Elizabeth Holmes Through the Director’s Lens
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.

Elizabeth Holmes.
"The Inventor," a chronicle of Theranos's storied downfall, premiered recently on HBO. Leapsmag reached out to director Alex Gibney, whom The New York Times has called "one of America's most successful and prolific documentary filmmakers," for his perspective on Elizabeth Holmes and the world she inhabited.
Do you think Elizabeth Holmes was a charismatic sociopath from the start — or is she someone who had good intentions, over-promised, and began the lies to keep her business afloat, a "fake it till you make it" entrepreneur like Thomas Edison?
I'm not qualified to say if EH was or is a sociopath. I don't think she started Theranos as a scam whose only purpose was to make money. If she had done so, she surely would have taken more money for herself along the way. I do think that she had good intentions and that she, as you say, "began the lies to keep her business afloat." ([Reporter John] Carreyrou's book points out that those lies began early.) I think that the Edison comparison is instructive for a lot of reasons.
First, Edison was the original "fake-it-till-you-make-it" entrepreneur. That puts this kind of behavior in the mainstream of American business. By saying that, I am NOT endorsing the ethic, just the opposite. As one Enron executive mused about the mendacity there, "Was it fraud or was it bad marketing?" That gives you a sense of how baked-in the "fake it" sensibility is.
"Having a thirst for fame and a noble cause enabled her to think it was OK to lie in service of those goals."
I think EH shares one other thing with Edison, which is a huge ego coupled with a talent for storytelling as long as she is the heroic, larger-than-life main character. It's interesting that EH calls her initial device "Edison." Edison was the world's most famous "inventor," both because of the devices that came out of his shop and and for his ability for "self-invention." As Randall Stross notes in "The Wizard of Menlo Park," he was the first celebrity businessman. In addition to her "good intentions," EH was certainly motivated by fame and glory and many of her lies were in service to those goals.
Having a thirst for fame and a noble cause enabled her to think it was OK to lie in service of those goals. That doesn't excuse the lies. But those noble goals may have allowed EH to excuse them for herself or, more perniciously, to make believe that they weren't lies at all. This is where we get into scary psychological territory.
But rather than thinking of it as freakish, I think it's more productive to think of it as an exaggeration of the way we all lie to others and to ourselves. That's the point of including the Dan Ariely experiment with the dice. In that experiment, most of the subjects cheated more when they thought they were doing it for a good cause. Even more disturbing, that "good cause" allowed them to lie much more effectively because they had come to believe they weren't doing anything wrong. As it turns out, economics isn't a rational practice; it's the practice of rationalizing.
Where EH and Edison differ is that Edison had a firm grip on reality. He knew he could find a way to make the incandescent lightbulb work. There is no evidence that EH was close to making her "Edison" work. But rather than face reality (and possibly adjust her goals) she pretended that her dream was real. That kind of "over-promising" or "bold vision" is one thing when you are making a prototype in the lab. It's a far more serious matter when you are using a deeply flawed system on real patients. EH can tell herself that she had to do that (Walgreens was ready to walk away if she hadn't "gone live") or else Theranos would have run out of money.
But look at the calculation she made: she thought it was worth putting lives at risk in order to make her dream come true. Now we're getting into the realm of the sociopath. But my experience leads me to believe that -- as in the case of the Milgram experiment -- most people don't do terrible things right away, they come to crimes gradually as they become more comfortable with bigger and bigger rationalizations. At Theranos, the more valuable the company became, the bigger grew the lies.
The two whistleblowers come across as courageous heroes, going up against the powerful and intimidating company. The contrast between their youth and lack of power and the old elite backers of Theronos is staggering, and yet justice triumphed. Were the whistleblowers hesitant or afraid to appear in the film, or were they eager to share their stories?
By the time I got to them, they were willing and eager to tell their stories, once I convinced them that I would honor their testimony. In the case of Erika and Tyler, they were nudged to participate by John Carreyrou, in whom they had enormous trust.
"It's simply crazy that no one demanded to see an objective demonstration of the magic box."
Why do you think so many elite veterans of politics and venture capitalism succumbed to Holmes' narrative in the first place, without checking into the details of its technology or financials?
