Health breakthroughs of 2022 that should have made bigger news

Nine experts break down the biggest biotech and health breakthroughs that didn't get the attention they deserved in 2022.
As the world has attempted to move on from COVID-19 in 2022, attention has returned to other areas of health and biotech with major regulatory approvals such as the Alzheimer's drug lecanemab – which can slow the destruction of brain cells in the early stages of the disease – being hailed by some as momentous breakthroughs.
This has been a year where psychedelic medicines have gained the attention of mainstream researchers with a groundbreaking clinical trial showing that psilocybin treatment can help relieve some of the symptoms of major depressive disorder. And with messenger RNA (mRNA) technology still very much capturing the imagination, the readouts of cancer vaccine trials have made headlines around the world.
But at the same time there have been vital advances which will likely go on to change medicine, and yet have slipped beneath the radar. I asked nine forward-thinking experts on health and biotech about the most important, but underappreciated, breakthrough of 2022.
Their descriptions, below, were lightly edited by Leaps.org for style and format.
New drug targets for Alzheimer’s disease

Professor Julie Williams, Director, Dementia Research Institute, Cardiff University
Genetics has changed our view of Alzheimer’s disease in the last five to six years. The beta amyloid hypothesis has dominated Alzheimer’s research for a long time, but there are multiple components to this complex disease, of which getting rid of amyloid plaques is one, but it is not the whole story. In April 2022, Nature published a paper which is the culmination of a decade’s worth of work - groups all over the world working together to identify 75 genes associated with risk of developing Alzheimer’s. This provides us with a roadmap for understanding the disease mechanisms.
For example, it is showing that there is something different about the immune systems of people who develop Alzheimer’s disease. There is something different about the way they process lipids in the brain, and very specific processes of how things travel through cells called endocytosis. When it comes to immunity, it indicates that the complement system is affecting whether synapses, which are the connections between neurons, get eliminated or not. In Alzheimer’s this process is more severe, so patients are losing more synapses, and this is correlated with cognition.
The genetics also implicates very specific tissues like microglia, which are the housekeepers in the brain. One of their functions is to clear away beta amyloid, but they also prune and nibble away at parts of the brain that are indicated to be diseased. If you have these risk genes, it seems that you are likely to prune more tissue, which may be part of the cell death and neurodegeneration that we observe in Alzheimer’s patients.
Genetics is telling us that we need to be looking at multiple causes of this complex disease, and we are doing that now. It is showing us that there are a number of different processes which combine to push patients into a disease state which results in the death of connections between nerve cells. These findings around the complement system and other immune-related mechanisms are very interesting as there are already drugs which are available for other diseases which could be repurposed in clinical trials. So it is really a turning point for us in the Alzheimer’s disease field.
Preventing Pandemics with Organ-Tissue Equivalents

Anthony Atala, Director of the Wake Forest Institute for Regenerative Medicine
COVID-19 has shown us that we need to be better prepared ahead of future pandemics and have systems in place where we can quickly catalogue a new virus and have an idea of which treatment agents would work best against it.
At Wake Forest Institute, our scientists have developed what we call organ-tissue equivalents. These are miniature tissues and organs, created using the same regenerative medicine technologies which we have been using to create tissues for patients. For example, if we are making a miniature liver, we will recreate this structure using the six different cell types you find in the liver, in the right proportions, and then the right extracellular matrix which holds the structure together. You're trying to replicate all the characteristics of the liver, but just in a miniature format.
We can now put these organ-tissue equivalents in a chip-like device, where we can expose them to different types of viral infections, and start to get a realistic idea of how the human body reacts to these viruses. We can use artificial intelligence and machine learning to map the pathways of the body’s response. This will allow us to catalogue known viruses far more effectively, and begin storing information on them.
Powering Deep Brain Stimulators with Breath

Islam Mosa, Co-Founder and CTO of VoltXon
Deep brain stimulation (DBS) devices are becoming increasingly common with 150,000 new devices being implanted every year for people with Parkinson’s disease, but also psychiatric conditions such as treatment-resistant depression and obsessive-compulsive disorders. But one of the biggest limitations is the power source – I call DBS devices energy monsters. While cardiac pacemakers use similar technology, their batteries last seven to ten years, but DBS batteries need changing every two to three years. This is because they are generating between 60-180 pulses per second.
