How a Nobel-Prize Winner Fought Her Family, Nazis, and Bombs to Change our Understanding of Cells Forever

Rita Levi-Montalcini survived the Nazis and eventually won a Nobel Prize for her work to understand why certain cells grow so quickly.
When Rita Levi-Montalcini decided to become a scientist, she was determined that nothing would stand in her way. And from the beginning, that determination was put to the test. Before Levi-Montalcini became a Nobel Prize-winning neurobiologist, the first to discover and isolate a crucial chemical called Neural Growth Factor (NGF), she would have to battle both the sexism within her own family as well as the racism and fascism that was slowly engulfing her country
Levi-Montalcini was born to two loving parents in Turin, Italy at the turn of the 20th century. She and her twin sister, Paola, were the youngest of the family's four children, and Levi-Montalcini described her childhood as "filled with love and reciprocal devotion." But while her parents were loving, supportive and "highly cultured," her father refused to let his three daughters engage in any schooling beyond the basics. "He loved us and had a great respect for women," she later explained, "but he believed that a professional career would interfere with the duties of a wife and mother."
At age 20, Levi-Montalcini had finally had enough. "I realized that I could not possibly adjust to a feminine role as conceived by my father," she is quoted as saying, and asked his permission to finish high school and pursue a career in medicine. When her father reluctantly agreed, Levi-Montalcini was ecstatic: In just under a year, she managed to catch up on her mathematics, graduate high school, and enroll in medical school in Turin.
By 1936, Levi-Montalcini had graduated medical school at the top of her class and decided to stay on at the University of Turin as a research assistant for histologist and human anatomy professor Guiseppe Levi. Levi-Montalcini started studying nerve cells and nerve fibers – the tiny, slender tendrils that are threaded throughout our nerves and that determine what information each nerve can transmit. But it wasn't long before another enormous obstacle to her scientific career reared its head.
Science Under a Fascist Regime
Two years into her research assistant position, Levi-Montalcini was fired, along with every other "non-Aryan Italian" who held an academic or professional career, thanks to a series of antisemitic laws passed by Italy's then-leader Benito Mussolini. Forced out of her academic position, Levi-Montalcini went to Belgium for a fellowship at a neurological institute in Brussels – but then was forced back to Turin when the German army invaded.
Levi-Montalcini decided to keep researching. She and Guiseppe Levi built a makeshift lab in Levi-Montalcini's apartment, borrowing chicken eggs from local farmers and using sewing needles to dissect them. By dissecting the chicken embryos from her bedroom laboratory, she was able to see how nerve fibers formed and died. The two continued this research until they were interrupted again – this time, by British air raids. Levi-Montalcini fled to a country cottage to continue her research, and then two years later was forced into hiding when the German army invaded Italy. Levi-Montalcini and her family assumed different identities and lived with non-Jewish friends in Florence to survive the Holocaust. Despite all of this, Levi-Montalcini continued her work, dissecting chicken embryos from her hiding place until the end of the war.
"The discovery of NGF really changed the world in which we live, because now we knew that cells talk to other cells, and that they use soluble factors. It was hugely important."
A Post-War Discovery
Several years after the war, when Levi-Montalcini was once again working at the University of Turin, a German embryologist named Viktor Hamburger invited her to Washington University in St. Louis. Hamburger was impressed by Levi-Montalcini's research with her chicken embryos, and secured an opportunity for her to continue her work in America. The invitation would "change the course of my life," Levi-Montalcini would later recall.
During her fellowship, Montalcini grew tumors in mice and then transferred them to chick embryos in order to see how it would affect the chickens. To her surprise, she noticed that introducing the tumor samples would cause nerve fibers to grow rapidly. From this, Levi-Montalcini discovered and was able to isolate a protein that she determined was able to cause this rapid growth. She later named this Nerve Growth Factor, or NGF.
From there, Levi-Montalcini and her team launched new experiments to test NGF, injecting it and repressing it to see the effect it had in a test subject's body. When the team injected NGF into embryonic mice, they observed nerve growth, as well as the mouse pups developing faster – their eyes opening earlier and their teeth coming in sooner – than the untreated group. When the team purified the NGF extract, however, it had no effect, leading the team to believe that something else in the crude extract of NGF was influencing the growth of the newborn mice. Stanley Cohen, Levi-Montalcini's colleague, identified another growth factor called EGF – epidermal growth factor – that caused the mouse pups' eyes and teeth to grow so quickly.
Levi-Montalcini continued to experiment with NGF for the next several decades at Washington University, illuminating how NGF works in our body. When Levi-Montalcini injected newborn mice with an antiserum for NGF, for example, her team found that it "almost completely deprived the animals of a sympathetic nervous system." Other experiments done by Levi-Montalcini and her colleagues helped show the role that NGF plays in other important biological processes, such as the regulation of our immune system and ovulation.
"The discovery of NGF really changed the world in which we live, because now we knew that cells talk to other cells, and that they use soluble factors. It was hugely important," said Bill Mobley, Chair of the Department of Neurosciences at the University of California, San Diego School of Medicine.