The reasons are all in the film. First, Channing Robertson and many of the old men on her board were clearly charmed by her and maybe attracted to her. They may have rationalized their attraction by convincing themselves it was for a good cause! Second, as Dan Ariely tells us, we all respond to stories -- more than graphs and data -- because they stir us emotionally. EH was a great storyteller. Third, the story of her as a female inventor and entrepreneur in male-dominated Silicon Valley is a tale that they wanted to invest in.
There may have been other factors. EH was very clever about the way she put together an ensemble of credibility. How could Channing Robertson, George Shultz, Henry Kissinger and Jim Mattis all be wrong? And when Walgreens put the Wellness Centers in stores, investors like Rupert Murdoch assumed that Walgreens must have done its due diligence. But they hadn't!
It's simply crazy that no one demanded to see an objective demonstration of the magic box. But that blind faith, as it turns out, is more a part of capitalism than we have been taught.
Do you think that Roger Parloff deserves any blame for the glowing Fortune story on Theranos, since he appears in the film to blame himself? Or was he just one more victim of Theranos's fraud?
He put her on the cover of Fortune so he deserves some blame for the fraud. He still blames himself. That willingness to hold himself to account shows how seriously he takes the job of a journalist. Unlike Elizabeth, Roger has the honesty and moral integrity to admit that he made a mistake. He owned up to it and published a mea culpa. That said, Roger was also a victim because Elizabeth lied to him.
Do you think investors in Silicon Valley, with their FOMO attitudes and deep pockets, are vulnerable to making the same mistake again with a shiny new startup, or has this saga been a sober reminder to do their due diligence first?
Many of the mistakes made with Theranos were the same mistakes made with Enron. We must learn to recognize that we are, by nature, trusting souls. Knowing that should lead us to a guiding slogan: "trust but verify."
The irony of Holmes dancing to "I Can't Touch This" is almost too perfect. How did you find that footage?
It was leaked to us.
"Elizabeth Holmes is now famous for her fraud. Who better to host the re-boot of 'The Apprentice.'"
Holmes is facing up to 20 years in prison for federal fraud charges, but Vanity Fair recently reported that she is seeking redemption, taking meetings with filmmakers for a possible documentary to share her "real" story. What do you think will become of Holmes in the long run?
It's usually a mistake to handicap a trial. My guess is that she will be convicted and do some prison time. But maybe she can convince jurors -- the way she convinced journalists, her board, and her investors -- that, on account of her noble intentions, she deserves to be found not guilty. "Somewhere, over the rainbow…"
After the trial, and possibly prison, I'm sure that EH will use her supporters (like Tim Draper) to find a way to use the virtual currency of her celebrity to rebrand herself and launch something new. Fitzgerald famously said that "there are no second acts in American lives." That may be the stupidest thing he ever said.
Donald Trump failed at virtually every business he ever embarked on. But he became a celebrity for being a fake businessman and used that celebrity -- and phony expertise -- to become president of the United States. Elizabeth Holmes is now famous for her fraud. Who better to host the re-boot of "The Apprentice." And then?
"You Can't Touch This!"
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.
Jamie Rettinger with his now fiance Amie Purnel-Davis, who helped him through the clinical trial.
Jamie Rettinger was still in his thirties when he first noticed a tiny streak of brown running through the thumbnail of his right hand. It slowly grew wider and the skin underneath began to deteriorate before he went to a local dermatologist in 2013. The doctor thought it was a wart and tried scooping it out, treating the affected area for three years before finally removing the nail bed and sending it off to a pathology lab for analysis.
"I have some bad news for you; what we removed was a five-millimeter melanoma, a cancerous tumor that often spreads," Jamie recalls being told on his return visit. "I'd never heard of cancer coming through a thumbnail," he says. None of his doctors had ever mentioned it either. "I just thought I was being treated for a wart." But nothing was healing and it continued to bleed.
A few months later a surgeon amputated the top half of his thumb. Lymph node biopsy tested negative for spread of the cancer and when the bandages finally came off, Jamie thought his medical issues were resolved.
Melanoma is the deadliest form of skin cancer. About 85,000 people are diagnosed with it each year in the U.S. and more than 8,000 die of the cancer when it spreads to other parts of the body, according to the Centers for Disease Control and Prevention (CDC).
There are two peaks in diagnosis of melanoma; one is in younger women ages 30-40 and often is tied to past use of tanning beds; the second is older men 60+ and is related to outdoor activity from farming to sports. Light-skinned people have a twenty-times greater risk of melanoma than do people with dark skin.
