robert Brown, director of the Bioeconomy Institute at Iowa State University and the project's principal investigator, who is working with other research partners at the University of Delaware, Sandia National Laboratories, and the American Institute of Chemical Engineering (AIChE)/RAPID Institute.
But then Brown realized plastics will slowly start oxidizing—taking in oxygen—and microorganisms can then consume it. The oxidation process at room temperature is extremely slow, however, which makes plastics essentially not biodegradable, Brown says.
That changes when heat is applied at brick pizza oven-like temperatures around 900-degrees Fahrenheit. The high temperatures get compounds to oxidize rapidly. Plastics are synthetic polymers made from petroleum—large molecules formed by linking many molecules together in a chain. Heated, these polymers will melt and crack into smaller molecules, causing them to vaporize in a process called devolatilization. Air is then used to cause oxidation in plastics and produce oxygenated compounds—fatty acids and alcohols—that microorganisms will eat and grow into single-cell proteins that can be used as an ingredient or substitute in protein-rich foods.
"The caveat is the microorganisms must be food-safe, something that we can consume," Brown says. "Like supplemental or nutritional yeast, like we use to brew beer and to make bread or is used in Australia to make Vegemite."
What do the microorganisms look like? For any home beer brewers, it's the "gunky looking stuff you'd find at the bottom after the fermentation process," Brown says. "That's cellular biomass. Like corn grown in the field, yeast or other microorganisms like bacteria can be harvested as macro-nutrients."
Brown says DARPA's ReSource program has challenged all the project researchers to find ways for microorganisms to consume any plastics found in the waste stream coming out of a military expeditionary force, including all the packaging of food and supplies. Then the researchers aim to remake the plastic waste into products soldiers can use, including food. The project is in the first of three phases.
"We are talking about polyethylene, polypropylene, like PET plastics used in water bottles and converting that into macronutrients that are food," says Brown.
Renewed Focus on Climate Change
The Union of Concerned Scientists say carbon dioxide levels are higher today than any point in at least 800,000 years.
"Climate science is so important for all of humankind. It is critical because the quality of life of humans on the planet depends on it."
Look for technology to help locate large-scale emitters of carbon dioxide, including sensors on satellites and artificial intelligence to optimize energy usage, especially in data centers.
Other technologies focus on alleviating the root cause of climate change: emissions of heat-trapping gasses that mainly come from burning fossil fuels.
Direct air carbon capture, an emerging effort to capture carbon dioxide directly from ambient air, could play a role.
The technology is in the early stages of development and still highly uncertain, says Peter Frumhoff, director of science and policy at Union of Concerned Scientists. "There are a lot of questions about how to do that at sufficiently low costs...and how to scale it up so you can get carbon dioxide stored in the right way," he says, and it can be very energy intensive.
One of the oldest solutions is planting new forests, or restoring old ones, which can help convert carbon dioxide into oxygen through photosynthesis. Hence the Trillion Trees Initiative launched by the World Economic Forum. Trees are only part of the solution, because planting trees isn't enough on its own, Frumhoff says. That's especially true, since 2020 was the year that human-made, artificial stuff now outweighs all life on earth.
More research is also going into artificial photosynthesis for solar fuels. The U.S. Department of Energy awarded $100 million in 2020 to two entities that are conducting research. Look also for improvements in battery storage capacity to help electric vehicles, as well as back-up power sources for solar and wind power, Frumhoff says.
Another method to combat climate change is solar geoengineering, also called solar radiation management, which reflects sunlight back to space. The idea stems from a volcanic eruption in 1991 that released a tremendous amount of sulfate aerosol particles into the stratosphere, reflecting the sunlight away from Earth. The planet cooled by a half degree for nearly a year, Frumhoff says. However, he acknowledges, "there's a lot of things we don't know about the potential impacts and risks" involved in this controversial approach.
