Fetuses can save their mothers' lives
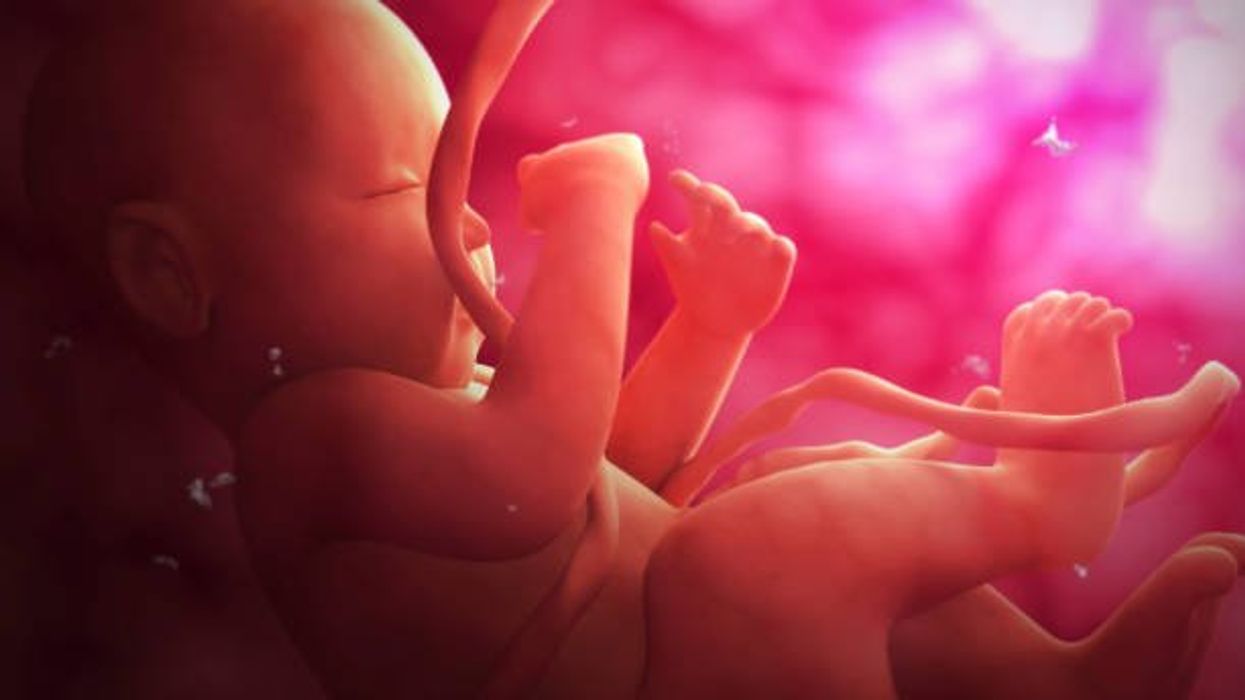
Stem cells from a fetus can travel to the heart and regenerate the muscle, essentially saving a mother’s life.
Story by Big Think
In rare cases, a woman’s heart can start to fail in the months before or after giving birth. The all-important muscle weakens as its chambers enlarge, reducing the amount of blood pumped with each beat. Peripartum cardiomyopathy can threaten the lives of both mother and child. Viral illness, nutritional deficiency, the bodily stress of pregnancy, or an abnormal immune response could all play a role, but the causes aren’t concretely known.
If there is a silver lining to peripartum cardiomyopathy, it’s that it is perhaps the most survivable form of heart failure. A remarkable 50% of women recover spontaneously. And there’s an even more remarkable explanation for that glowing statistic: The fetus‘ stem cells migrate to the heart and regenerate the beleaguered muscle. In essence, the developing or recently born child saves its mother’s life.
Saving mama
While this process has not been observed directly in humans, it has been witnessed in mice. In a 2015 study, researchers tracked stem cells from fetal mice as they traveled to mothers’ damaged cardiac cells and integrated themselves into hearts.
Evolutionarily, this function makes sense: It is in the fetus’ best interest that its mother remains healthy.
Scientists also have spotted cells from the fetus within the hearts of human mothers, as well as countless other places inside the body, including the skin, spleen, liver, brain, lung, kidney, thyroid, lymph nodes, salivary glands, gallbladder, and intestine. These cells essentially get everywhere. While most are eliminated by the immune system during pregnancy, some can persist for an incredibly long time — up to three decades after childbirth.