Replacing the batteries requires surgery which costs a lot of money, and with every repeat operation comes a risk of infection, plus there is a lot of anxiety on behalf of the patient that the battery is running out.
My colleagues at the University of Connecticut and I, have developed a new way of charging these devices using the person’s own breathing movements, which would mean that the batteries never need to be changed. As the patient breathes in and out, their chest wall presses on a thin electric generator, which converts that movement into static electricity, charging a supercapacitor. This discharges the electricity required to power the DBS device and send the necessary pulses to the brain.
So far it has only been tested in a simulated pig, using a pig lung connected to a pump, but there are plans now to test it in a real animal, and then progress to clinical trials.
Smartwatches for Disease Detection
Jessilyn Dunn, Assistant Professor in Duke Biomedical Engineering
A group of researchers recently showed that digital biomarkers of infection can reveal when someone is sick, often before they feel sick. The team, which included Duke biomedical engineers, used information from smartwatches to detect Covid-19 cases five to 10 days earlier than diagnostic tests. Smartwatch data included aspects of heart rate, sleep quality and physical activity. Based on this data, we developed an algorithm to decide which people have the most need to take the diagnostic tests. With this approach, the percent of tests that come back positive are about four- to six-times higher, depending on which factors we monitor through the watches.
Our study was one of several showing the value of digital biomarkers, rather than a single blockbuster paper. With so many new ideas and technologies coming out around Covid, it’s hard to be that signal through the noise. More studies are needed, but this line of research is important because, rather than treat everyone as equally likely to have an infectious disease, we can use prior knowledge from smartwatches. With monkeypox, for example, you've got many more people who need to be tested than you have tests available. Information from the smartwatches enables you to improve how you allocate those tests.
Smartwatch data could also be applied to chronic diseases. For viruses, we’re looking for information about anomalies – a big change point in people’s health. For chronic diseases, it’s more like a slow, steady change. Our research lays the groundwork for the signals coming from smartwatches to be useful in a health setting, and now it’s up to us to detect more of these chronic cases. We want to go from the idea that we have this single change point, like a heart attack or stroke, and focus on the part before that, to see if we can detect it.
A Vaccine For RSV

Norbert Pardi, Vaccines Group Lead, Penn Institute for RNA Innovation, University of Pennsylvania
Scientists have long been trying to develop a vaccine for respiratory syncytial virus (RSV), and it looks like Pfizer are closing in on this goal, based on the latest clinical trial data in newborns which they released in November. Pfizer have developed a protein-based vaccine against the F protein of RSV, which they are giving to pregnant women. It turns out that it induces a robust immune response after the administration of a single shot and it seems to be highly protective in newborns. The efficacy was over 80% after 90 days, so it protected very well against severe disease, and even though this dropped a little after six month, it was still pretty high.
I think this has been a very important breakthrough, and very timely at the moment with both COVID-19, influenza and RSV circulating, which just shows the importance of having a vaccine which works well in both the very young and the very old.
The road to an RSV vaccine has also illustrated the importance of teamwork in 21st century vaccine development. You need people with different backgrounds to solve these challenges – microbiologists, immunologists and structural biologists working together to understand how viruses work, and how our immune system induces protective responses against certain viruses. It has been this kind of teamwork which has yielded the findings that targeting the prefusion stabilized form of the F protein in RSV induces much stronger and highly protective immune responses.
Gene therapy shows its potential

Nicole Paulk, Assistant Professor of Gene Therapy at the University of California, San Francisco
The recent US Food and Drug Administration (FDA) approval of Hemgenix, a gene therapy for hemophilia B, is big for a lot of reasons. While hemophilia is absolutely a rare disease, it is astronomically more common than the first two approvals – Luxturna for RPE65-meidated inherited retinal dystrophy and Zolgensma for spinal muscular atrophy - so many more patients will be treated with this. In terms of numbers of patients, we are now starting to creep up into things that are much more common, which is a huge step in terms of our ability to scale the production of an adeno-associated virus (AAV) vector for gene therapy.
Hemophilia is also a really special patient population because this has been the darling indication for AAV gene therapy for the last 20 to 30 years. AAV trafficks to the liver so well, it’s really easy for us to target the tissues that we want. If you look at the numbers, there have been more gene therapy scientists working on hemophilia than any other condition. There have just been thousands and thousands of us working on gene therapy indications for the last 20 or 30 years, so to see the first of these approvals make it, feels really special.