"When I graduated from medical school, in 2005, melanoma was a death sentence" --Diwakar Davar.
Jamie had a follow up PET scan about six months after his surgery. A suspicious spot on his lung led to a biopsy that came back positive for melanoma. The cancer had spread. Treatment with a monoclonal antibody (nivolumab/Opdivo®) didn't prove effective and he was referred to the UPMC Hillman Cancer Center in Pittsburgh, a four-hour drive from his home in western Ohio.
An alternative monoclonal antibody treatment brought on such bad side effects, diarrhea as often as 15 times a day, that it took more than a week of hospitalization to stabilize his condition. The only options left were experimental approaches in clinical trials.
Early research
"When I graduated from medical school, in 2005, melanoma was a death sentence" with a cure rate in the single digits, says Diwakar Davar, 39, an oncologist at UPMC Hillman Cancer Center who specializes in skin cancer. That began to change in 2010 with introduction of the first immunotherapies, monoclonal antibodies, to treat cancer. The antibodies attach to PD-1, a receptor on the surface of T cells of the immune system and on cancer cells. Antibody treatment boosted the melanoma cure rate to about 30 percent. The search was on to understand why some people responded to these drugs and others did not.
At the same time, there was a growing understanding of the role that bacteria in the gut, the gut microbiome, plays in helping to train and maintain the function of the body's various immune cells. Perhaps the bacteria also plays a role in shaping the immune response to cancer therapy.
One clue came from genetically identical mice. Animals ordered from different suppliers sometimes responded differently to the experiments being performed. That difference was traced to different compositions of their gut microbiome; transferring the microbiome from one animal to another in a process known as fecal transplant (FMT) could change their responses to disease or treatment.
When researchers looked at humans, they found that the patients who responded well to immunotherapies had a gut microbiome that looked like healthy normal folks, but patients who didn't respond had missing or reduced strains of bacteria.
Davar and his team knew that FMT had a very successful cure rate in treating the gut dysbiosis of Clostridioides difficile, a persistant intestinal infection, and they wondered if a fecal transplant from a patient who had responded well to cancer immunotherapy treatment might improve the cure rate of patients who did not originally respond to immunotherapies for melanoma.
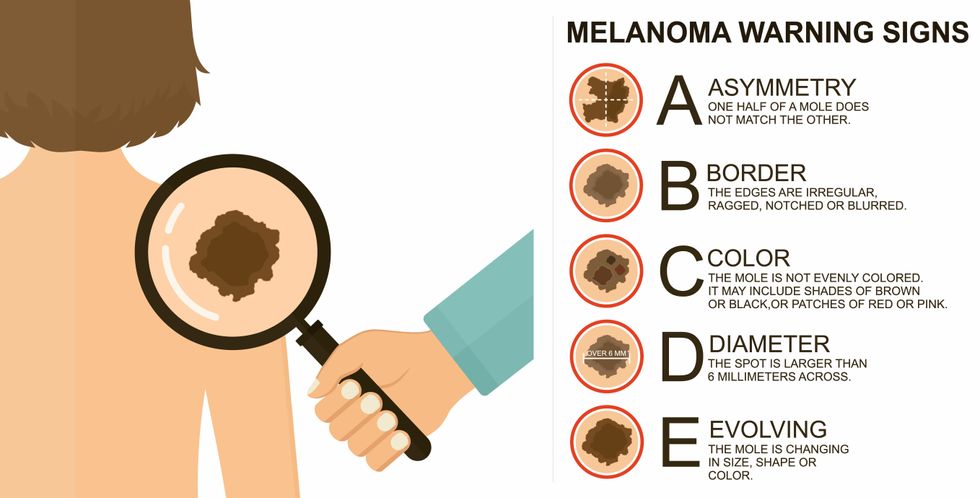
The ABCDE of melanoma detection
Adobe Stock
Clinical trial
"It was pretty weird, I was totally blasted away. Who had thought of this?" Jamie first thought when the hypothesis was explained to him. But Davar's explanation that the procedure might restore some of the beneficial bacterial his gut was lacking, convinced him to try. He quickly signed on in October 2018 to be the first person in the clinical trial.