This integration of the fetus’ cells into the mother’s body has been given a name: fetal microchimerism. The process appears to start between the fourth and sixth week of gestation in humans. Scientists are actively trying to suss out its purpose. Fetal stem cells, which can differentiate into all sorts of specialized cells, appear to target areas of injury. So their role in healing seems apparent. Evolutionarily, this function makes sense: It is in the fetus’ best interest that its mother remains healthy.
Sending cells into the mother’s body may also prime her immune system to grow more tolerant of the developing fetus. Successful pregnancy requires that the immune system not see the fetus as an interloper and thus dispatch cells to attack it.
Fetal microchimerism
But fetal microchimerism might not be entirely beneficial. Greater concentrations of the cells have been associated with various autoimmune diseases such as lupus, Sjogren’s syndrome, and even multiple sclerosis. After all, they are foreign cells living in the mother’s body, so it’s possible that they might trigger subtle, yet constant inflammation. Fetal cells also have been linked to cancer, although it isn’t clear whether they abet or hinder the disease.
A team of Spanish scientists summarized the apparent give and take of fetal microchimerism in a 2022 review article. “On the one hand, fetal microchimerism could be a source of progenitor cells with a beneficial effect on the mother’s health by intervening in tissue repair, angiogenesis, or neurogenesis. On the other hand, fetal microchimerism might have a detrimental function by activating the immune response and contributing to autoimmune diseases,” they wrote.
Regardless of a fetus’ cells net effect, their existence alone is intriguing. In a paper published earlier this year, University of London biologist Francisco Úbeda and University of Western Ontario mathematical biologist Geoff Wild noted that these cells might very well persist within mothers for life.
“Therefore, throughout their reproductive lives, mothers accumulate fetal cells from each of their past pregnancies including those resulting in miscarriages. Furthermore, mothers inherit, from their own mothers, a pool of cells contributed by all fetuses carried by their mothers, often referred to as grandmaternal microchimerism.”
So every mother may carry within her literal pieces of her ancestors.
Too much of this ingredient leads to autoimmune diseases, new research shows. Here's how to cut back.
Scientists are looking at how salt affects our cells, and they're finding more reasons to avoid htoo much of it.
For more than a century, doctors have warned that too much salt in your diet can lead to high blood pressure, heart disease and stroke - and many of the reasons for these effects are well known. But recently scientists have been looking deeper, into the cellular level, and they are finding additional reasons to minimize sodium intake; it is bad for immune cells, creating patterns of gene expression and activity seen in a variety of autoimmune diseases such as multiple sclerosis, lupus, rheumatoid arthritis, and type-1 diabetes.
Salt is a major part of the ocean from which life evolved on this planet. We carry that legacy in our blood, which tastes salty. It is an important element for conducting electrical signals along nerves and balancing water and metabolites transported throughout our bodies. We need to consume about 500 milligrams of salt each day to maintain these functions, more with exercise and heavy sweating as that is a major way the body loses salt. The problem is that most Americans eating a modern western diet consume about 3400 milligrams, 1.5 teaspoons per day.
Evidence has been accumulating over the last few years that elevated levels of sodium can be harmful to at least some types of immune cells. The first signal came in monocytes, which are immune cells that travel to various tissues in the body, where some of them turn into macrophages, a subset of white blood cells that can directly kill microorganisms and make chemical signals that bring other types of immune cells into play.
Two years ago, Dominik N. Müller from the Max-Delbrueck-Center in Berlin, Germany and Markus Kleinewietfeld, an immunologist at Hasselt University in Belgium, ran a study where they fed people pizza and then measured their immune cell function. “We saw that in any monocytes, metabolic function was down, even after a single salty meal,” Kleinewietfeld says. It seemed to be the cellular equivalent of the sluggish feeling we get after eating too much. The cells were able to recover but more research is needed to answer questions about what dose of sodium causes impairment, how long the damage lasts, and whether there is a cumulative effect of salt toxicity.
Kleinewietfeld and his colleagues have hypothesized that too much salt could be a significant factor in the increased number of autoimmune diseases and allergies over the last few generations.
The latest series of experiments focused on a type of T cell called T regulatory cells, or Tregs. Most T cells release inflammatory mediators to fight pathogens and, once that job is done, Tregs come along to calm down their hyperactive brethren. Failure to do so can result in continued inflammation and possibly autoimmune diseases.
In the lab, Kleinewietfeld and his large team of international collaborators saw that high levels of sodium had a huge effect on Tregs, upregulating 1250 genes and downregulating an additional 1380 genes so that they looked similar to patterns of gene expression seen in autoimmune diseases.