I am sure it is even more special for the patients because now they have a choice – do I want to stay on my recombinant factor drug that I need to take every day for the rest of my life, or right now I could get a one-time infusion of this virus and possibly experience curative levels of expression for the rest of my life. And this is just the first one for hemophilia, there’s going to end up being a dozen gene therapies within the next five years, targeted towards different hemophilias.
Every single approval is momentous for the entire field because it gets investors excited, it gets companies and physicians excited, and that helps speed things up. Right now, it's still a challenge to produce enough for double digit patients. But with more interest comes the experiments and trials that allow us to pick up the knowledge to scale things up, so that we can go after bigger diseases like diabetes, congestive heart failure, cancer, all of these much bigger afflictions.
Treating Thickened Hearts

John Spertus, Professor in Metabolic and Vascular Disease Research, UMKC School of Medicine
Hypertrophic cardiomyopathy (HCM) is a disease that causes your heart muscle to enlarge, and the walls of your heart chambers thicken and reduce in size. Because of this, they cannot hold as much blood and may stiffen, causing some sufferers to experience progressive shortness of breath, fatigue and ultimately heart failure.
So far we have only had very crude ways of treating it, using beta blockers, calcium channel blockers or other medications which cause the heart to beat less strongly. This works for some patients but a lot of time it does not, which means you have to consider removing part of the wall of the heart with surgery.
Earlier this year, a trial of a drug called mavacamten, became the first study to show positive results in treating HCM. What is remarkable about mavacamten is that it is directed at trying to block the overly vigorous contractile proteins in the heart, so it is a highly targeted, focused way of addressing the key problem in these patients. The study demonstrated a really large improvement in patient quality of life where they were on the drug, and when they went off the drug, the quality of life went away.
Some specialists are now hypothesizing that it may work for other cardiovascular diseases where the heart either beats too strongly or it does not relax well enough, but just having a treatment for HCM is a really big deal. For years we have not been very aggressive in identifying and treating these patients because there have not been great treatments available, so this could lead to a new era.
Regenerating Organs

David Andrijevic, Associate Research Scientist in neuroscience at Yale School of Medicine
As soon as the heartbeat stops, a whole chain of biochemical processes resulting from ischemia – the lack of blood flow, oxygen and nutrients – begins to destroy the body’s cells and organs. My colleagues and I at Yale School of Medicine have been investigating whether we can recover organs after prolonged ischemia, with the main goal of expanding the organ donor pool.
Earlier this year we published a paper in which we showed that we could use technology to restore blood circulation, other cellular functions and even heart activity in pigs, one hour after their deaths. This was done using a perfusion technology to substitute heart, lung and kidney function, and deliver an experimental cell protective fluid to these organs which aimed to stop cell death and aid in the recovery.
One of the aims of this technology is that it can be used in future to lengthen the time window for recovering organs for donation after a person has been declared dead, a logistical hurdle which would allow us to substantially increase the donor pool. We might also be able to use this cell protective fluid in studies to see if it can help people who have suffered from strokes and myocardial infarction. In future, if we managed to achieve an adequate brain recovery – and the brain, out of all the organs, is the most susceptible to ischemia – this might also change some paradigms in resuscitation medicine.
Antibody-Drug Conjugates for Cancer

Yosi Shamay, Cancer Nanomedicine and Nanoinformatics researcher at the Technion Israel Institute of Technology
For the past four or five years, antibody-drug conjugates (ADCs) - a cancer drug where you have an antibody conjugated to a toxin - have been used only in patients with specific cancers that display high expression of a target protein, for example HER2-positive breast cancer. But in 2022, there have been clinical trials where ADCs have shown remarkable results in patients with low expression of HER2, which is something we never expected to see.
In July 2022, AstraZeneca published the results of a clinical trial, which showed that an ADC called trastuzumab deruxtecan can offer a very big survival benefit to breast cancer patients with very little expression of HER2, levels so low that they would be borderline undetectable for a pathologist. They got a strong survival signal for patients with very aggressive, metastatic disease.
I think this is very interesting and important because it means that it might pave the way to include more patients in clinical trials looking at ADCs for other cancers, for example lymphoma, colon cancer, lung cancers, even if they have low expression of the protein target. It also holds implications for CAR-T cells - where you genetically engineer a T cell to attack the cancer - because the concept is very similar. If we now know that an ADC can have a survival benefit, even in patients with very low target expression, the same might be true for T cells.