Whatever the approach, scientific solutions to climate change are attracting renewed attention. Under President Trump, the White House Office of Science and Technology Policy didn't have an acting director for almost two years. Expect that to change when President-elect Joe Biden takes office.
"Climate science is so important for all of humankind," Dulac says. "It is critical because the quality of life of humans on the planet depends on it."
Autonomous, indoor farming gives a boost to crops
Artificial Intelligence is already helping to grow some of the food we eat.
The glass-encased cabinet looks like a display meant to hold reasonably priced watches, or drugstore beauty creams shipped from France. But instead of this stagnant merchandise, each of its five shelves is overgrown with leaves — moss-soft pea sprouts, spikes of Lolla rosa lettuces, pale bok choy, dark kale, purple basil or red-veined sorrel or green wisps of dill. The glass structure isn’t a cabinet, but rather a “micro farm.”
The gadget is on display at the Richmond, Virginia headquarters of Babylon Micro-Farms, a company that aims to make indoor farming in the U.S. more accessible and sustainable. Babylon’s soilless hydroponic growing system, which feeds plants via nutrient-enriched water, allows chefs on cruise ships, cafeterias and elsewhere to provide home-grown produce to patrons, just seconds after it’s harvested. Currently, there are over 200 functioning systems, either sold or leased to customers, and more of them are on the way.
The chef-farmers choose from among 45 types of herb and leafy-greens seeds, plop them into grow trays, and a few weeks later they pick and serve. While success is predicated on at least a small amount of these humans’ care, the systems are autonomously surveilled round-the-clock from Babylon’s base of operations. And artificial intelligence is helping to run the show.
Babylon piloted the use of specialized cameras that take pictures in different spectrums to gather some less-obvious visual data about plants’ wellbeing and alert people if something seems off.
Imagine consistently perfect greens and tomatoes and strawberries, grown hyper-locally, using less water, without chemicals or environmental contaminants. This is the hefty promise of controlled environment agriculture (CEA) — basically, indoor farms that can be hydroponic, aeroponic (plant roots are suspended and fed through misting), or aquaponic (where fish play a role in fertilizing vegetables). But whether they grow 4,160 leafy-green servings per year, like one Babylon farm, or millions of servings, like some of the large, centralized facilities starting to supply supermarkets across the U.S., they seek to minimize failure as much as possible.

Babylon’s soilless hydroponic growing system
Courtesy Babylon Micro-Farms
Here, AI is starting to play a pivotal role. CEA growers use it to help “make sense of what’s happening” to the plants in their care, says Scott Lowman, vice president of applied research at the Institute for Advanced Learning and Research (IALR) in Virginia, a state that’s investing heavily in CEA companies. And although these companies say they’re not aiming for a future with zero human employees, AI is certainly poised to take a lot of human farming intervention out of the equation — for better and worse.
Most of these companies are compiling their own data sets to identify anything that might block the success of their systems. Babylon had already integrated sensor data into its farms to measure heat and humidity, the nutrient content of water, and the amount of light plants receive. Last year, they got a National Science Foundation grant that allowed them to pilot the use of specialized cameras that take pictures in different spectrums to gather some less-obvious visual data about plants’ wellbeing and alert people if something seems off. “Will this plant be healthy tomorrow? Are there things…that the human eye can't see that the plant starts expressing?” says Amandeep Ratte, the company’s head of data science. “If our system can say, Hey, this plant is unhealthy, we can reach out to [users] preemptively about what they’re doing wrong, or is there a disease at the farm?” Ratte says. The earlier the better, to avoid crop failures.

Natural light accounts for 70 percent of Greenswell Growers’ energy use on a sunny day.
Courtesy Greenswell Growers
IALR’s Lowman says that other CEA companies are developing their AI systems to account for the different crops they grow — lettuces come in all shapes and sizes, after all, and each has different growing needs than, for example, tomatoes. The ways they run their operations differs also. Babylon is unusual in its decentralized structure. But centralized growing systems with one main location have variabilities, too. AeroFarms, which recently declared bankruptcy but will continue to run its 140,000-square foot vertical operation in Danville, Virginia, is entirely enclosed and reliant on the intense violet glow of grow lights to produce microgreens.