Fecal donations go through the same safety procedures of screening for and inactivating diseases that are used in processing blood donations to make them safe for transfusion. The procedure itself uses a standard hollow colonoscope designed to screen for colon cancer and remove polyps. The transplant is inserted through the center of the flexible tube.
Most patients are sedated for procedures that use a colonoscope but Jamie doesn't respond to those drugs: "You can't knock me out. I was watching them on the TV going up my own butt. It was kind of unreal at that point," he says. "There were about twelve people in there watching because no one had seen this done before."
A test two weeks after the procedure showed that the FMT had engrafted and the once-missing bacteria were thriving in his gut. More importantly, his body was responding to another monoclonal antibody (pembrolizumab/Keytruda®) and signs of melanoma began to shrink. Every three months he made the four-hour drive from home to Pittsburgh for six rounds of treatment with the antibody drug.
"We were very, very lucky that the first patient had a great response," says Davar. "It allowed us to believe that even though we failed with the next six, we were on the right track. We just needed to tweak the [fecal] cocktail a little better" and enroll patients in the study who had less aggressive tumor growth and were likely to live long enough to complete the extensive rounds of therapy. Six of 15 patients responded positively in the pilot clinical trial that was published in the journal Science.
Davar believes they are beginning to understand the biological mechanisms of why some patients initially do not respond to immunotherapy but later can with a FMT. It is tied to the background level of inflammation produced by the interaction between the microbiome and the immune system. That paper is not yet published.
Surviving cancer
It has been almost a year since the last in his series of cancer treatments and Jamie has no measurable disease. He is cautiously optimistic that his cancer is not simply in remission but is gone for good. "I'm still scared every time I get my scans, because you don't know whether it is going to come back or not. And to realize that it is something that is totally out of my control."
"It was hard for me to regain trust" after being misdiagnosed and mistreated by several doctors he says. But his experience at Hillman helped to restore that trust "because they were interested in me, not just fixing the problem."
He is grateful for the support provided by family and friends over the last eight years. After a pause and a sigh, the ruggedly built 47-year-old says, "If everyone else was dead in my family, I probably wouldn't have been able to do it."
"I never hesitated to ask a question and I never hesitated to get a second opinion." But Jamie acknowledges the experience has made him more aware of the need for regular preventive medical care and a primary care physician. That person might have caught his melanoma at an earlier stage when it was easier to treat.
Davar continues to work on clinical studies to optimize this treatment approach. Perhaps down the road, screening the microbiome will be standard for melanoma and other cancers prior to using immunotherapies, and the FMT will be as simple as swallowing a handful of freeze-dried capsules off the shelf rather than through a colonoscopy. Earlier this year, the Food and Drug Administration approved the first oral fecal microbiota product for C. difficile, hopefully paving the way for more.
An older version of this hit article was first published on May 18, 2021
All organisms can repair damaged tissue, but none do it better than salamanders and newts. A surprising area of science could tell us how they manage this feat - and perhaps even help us develop a similar ability.
All organisms have the capacity to repair or regenerate tissue damage. None can do it better than salamanders or newts, which can regenerate an entire severed limb.
That feat has amazed and delighted man from the dawn of time and led to endless attempts to understand how it happens – and whether we can control it for our own purposes. An exciting new clue toward that understanding has come from a surprising source: research on the decline of cells, called cellular senescence.
Senescence is the last stage in the life of a cell. Whereas some cells simply break up or wither and die off, others transition into a zombie-like state where they can no longer divide. In this liminal phase, the cell still pumps out many different molecules that can affect its neighbors and cause low grade inflammation. Senescence is associated with many of the declining biological functions that characterize aging, such as inflammation and genomic instability.
Oddly enough, newts are one of the few species that do not accumulate senescent cells as they age, according to research over several years by Maximina Yun. A research group leader at the Center for Regenerative Therapies Dresden and the Max Planck Institute of Molecular and Cell Biology and Genetics, in Dresden, Germany, Yun discovered that senescent cells were induced at some stages of regeneration of the salamander limb, “and then, as the regeneration progresses, they disappeared, they were eliminated by the immune system,” she says. “They were present at particular times and then they disappeared.”
Senescent cells added to the edges of the wound helped the healthy muscle cells to “dedifferentiate,” essentially turning back the developmental clock of those cells into more primitive states.