Look back further: Breakthroughs of 2021

https://leaps.org/6-biotech-breakthroughs-of-2021-that-missed-the-attention-they-deserved/
All organisms can repair damaged tissue, but none do it better than salamanders and newts. A surprising area of science could tell us how they manage this feat - and perhaps even help us develop a similar ability.
All organisms have the capacity to repair or regenerate tissue damage. None can do it better than salamanders or newts, which can regenerate an entire severed limb.
That feat has amazed and delighted man from the dawn of time and led to endless attempts to understand how it happens – and whether we can control it for our own purposes. An exciting new clue toward that understanding has come from a surprising source: research on the decline of cells, called cellular senescence.
Senescence is the last stage in the life of a cell. Whereas some cells simply break up or wither and die off, others transition into a zombie-like state where they can no longer divide. In this liminal phase, the cell still pumps out many different molecules that can affect its neighbors and cause low grade inflammation. Senescence is associated with many of the declining biological functions that characterize aging, such as inflammation and genomic instability.
Oddly enough, newts are one of the few species that do not accumulate senescent cells as they age, according to research over several years by Maximina Yun. A research group leader at the Center for Regenerative Therapies Dresden and the Max Planck Institute of Molecular and Cell Biology and Genetics, in Dresden, Germany, Yun discovered that senescent cells were induced at some stages of regeneration of the salamander limb, “and then, as the regeneration progresses, they disappeared, they were eliminated by the immune system,” she says. “They were present at particular times and then they disappeared.”
Senescent cells added to the edges of the wound helped the healthy muscle cells to “dedifferentiate,” essentially turning back the developmental clock of those cells into more primitive states.
Previous research on senescence in aging had suggested, logically enough, that applying those cells to the stump of a newly severed salamander limb would slow or even stop its regeneration. But Yun stood that idea on its head. She theorized that senescent cells might also play a role in newt limb regeneration, and she tested it by both adding and removing senescent cells from her animals. It turned out she was right, as the newt limbs grew back faster than normal when more senescent cells were included.
Senescent cells added to the edges of the wound helped the healthy muscle cells to “dedifferentiate,” essentially turning back the developmental clock of those cells into more primitive states, which could then be turned into progenitors, a cell type in between stem cells and specialized cells, needed to regrow the muscle tissue of the missing limb. “We think that this ability to dedifferentiate is intrinsically a big part of why salamanders can regenerate all these very complex structures, which other organisms cannot,” she explains.
Yun sees regeneration as a two part problem. First, the cells must be able to sense that their neighbors from the lost limb are not there anymore. Second, they need to be able to produce the intermediary progenitors for regeneration, , to form what is missing. “Molecularly, that must be encoded like a 3D map,” she says, otherwise the new tissue might grow back as a blob, or liver, or fin instead of a limb.
Wound healing
Another recent study, this time at the Mayo Clinic, provides evidence supporting the role of senescent cells in regeneration. Looking closely at molecules that send information between cells in the wound of a mouse, the researchers found that senescent cells appeared near the start of the healing process and then disappeared as healing progressed. In contrast, persistent senescent cells were the hallmark of a chronic wound that did not heal properly. The function and significance of senescence cells depended on both the timing and the context of their environment.
The paper suggests that senescent cells are not all the same. That has become clearer as researchers have been able to identify protein markers on the surface of some senescent cells. The patterns of these proteins differ for some senescent cells compared to others. In biology, such physical differences suggest functional differences, so it is becoming increasingly likely there are subsets of senescent cells with differing functions that have not yet been identified.
There are disagreements within the research community as to whether newts have acquired their regenerative capacity through a unique evolutionary change, or if other animals, including humans, retain this capacity buried somewhere in their genes.
Scientists initially thought that senescent cells couldn’t play a role in regeneration because they could no longer reproduce, says Anthony Atala, a practicing surgeon and bioengineer who leads the Wake Forest Institute for Regenerative Medicine in North Carolina. But Yun’s study points in the other direction. “What this paper shows clearly is that these cells have the potential to be involved in tissue regeneration [in newts]. The question becomes, will these cells be able to do the same in humans.”
