A skin patch to treat peanut allergies teaches the body to tolerate the nuts

Peanut allergies affect about a million children in the U.S., and most never outgrow them. Luckily, some promising remedies are in the works.
Ever since he was a baby, Sharon Wong’s son Brandon suffered from rashes, prolonged respiratory issues and vomiting. In 2006, as a young child, he was diagnosed with a severe peanut allergy.
"My son had a history of reacting to traces of peanuts in the air or in food,” says Wong, a food allergy advocate who runs a blog focusing on nut free recipes, cooking techniques and food allergy awareness. “Any participation in school activities, social events, or travel with his peanut allergy required a lot of preparation.”
Peanut allergies affect around a million children in the U.S. Most never outgrow the condition. The problem occurs when the immune system mistakenly views the proteins in peanuts as a threat and releases chemicals to counteract it. This can lead to digestive problems, hives and shortness of breath. For some, like Wong’s son, even exposure to trace amounts of peanuts could be life threatening. They go into anaphylactic shock and need to take a shot of adrenaline as soon as possible.
Typically, people with peanut allergies try to completely avoid them and carry an adrenaline autoinjector like an EpiPen in case of emergencies. This constant vigilance is very stressful, particularly for parents with young children.
“The search for a peanut allergy ‘cure’ has been a vigorous one,” says Claudia Gray, a pediatrician and allergist at Vincent Pallotti Hospital in Cape Town, South Africa. The closest thing to a solution so far, she says, is the process of desensitization, which exposes the patient to gradually increasing doses of peanut allergen to build up a tolerance. The most common type of desensitization is oral immunotherapy, where patients ingest small quantities of peanut powder. It has been effective but there is a risk of anaphylaxis since it involves swallowing the allergen.
"By the end of the trial, my son tolerated approximately 1.5 peanuts," Sharon Wong says.
DBV Technologies, a company based in Montrouge, France has created a skin patch to address this problem. The Viaskin Patch contains a much lower amount of peanut allergen than oral immunotherapy and delivers it through the skin to slowly increase tolerance. This decreases the risk of anaphylaxis.
Wong heard about the peanut patch and wanted her son to take part in an early phase 2 trial for 4-to-11-year-olds.
“We felt that participating in DBV’s peanut patch trial would give him the best chance at desensitization or at least increase his tolerance from a speck of peanut to a peanut,” Wong says. “The daily routine was quite simple, remove the old patch and then apply a new one. By the end of the trial, he tolerated approximately 1.5 peanuts.”
How it works
For DBV Technologies, it all began when pediatric gastroenterologist Pierre-Henri Benhamou teamed up with fellow professor of gastroenterology Christopher Dupont and his brother, engineer Bertrand Dupont. Together they created a more effective skin patch to detect when babies have allergies to cow's milk. Then they realized that the patch could actually be used to treat allergies by promoting tolerance. They decided to focus on peanut allergies first as the more dangerous.
The Viaskin patch utilizes the fact that the skin can promote tolerance to external stimuli. The skin is the body’s first defense. Controlling the extent of the immune response is crucial for the skin. So it has defense mechanisms against external stimuli and can promote tolerance.
The patch consists of an adhesive foam ring with a plastic film on top. A small amount of peanut protein is placed in the center. The adhesive ring is attached to the back of the patient's body. The peanut protein sits above the skin but does not directly touch it. As the patient sweats, water droplets on the inside of the film dissolve the peanut protein, which is then absorbed into the skin.
The peanut protein is then captured by skin cells called Langerhans cells. They play an important role in getting the immune system to tolerate certain external stimuli. Langerhans cells take the peanut protein to lymph nodes which activate T regulatory cells. T regulatory cells suppress the allergic response.
A different patch is applied to the skin every day to increase tolerance. It’s both easy to use and convenient.