Digging deeper, they found that sodium affected mitochondria, the tiny organelles inside of cells that produce much of its energy. The sodium was interfering with how the mitochondria use oxygen, which resulted in increased levels of an unstable form of oxygen that can damage cell function. The researchers injected those damaged Tregs into mice and found that they impaired the animals' immune function, allowing the inflammation to continue rather than shutting it down.
That finding dovetailed nicely with a 2019 paper in Nature from Navdeep Chandel's lab at Northwestern University, which showed in mice that inhibiting the mitochondrial use of oxygen reduced the ability of Tregs to regulate other T cells. “Mitochondria were controlling directly the immunosuppressive program, they were this master regulator tuning the right amount of genes to give you proper immunosuppression,” Chandel said. “And if you lose that function, then you get autoimmunity.”
Kleinewietfeld's team studied the Treg cells of humans and found that sodium can similarly decrease mitochondrial use of oxygen and immunosuppressive activity. “I would have never predicted that myself,” Chandel says, but now researchers can look at the mitochondria of patients with autoimmune disease and see if their gene expression also changes under high salt conditions. He sees the link between the patterns of gene expression in Tregs generated by high salt exposure and those patterns seen in autoimmune diseases, but he is cautious about claiming a causal effect.
Kleinewietfeld and his colleagues have hypothesized that too much salt could be a significant factor in the increased number of autoimmune diseases and allergies over the last few generations. He says a high salt diet could also have an indirect effect on immune function through the way it affects the gut microbiome and the molecules made by microbes when they break down food. But the research results are too preliminary to say that for sure, much less parse out the role of salt compared with other possible factors. “It is still an exciting journey to try to understand this field,” he says.
Additionally, it is difficult to say precisely how this research in animals and human cell cultures will translate into a whole human body. Individual differences in genetics can affect how the body absorbs, transports, and gets rid of sodium, such that some people are more sensitive to salt than are others.
So how should people apply these research findings to daily life?
Salt is obvious when we sprinkle it on at the table or eat tasty things like potato chips, but we may be unaware of sodium hidden in packaged foods. That's because salt is an easy and cheap way to boost the flavor of foods. And if we do read the labeled salt content on a package, we focus on the number for a single serving, but then eat more than that.
Last September, the U.S. Food and Drug Administration (FDA) began a process to update labels on the content of food, including what is meant by the word “healthy” and how food manufacturers can use the term. Many in the food industry are resisting those proposed changes.
Chandel cautions against trying to counter the effects of salt by reaching for foods or supplements full of antioxidants, which, in theory, could reduce the harmful effects on mitochondria caused by a heavy hand with the salt shaker.
Until labels are updated, it would be prudent to try to reduce sodium intake by cutting down on packaged foods while making your own food at home, where you know just how much salt has been added. The Mayo Clinic offers guidance on how to become more aware of the sodium in your diet and eat less of it.
Chandel thinks many people will struggle with minimizing salt in their diets. It’s similar to the challenge of eating less sugar, in that the body craves both, and it is difficult to fight that. He cautions against trying to counter the effects of salt by reaching for foods or supplements full of antioxidants, which, in theory, could reduce the harmful effects on mitochondria caused by a heavy hand with the salt shaker. “Dietary antioxidants have failed in just about every clinical trial, yet the public continues to take them,” Chandel says. But he is optimistic that research will lead us to a better understanding of how Tregs function, and uncover new targets for treating autoimmune diseases.
In this week's Friday Five, new research that could help prevent Alzheimer's. Plus, why you should care about smart senior towns, how to reverse being drunk, money can make you happier if you're this type of person, and personalized anxiety medicine.
The Friday Five covers five stories in research that you may have missed this week. There are plenty of controversies and troubling ethical issues in science – and we get into many of them in our online magazine – but this news roundup focuses on scientific creativity and progress to give you a therapeutic dose of inspiration headed into the weekend.
Here are the promising studies covered in this week's Friday Five, featuring interviews with Dr. Christopher Martens, director of the Delaware Center for Cogntiive Aging Research and professor of kinesiology and applied physiology at the University of Delaware, and Dr. Ilona Matysiak, visiting scholar at Iowa State University and associate professor of sociology at Maria Grzegorzewska University.
Listen on Apple | Listen on Spotify | Listen on Stitcher | Listen on Amazon | Listen on Google
- Could this supplement help prevent Alzheimer's?
- Why you should care about smart senior towns
- Here's how to reverse being drunk
- Money can make you happy - if you're this type of person
- Personalized anxiety medicine