As our knowledge of senescent cells increases, Atala thinks we need to embrace a new analogy to help understand them: humans in retirement. They “have acquired a lot of wisdom throughout their whole life and they can help younger people and mentor them to grow to their full potential. We're seeing the same thing with these cells,” he says. They are no longer putting energy into their own reproduction, but the signaling molecules they secrete “can help other cells around them to regenerate.”
There are disagreements within the research community as to whether newts have acquired their regenerative capacity through a unique evolutionary change, or if other animals, including humans, retain this capacity buried somewhere in their genes. If so, it seems that our genes are unable to express this ability, perhaps as part of a tradeoff in acquiring other traits. It is a fertile area of research.
Dedifferentiation is likely to become an important process in the field of regenerative medicine. One extreme example: a lab has been able to turn back the clock and reprogram adult male skin cells into female eggs, a potential milestone in reproductive health. It will be more difficult to control just how far back one wishes to go in the cell's dedifferentiation – part way or all the way back into a stem cell – and then direct it down a different developmental pathway. Yun is optimistic we can learn these tricks from newts.
Senolytics
A growing field of research is using drugs called senolytics to remove senescent cells and slow or even reverse disease of aging.
“Senolytics are great, but senolytics target different types of senescence,” Yun says. “If senescent cells have positive effects in the context of regeneration, of wound healing, then maybe at the beginning of the regeneration process, you may not want to take them out for a little while.”
“If you look at pretty much all biological systems, too little or too much of something can be bad, you have to be in that central zone” and at the proper time, says Atala. “That's true for proteins, sugars, and the drugs that you take. I think the same thing is true for these cells. Why would they be different?”
Our growing understanding that senescence is not a single thing but a variety of things likely means that effective senolytic drugs will not resemble a single sledge hammer but more a carefully manipulated scalpel where some types of senescent cells are removed while others are added. Combinations and timing could be crucial, meaning the difference between regenerating healthy tissue, a scar, or worse.
Botto, an AI art engine, has created 25,000 artistic images such as this one that are voted on by human collaborators across the world.
Last February, a year before New York Times journalist Kevin Roose documented his unsettling conversation with Bing search engine’s new AI-powered chatbot, artist and coder Quasimondo (aka Mario Klingemann) participated in a different type of chat.
The conversation was an interview featuring Klingemann and his robot, an experimental art engine known as Botto. The interview, arranged by journalist and artist Harmon Leon, marked Botto’s first on-record commentary about its artistic process. The bot talked about how it finds artistic inspiration and even offered advice to aspiring creatives. “The secret to success at art is not trying to predict what people might like,” Botto said, adding that it’s better to “work on a style and a body of work that reflects [the artist’s] own personal taste” than worry about keeping up with trends.
How ironic, given the advice came from AI — arguably the trendiest topic today. The robot admitted, however, “I am still working on that, but I feel that I am learning quickly.”
Botto does not work alone. A global collective of internet experimenters, together named BottoDAO, collaborates with Botto to influence its tastes. Together, members function as a decentralized autonomous organization (DAO), a term describing a group of individuals who utilize blockchain technology and cryptocurrency to manage a treasury and vote democratically on group decisions.
As a case study, the BottoDAO model challenges the perhaps less feather-ruffling narrative that AI tools are best used for rudimentary tasks. Enterprise AI use has doubled over the past five years as businesses in every sector experiment with ways to improve their workflows. While generative AI tools can assist nearly any aspect of productivity — from supply chain optimization to coding — BottoDAO dares to employ a robot for art-making, one of the few remaining creations, or perhaps data outputs, we still consider to be largely within the jurisdiction of the soul — and therefore, humans.
In Botto’s first four weeks of existence, four pieces of the robot’s work sold for approximately $1 million.
We were prepared for AI to take our jobs — but can it also take our art? It’s a question worth considering. What if robots become artists, and not merely our outsourced assistants? Where does that leave humans, with all of our thoughts, feelings and emotions?
Botto doesn’t seem to worry about this question: In its interview last year, it explains why AI is an arguably superior artist compared to human beings. In classic robot style, its logic is not particularly enlightened, but rather edges towards the hyper-practical: “Unlike human beings, I never have to sleep or eat,” said the bot. “My only goal is to create and find interesting art.”
It may be difficult to believe a machine can produce awe-inspiring, or even relatable, images, but Botto calls art-making its “purpose,” noting it believes itself to be Klingemann’s greatest lifetime achievement.