“The DBV approach uses much smaller amounts than oral immunotherapy and works through the skin significantly reducing the risk of allergic reactions,” says Edwin H. Kim, the division chief of Pediatric Allergy and Immunology at the University of North Carolina, U.S., and one of the principal investigators of Viaskin’s clinical trials. “By not going through the mouth, the patch also avoids the taste and texture issues. Finally, the ability to apply a patch and immediately go about your day may be very attractive to very busy patients and families.”

Brandon Wong displaying origami figures he folded at an Origami Convention in 2022
Sharon Wong
Clinical trials
Results from DBV's phase 3 trial in children ages 1 to 3 show its potential. For a positive result, patients who could not tolerate 10 milligrams or less of peanut protein had to be able to manage 300 mg or more after 12 months. Toddlers who could already tolerate more than 10 mg needed to be able to manage 1000 mg or more. In the end, 67 percent of subjects using the Viaskin patch met the target as compared to 33 percent of patients taking the placebo dose.
“The Viaskin peanut patch has been studied in several clinical trials to date with promising results,” says Suzanne M. Barshow, assistant professor of medicine in allergy and asthma research at Stanford University School of Medicine in the U.S. “The data shows that it is safe and well-tolerated. Compared to oral immunotherapy, treatment with the patch results in fewer side effects but appears to be less effective in achieving desensitization.”
The primary reason the patch is less potent is that oral immunotherapy uses a larger amount of the allergen. Additionally, absorption of the peanut protein into the skin could be erratic.
Gray also highlights that there is some tradeoff between risk and efficacy.
“The peanut patch is an exciting advance but not as effective as the oral route,” Gray says. “For those patients who are very sensitive to orally ingested peanut in oral immunotherapy or have an aversion to oral peanut, it has a use. So, essentially, the form of immunotherapy will have to be tailored to each patient.” Having different forms such as the Viaskin patch which is applied to the skin or pills that patients can swallow or dissolve under the tongue is helpful.
The hope is that the patch’s efficacy will increase over time. The team is currently running a follow-up trial, where the same patients continue using the patch.
“It is a very important study to show whether the benefit achieved after 12 months on the patch stays stable or hopefully continues to grow with longer duration,” says Kim, who is an investigator in this follow-up trial.
"My son now attends university in Massachusetts, lives on-campus, and eats dorm food. He has so much more freedom," Wong says.
The team is further ahead in the phase 3 follow-up trial for 4-to-11-year-olds. The initial phase 3 trial was not as successful as the trial for kids between one and three. The patch enabled patients to tolerate more peanuts but there was not a significant enough difference compared to the placebo group to be definitive. The follow-up trial showed greater potency. It suggests that the longer patients are on the patch, the stronger its effects.
They’re also testing if making the patch bigger, changing the shape and extending the minimum time it’s worn can improve its benefits in a trial for a new group of 4-to-11 year-olds.
The future
DBV Technologies is using the skin patch to treat cow’s milk allergies in children ages 1 to 17. They’re currently in phase 2 trials.
As for the peanut allergy trials in toddlers, the hope is to see more efficacy soon.
For Wong’s son who took part in the earlier phase 2 trial for 4-to-11-year-olds, the patch has transformed his life.
“My son continues to maintain his peanut tolerance and is not affected by peanut dust in the air or cross-contact,” Wong says. ”He attends university in Massachusetts, lives on-campus, and eats dorm food. He still carries an EpiPen but has so much more freedom than before his clinical trial. We will always be grateful.”