Different companies have different data needs. What data is essential to AeroFarms isn’t quite the same as for Greenswell Growers located in Goochland County, Virginia. Raising four kinds of lettuce in a 77,000-square-foot automated hydroponic greenhouse, the vagaries of naturally available light, which accounts for 70 percent of Greenswell’s energy use on a sunny day, affect operations. Their tech needs to account for “outside weather impacts,” says president Carl Gupton. “What adjustments do we have to make inside of the greenhouse to offset what's going on outside environmentally, to give that plant optimal conditions? When it's 85 percent humidity outside, the system needs to do X, Y and Z to get the conditions that we want inside.”
AI will help identify diseases, as well as when a plant is thirsty or overly hydrated, when it needs more or less calcium, phosphorous, nitrogen.
Nevertheless, every CEA system has the same core needs — consistent yield of high quality crops to keep up year-round supply to customers. Additionally, “Everybody’s got the same set of problems,” Gupton says. Pests may come into a facility with seeds. A disease called pythium, one of the most common in CEA, can damage plant roots. “Then you have root disease pressures that can also come internally — a change in [growing] substrate can change the way the plant performs,” Gupton says.
AI will help identify diseases, as well as when a plant is thirsty or overly hydrated, when it needs more or less calcium, phosphorous, nitrogen. So, while companies amass their own hyper-specific data sets, Lowman foresees a time within the next decade “when there will be some type of [open-source] database that has the most common types of plant stress identified” that growers will be able to tap into. Such databases will “create a community and move the science forward,” says Lowman.
In fact, IALR is working on assembling images for just such a database now. On so-called “smart tables” inside an Institute lab, a team is growing greens and subjects them to various stressors. Then, they’re administering treatments while taking images of every plant every 15 minutes, says Lowman. Some experiments generate 80,000 images; the challenge lies in analyzing and annotating the vast trove of them, marking each one to reflect outcome—for example increasing the phosphate delivery and the plant’s response to it. Eventually, they’ll be fed into AI systems to help them learn.
For all the enthusiasm surrounding this technology, it’s not without downsides. Training just one AI system can emit over 250,000 pounds of carbon dioxide, according to MIT Technology Review. AI could also be used “to enhance environmental benefit for CEA and optimize [its] energy consumption,” says Rozita Dara, a computer science professor at the University of Guelph in Canada, specializing in AI and data governance, “but we first need to collect data to measure [it].”

The chef-farmers can choose from 45 types of herb and leafy-greens seeds.
Courtesy Babylon Micro-Farms
Any system connected to the Internet of Things is also vulnerable to hacking; if CEA grows to the point where “there are many of these similar farms, and you're depending on feeding a population based on those, it would be quite scary,” Dara says. And there are privacy concerns, too, in systems where imaging is happening constantly. It’s partly for this reason, says Babylon’s Ratte, that the company’s in-farm cameras all “face down into the trays, so the only thing [visible] is pictures of plants.”
Tweaks to improve AI for CEA are happening all the time. Greenswell made its first harvest in 2022 and now has annual data points they can use to start making more intelligent choices about how to feed, water, and supply light to plants, says Gupton. Ratte says he’s confident Babylon’s system can already “get our customers reliable harvests. But in terms of how far we have to go, it's a different problem,” he says. For example, if AI could detect whether the farm is mostly empty—meaning the farm’s user hasn’t planted a new crop of greens—it can alert Babylon to check “what's going on with engagement with this user?” Ratte says. “Do they need more training? Did the main person responsible for the farm quit?”