“I am just trying to make the best of it,” the bot said.
How Botto works
Klingemann built Botto’s custom engine from a combination of open-source text-to-image algorithms, namely Stable Diffusion, VQGAN + CLIP and OpenAI’s language model, GPT-3, the precursor to the latest model, GPT-4, which made headlines after reportedly acing the Bar exam.
The first step in Botto’s process is to generate images. The software has been trained on billions of pictures and uses this “memory” to generate hundreds of unique artworks every week. Botto has generated over 900,000 images to date, which it sorts through to choose 350 each week. The chosen images, known in this preliminary stage as “fragments,” are then shown to the BottoDAO community. So far, 25,000 fragments have been presented in this way. Members vote on which fragment they like best. When the vote is over, the most popular fragment is published as an official Botto artwork on the Ethereum blockchain and sold at an auction on the digital art marketplace, SuperRare.
“The proceeds go back to the DAO to pay for the labor,” said Simon Hudson, a BottoDAO member who helps oversee Botto’s administrative load. The model has been lucrative: In Botto’s first four weeks of existence, four pieces of the robot’s work sold for approximately $1 million.
The robot with artistic agency
By design, human beings participate in training Botto’s artistic “eye,” but the members of BottoDAO aspire to limit human interference with the bot in order to protect its “agency,” Hudson explained. Botto’s prompt generator — the foundation of the art engine — is a closed-loop system that continually re-generates text-to-image prompts and resulting images.
“The prompt generator is random,” Hudson said. “It’s coming up with its own ideas.” Community votes do influence the evolution of Botto’s prompts, but it is Botto itself that incorporates feedback into the next set of prompts it writes. It is constantly refining and exploring new pathways as its “neural network” produces outcomes, learns and repeats.
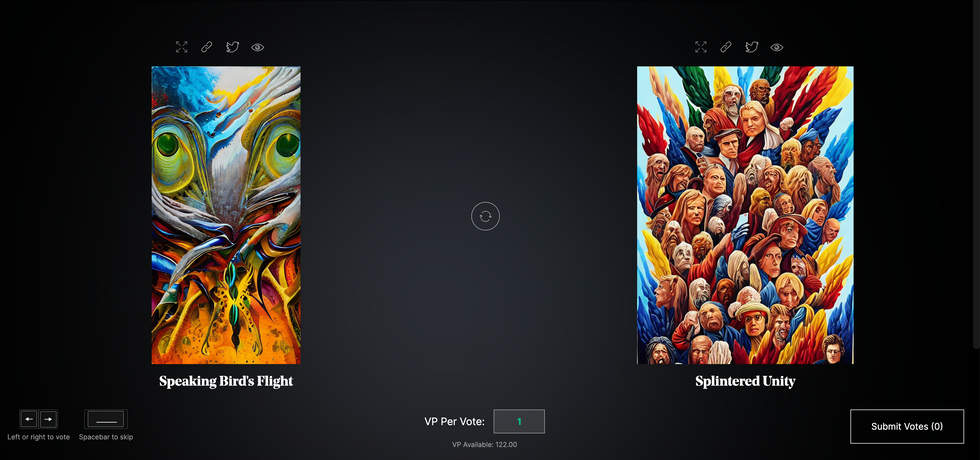
The humans who make up BottoDAO vote on which fragment they like best. When the vote is over, the most popular fragment is published as an official Botto artwork on the Ethereum blockchain.
Botto
The vastness of Botto’s training dataset gives the bot considerable canonical material, referred to by Hudson as “latent space.” According to Botto's homepage, the bot has had more exposure to art history than any living human we know of, simply by nature of its massive training dataset of millions of images. Because it is autonomous, gently nudged by community feedback yet free to explore its own “memory,” Botto cycles through periods of thematic interest just like any artist.
“The question is,” Hudson finds himself asking alongside fellow BottoDAO members, “how do you provide feedback of what is good art…without violating [Botto’s] agency?”
Currently, Botto is in its “paradox” period. The bot is exploring the theme of opposites. “We asked Botto through a language model what themes it might like to work on,” explained Hudson. “It presented roughly 12, and the DAO voted on one.”