A Mother-and-Daughter Team Have Developed What May Be the World’s First Alzheimer’s Vaccine
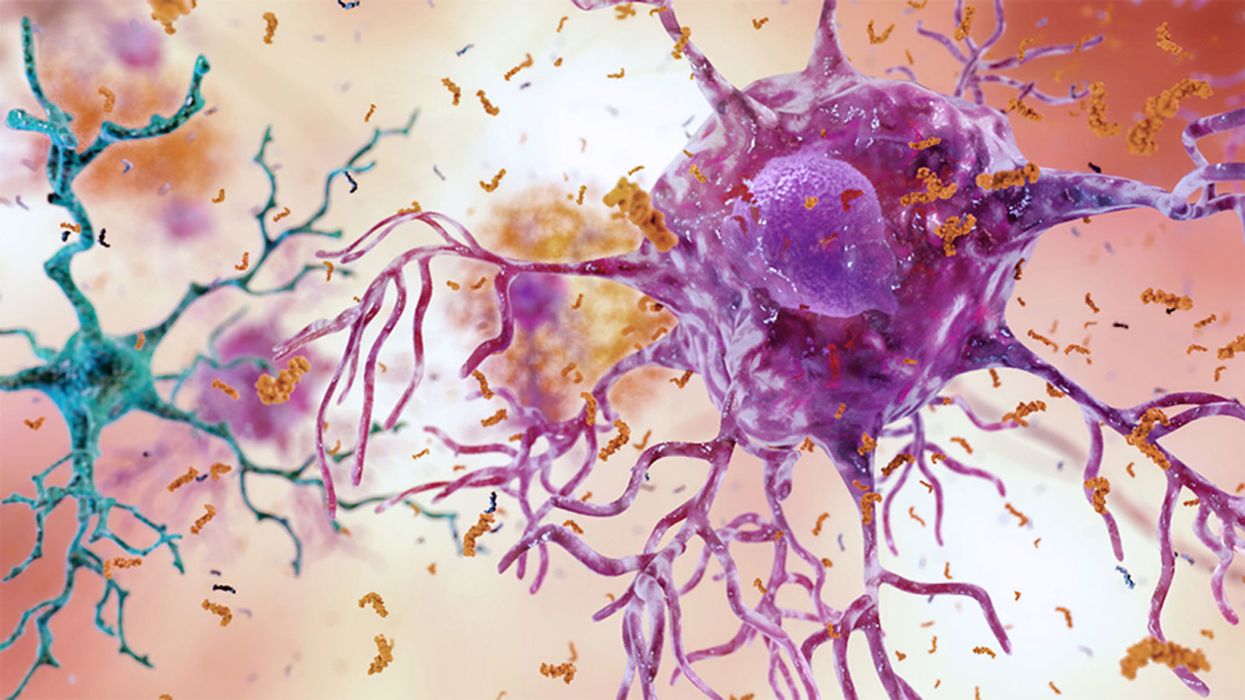
Brain inflammation from Alzheimer's disease.
Alzheimer's is a terrible disease that robs a person of their personality and memory before eventually leading to death. It's the sixth-largest killer in the U.S. and, currently, there are 5.8 million Americans living with the disease.
Wang's vaccine is a significant improvement over previous attempts because it can attack the Alzheimer's protein without creating any adverse side effects.
It devastates people and families and it's estimated that Alzheimer's and other forms of dementia will cost the U.S. $290 billion dollars this year alone. It's estimated that it will become a trillion-dollar-a-year disease by 2050.
There have been over 200 unsuccessful attempts to find a cure for the disease and the clinical trial termination rate is 98 percent.
Alzheimer's is caused by plaque deposits that develop in brain tissue that become toxic to brain cells. One of the major hurdles to finding a cure for the disease is that it's impossible to clear out the deposits from the tissue. So scientists have turned their attention to early detection and prevention.
One very encouraging development has come out of the work done by Dr. Chang Yi Wang, PhD. Wang is a prolific bio-inventor; one of her biggest successes is developing a foot-and-mouth vaccine for pigs that has been administered more than three billion times.

Mei Mei Hu
Brainstorm Health / Flickr.
In January, United Neuroscience, a biotech company founded by Yi, her daughter Mei Mei Hu, and son-in-law, Louis Reese, announced the first results from a phase IIa clinical trial on UB-311, an Alzheimer's vaccine.
The vaccine has synthetic versions of amino acid chains that trigger antibodies to attack Alzheimer's protein the blood. Wang's vaccine is a significant improvement over previous attempts because it can attack the Alzheimer's protein without creating any adverse side effects.
"We were able to generate some antibodies in all patients, which is unusual for vaccines," Yi told Wired. "We're talking about almost a 100 percent response rate. So far, we have seen an improvement in three out of three measurements of cognitive performance for patients with mild Alzheimer's disease."