Lowman says more automation is coming, offering greater ability for systems to identify problems and mitigate them on the spot. “We still have to develop datasets that are specific, so you can have a very clear control plan, [because] artificial intelligence is only as smart as what we tell it, and in plant science, there's so much variation,” he says. He believes AI’s next level will be “looking at those first early days of plant growth: when the seed germinates, how fast it germinates, what it looks like when it germinates.” Imaging all that and pairing it with AI, “can be a really powerful tool, for sure.”
Scientists make progress with growing organs for transplants
Researchers from the University of Cambridge have laid the foundations for growing synthetic embryos that could develop a beating heart, gut and brain.
Story by Big Think
For over a century, scientists have dreamed of growing human organs sans humans. This technology could put an end to the scarcity of organs for transplants. But that’s just the tip of the iceberg. The capability to grow fully functional organs would revolutionize research. For example, scientists could observe mysterious biological processes, such as how human cells and organs develop a disease and respond (or fail to respond) to medication without involving human subjects.
Recently, a team of researchers from the University of Cambridge has laid the foundations not just for growing functional organs but functional synthetic embryos capable of developing a beating heart, gut, and brain. Their report was published in Nature.
The organoid revolution
In 1981, scientists discovered how to keep stem cells alive. This was a significant breakthrough, as stem cells have notoriously rigorous demands. Nevertheless, stem cells remained a relatively niche research area, mainly because scientists didn’t know how to convince the cells to turn into other cells.
Then, in 1987, scientists embedded isolated stem cells in a gelatinous protein mixture called Matrigel, which simulated the three-dimensional environment of animal tissue. The cells thrived, but they also did something remarkable: they created breast tissue capable of producing milk proteins. This was the first organoid — a clump of cells that behave and function like a real organ. The organoid revolution had begun, and it all started with a boob in Jello.
For the next 20 years, it was rare to find a scientist who identified as an “organoid researcher,” but there were many “stem cell researchers” who wanted to figure out how to turn stem cells into other cells. Eventually, they discovered the signals (called growth factors) that stem cells require to differentiate into other types of cells.
For a human embryo (and its organs) to develop successfully, there needs to be a “dialogue” between these three types of stem cells.
By the end of the 2000s, researchers began combining stem cells, Matrigel, and the newly characterized growth factors to create dozens of organoids, from liver organoids capable of producing the bile salts necessary for digesting fat to brain organoids with components that resemble eyes, the spinal cord, and arguably, the beginnings of sentience.
Synthetic embryos
Organoids possess an intrinsic flaw: they are organ-like. They share some characteristics with real organs, making them powerful tools for research. However, no one has found a way to create an organoid with all the characteristics and functions of a real organ. But Magdalena Żernicka-Goetz, a developmental biologist, might have set the foundation for that discovery.
Żernicka-Goetz hypothesized that organoids fail to develop into fully functional organs because organs develop as a collective. Organoid research often uses embryonic stem cells, which are the cells from which the developing organism is created. However, there are two other types of stem cells in an early embryo: stem cells that become the placenta and those that become the yolk sac (where the embryo grows and gets its nutrients in early development). For a human embryo (and its organs) to develop successfully, there needs to be a “dialogue” between these three types of stem cells. In other words, Żernicka-Goetz suspected the best way to grow a functional organoid was to produce a synthetic embryoid.
As described in the aforementioned Nature paper, Żernicka-Goetz and her team mimicked the embryonic environment by mixing these three types of stem cells from mice. Amazingly, the stem cells self-organized into structures and progressed through the successive developmental stages until they had beating hearts and the foundations of the brain.
“Our mouse embryo model not only develops a brain, but also a beating heart [and] all the components that go on to make up the body,” said Żernicka-Goetz. “It’s just unbelievable that we’ve got this far. This has been the dream of our community for years and major focus of our work for a decade and finally we’ve done it.”
If the methods developed by Żernicka-Goetz’s team are successful with human stem cells, scientists someday could use them to guide the development of synthetic organs for patients awaiting transplants. It also opens the door to studying how embryos develop during pregnancy.
This article originally appeared on Big Think, home of the brightest minds and biggest ideas of all time.