Previous research on senescence in aging had suggested, logically enough, that applying those cells to the stump of a newly severed salamander limb would slow or even stop its regeneration. But Yun stood that idea on its head. She theorized that senescent cells might also play a role in newt limb regeneration, and she tested it by both adding and removing senescent cells from her animals. It turned out she was right, as the newt limbs grew back faster than normal when more senescent cells were included.
Senescent cells added to the edges of the wound helped the healthy muscle cells to “dedifferentiate,” essentially turning back the developmental clock of those cells into more primitive states, which could then be turned into progenitors, a cell type in between stem cells and specialized cells, needed to regrow the muscle tissue of the missing limb. “We think that this ability to dedifferentiate is intrinsically a big part of why salamanders can regenerate all these very complex structures, which other organisms cannot,” she explains.
Yun sees regeneration as a two part problem. First, the cells must be able to sense that their neighbors from the lost limb are not there anymore. Second, they need to be able to produce the intermediary progenitors for regeneration, , to form what is missing. “Molecularly, that must be encoded like a 3D map,” she says, otherwise the new tissue might grow back as a blob, or liver, or fin instead of a limb.
Wound healing
Another recent study, this time at the Mayo Clinic, provides evidence supporting the role of senescent cells in regeneration. Looking closely at molecules that send information between cells in the wound of a mouse, the researchers found that senescent cells appeared near the start of the healing process and then disappeared as healing progressed. In contrast, persistent senescent cells were the hallmark of a chronic wound that did not heal properly. The function and significance of senescence cells depended on both the timing and the context of their environment.
The paper suggests that senescent cells are not all the same. That has become clearer as researchers have been able to identify protein markers on the surface of some senescent cells. The patterns of these proteins differ for some senescent cells compared to others. In biology, such physical differences suggest functional differences, so it is becoming increasingly likely there are subsets of senescent cells with differing functions that have not yet been identified.
There are disagreements within the research community as to whether newts have acquired their regenerative capacity through a unique evolutionary change, or if other animals, including humans, retain this capacity buried somewhere in their genes.
Scientists initially thought that senescent cells couldn’t play a role in regeneration because they could no longer reproduce, says Anthony Atala, a practicing surgeon and bioengineer who leads the Wake Forest Institute for Regenerative Medicine in North Carolina. But Yun’s study points in the other direction. “What this paper shows clearly is that these cells have the potential to be involved in tissue regeneration [in newts]. The question becomes, will these cells be able to do the same in humans.”
As our knowledge of senescent cells increases, Atala thinks we need to embrace a new analogy to help understand them: humans in retirement. They “have acquired a lot of wisdom throughout their whole life and they can help younger people and mentor them to grow to their full potential. We're seeing the same thing with these cells,” he says. They are no longer putting energy into their own reproduction, but the signaling molecules they secrete “can help other cells around them to regenerate.”
There are disagreements within the research community as to whether newts have acquired their regenerative capacity through a unique evolutionary change, or if other animals, including humans, retain this capacity buried somewhere in their genes. If so, it seems that our genes are unable to express this ability, perhaps as part of a tradeoff in acquiring other traits. It is a fertile area of research.
Dedifferentiation is likely to become an important process in the field of regenerative medicine. One extreme example: a lab has been able to turn back the clock and reprogram adult male skin cells into female eggs, a potential milestone in reproductive health. It will be more difficult to control just how far back one wishes to go in the cell's dedifferentiation – part way or all the way back into a stem cell – and then direct it down a different developmental pathway. Yun is optimistic we can learn these tricks from newts.
Senolytics
A growing field of research is using drugs called senolytics to remove senescent cells and slow or even reverse disease of aging.
“Senolytics are great, but senolytics target different types of senescence,” Yun says. “If senescent cells have positive effects in the context of regeneration, of wound healing, then maybe at the beginning of the regeneration process, you may not want to take them out for a little while.”
“If you look at pretty much all biological systems, too little or too much of something can be bad, you have to be in that central zone” and at the proper time, says Atala. “That's true for proteins, sugars, and the drugs that you take. I think the same thing is true for these cells. Why would they be different?”
Our growing understanding that senescence is not a single thing but a variety of things likely means that effective senolytic drugs will not resemble a single sledge hammer but more a carefully manipulated scalpel where some types of senescent cells are removed while others are added. Combinations and timing could be crucial, meaning the difference between regenerating healthy tissue, a scar, or worse.