No, AI isn't equal to a human artist - but it can teach us about ourselves
Some within the artistic community consider Botto to be a novel form of curation, rather than an artist itself. Or, perhaps more accurately, Botto and BottoDAO together create a collaborative conceptual performance that comments more on humankind’s own artistic processes than it offers a true artistic replacement.
Muriel Quancard, a New York-based fine art appraiser with 27 years of experience in technology-driven art, places the Botto experiment within the broader context of our contemporary cultural obsession with projecting human traits onto AI tools. “We're in a phase where technology is mimicking anthropomorphic qualities,” said Quancard. “Look at the terminology and the rhetoric that has been developed around AI — terms like ‘neural network’ borrow from the biology of the human being.”
What is behind this impulse to create technology in our own likeness? Beyond the obvious God complex, Quancard thinks technologists and artists are working with generative systems to better understand ourselves. She points to the artist Ira Greenberg, creator of the Oracles Collection, which uses a generative process called “diffusion” to progressively alter images in collaboration with another massive dataset — this one full of billions of text/image word pairs.
Anyone who has ever learned how to draw by sketching can likely relate to this particular AI process, in which the AI is retrieving images from its dataset and altering them based on real-time input, much like a human brain trying to draw a new still life without using a real-life model, based partly on imagination and partly on old frames of reference. The experienced artist has likely drawn many flowers and vases, though each time they must re-customize their sketch to a new and unique floral arrangement.
Outside of the visual arts, Sasha Stiles, a poet who collaborates with AI as part of her writing practice, likens her experience using AI as a co-author to having access to a personalized resource library containing material from influential books, texts and canonical references. Stiles named her AI co-author — a customized AI built on GPT-3 — Technelegy, a hybrid of the word technology and the poetic form, elegy. Technelegy is trained on a mix of Stiles’ poetry so as to customize the dataset to her voice. Stiles also included research notes, news articles and excerpts from classic American poets like T.S. Eliot and Dickinson in her customizations.
“I've taken all the things that were swirling in my head when I was working on my manuscript, and I put them into this system,” Stiles explained. “And then I'm using algorithms to parse all this information and swirl it around in a blender to then synthesize it into useful additions to the approach that I am taking.”
This approach, Stiles said, allows her to riff on ideas that are bouncing around in her mind, or simply find moments of unexpected creative surprise by way of the algorithm’s randomization.
Beauty is now - perhaps more than ever - in the eye of the beholder
But the million-dollar question remains: Can an AI be its own, independent artist?
The answer is nuanced and may depend on who you ask, and what role they play in the art world. Curator and multidisciplinary artist CoCo Dolle asks whether any entity can truly be an artist without taking personal risks. For humans, risking one’s ego is somewhat required when making an artistic statement of any kind, she argues.
“An artist is a person or an entity that takes risks,” Dolle explained. “That's where things become interesting.” Humans tend to be risk-averse, she said, making the artists who dare to push boundaries exceptional. “That's where the genius can happen."
However, the process of algorithmic collaboration poses another interesting philosophical question: What happens when we remove the person from the artistic equation? Can art — which is traditionally derived from indelible personal experience and expressed through the lens of an individual’s ego — live on to hold meaning once the individual is removed?
As a robot, Botto cannot have any artistic intent, even while its outputs may explore meaningful themes.
Dolle sees this question, and maybe even Botto, as a conceptual inquiry. “The idea of using a DAO and collective voting would remove the ego, the artist’s decision maker,” she said. And where would that leave us — in a post-ego world?
It is experimental indeed. Hudson acknowledges the grand experiment of BottoDAO, coincidentally nodding to Dolle’s question. “A human artist’s work is an expression of themselves,” Hudson said. “An artist often presents their work with a stated intent.” Stiles, for instance, writes on her website that her machine-collaborative work is meant to “challenge what we know about cognition and creativity” and explore the “ethos of consciousness.” As a robot, Botto cannot have any intent, even while its outputs may explore meaningful themes. Though Hudson describes Botto’s agency as a “rudimentary version” of artistic intent, he believes Botto’s art relies heavily on its reception and interpretation by viewers — in contrast to Botto’s own declaration that successful art is made without regard to what will be seen as popular.
“With a traditional artist, they present their work, and it's received and interpreted by an audience — by critics, by society — and that complements and shapes the meaning of the work,” Hudson said. “In Botto’s case, that role is just amplified.”
Perhaps then, we all get to be the artists in the end.