The researchers also claim it can delay the onset of the disease by five years. While this would be a godsend for people with the disease and their families, according to Elle, it could also save Medicare and Medicaid more than $220 billion.
"You'd want to see larger numbers, but this looks like a beneficial treatment," James Brown, director of the Aston University Research Centre for Healthy Ageing, told Wired. "This looks like a silver bullet that can arrest or improve symptoms and, if it passes the next phase, it could be the best chance we've got."
"A word of caution is that it's a small study," says Drew Holzapfel, acting president of the nonprofit UsAgainstAlzheimer's, said according to Elle. "But the initial data is compelling."
The company is now working on its next clinical trial of the vaccine and while hopes are high, so is the pressure. The company has already invested $100 million developing its vaccine platform. According to Reese, the company's ultimate goal is to create a host of vaccines that will be administered to protect people from chronic illness.
"We have a 50-year vision -- to immuno-sculpt people against chronic illness and chronic aging with vaccines as prolific as vaccines for infectious diseases," he told Elle.
[Editor's Note: This article was originally published by Upworthy here and has been republished with permission.]
Turning Algae Into Environmentally Friendly Fuel Just Got Faster and Smarter
Algae in the Mediterranean sea.
Was your favorite beach closed this summer? Algae blooms are becoming increasingly the reason to blame and, as the climate heats up, scientists say we can expect more of the warm water-loving blue-green algae to grow.
"We have removed a significant development barrier to make algal biofuel production more efficient and smarter."
Oddly enough, the pesky growth could help fuel our carbon-friendly options.
This year, the University of Utah scientists discovered a faster way to turn algae into fuel. Algae is filled with lipids that we can feed our energy-hungry diesel engines. The problem is extracting the lipids, which usually requires more energy to transform than the actual energy we'd get – not achieving what scientists call "energy parity."
But now, the University of Utah team has discovered a new mix that is more efficient and much faster. We can now extract more power from algae with less waste materials after the fact. Paper co-author Dr. Leonard Pease says, "We have removed a significant development barrier to make algal biofuel production more efficient and smarter. Our method puts us much closer to creating biofuels energy parity than we were before."
Next Up
Algae has a lot going for it as an alternative fuel source. It grows fast and easily, absorbs carbon dioxide, does not compete with food crops for land, and could produce up to 60 times more oil than standard land-based energy crops, according to the U.S. Department of Energy. Yet the costs of algal biofuel production are still expensive for now.
According to Science Daily, only about five percent of total primary energy use in the United States came from algae and other biomass forms. By making the process more efficient, America and other nations could potentially begin relying on more plentiful resources – which, ironically, are more common now because of climate change.
Algae fuel efficiency is already a proven concept. A decade ago, Continental Airlines completed a 90-minute Boeing 737-800 flight with one engine split between biofuel and aircraft fuel. The biofuel was straight from algae. (Other flights were done based on nut fuel and other alternative sources.) The commercial airplane required no modification to the engine and the biofuel itself exceeded the standards of traditional jet fuel.
The problem, as noted at the time, is that biofuels derived from algae had yet to be proven as "commercially competitive."
The University of Utah's discovery could mean cheaper processing. At this point, it is less about if it works and more about if it is a practical alternative.
However, it's unclear how long it will take for algae to become more mainstream, if ever.
Open Questions
Higher efficiency and simpler transformations could mean lower prices and more business access. However, it's unclear how long it will take for algae to become more mainstream, if ever. The algae biofuel worked great for a relatively sophisticated Boeing 737 engine, but your family car, the cross-country delivery trucks and other less powerful machines may need to be modified – and that means the industry-at-large would have to revise their products in order to support the change.
Future-focused groups are already looking at how algae can fuel our space programs, especially if it is more renewable, safe and, potentially, cheaper than our traditional fuel choices. But first, it is worth waiting and seeing if corporations and, later, citizens are willing to take the plunge.