Her Lasting Legacy
After years of setbacks, Levi-Montalcini's groundbreaking work was recognized in 1986, when she was awarded the Nobel Prize in Medicine for her discovery of NGF (Cohen, her colleague who discovered EGF, shared the prize). Researchers continue to study NGF even to this day, and the continued research has been able to increase our understanding of diseases like HIV and Alzheimer's.
Levi-Montalcini never stopped researching either: In January 2012, at the age of 102, Levi-Montalcini published her last research paper in the journal PNAS, making her the oldest member of the National Academy of Science to do so. Before she died in December 2012, she encouraged other scientists who would suffer setbacks in their careers to keep pursuing their passions. "Don't fear the difficult moments," Levi-Montalcini is quoted as saying. "The best comes from them."
A model of a human kidney.
Science's dream of creating perfect custom organs on demand as soon as a patient needs one is still a long way off. But tiny versions are already serving as useful research tools and stepping stones toward full-fledged replacements.
Although organoids cannot yet replace kidneys, they are invaluable tools for research.
The Lowdown
Australian researchers have grown hundreds of mini human kidneys in the past few years. Known as organoids, they function much like their full-grown counterparts, minus a few features due to a lack of blood supply.
Cultivated in a petri dish, these kidneys are still a shadow of their human counterparts. They grow no larger than one-sixth of an inch in diameter; fully developed organs are up to five inches in length. They contain no more than a few dozen nephrons, the kidney's individual blood-filtering unit, whereas a fully-grown kidney has about 1 million nephrons. And the dish variety live for just a few weeks.
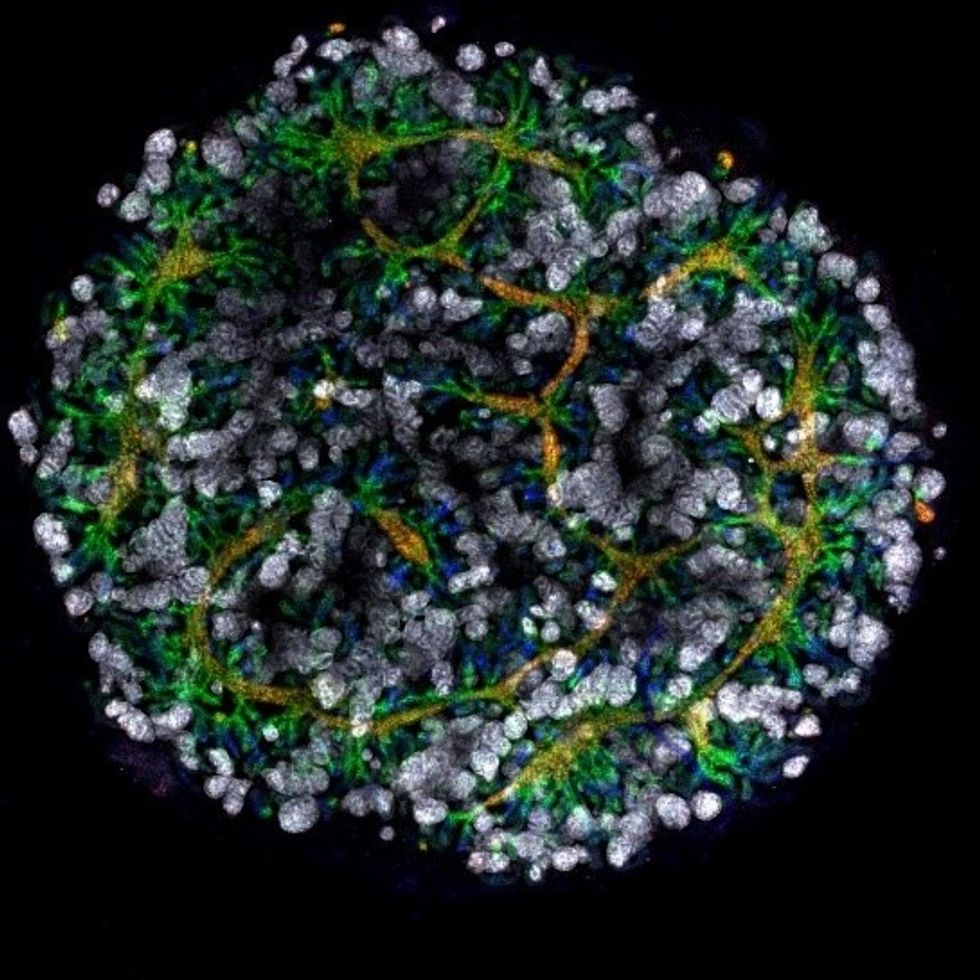
An organoid kidney created by the Murdoch Children's Institute in Melbourne, Australia.
Photo Credit: Shahnaz Khan.
But Melissa Little, head of the kidney research laboratory at the Murdoch Children's Institute in Melbourne, says these organoids are invaluable tools for research. Although renal failure is rare in children, more than half of those who suffer from such a disorder inherited it.
The mini kidneys enable scientists to better understand the progression of such disorders because they can be grown with a patient's specific genetic condition.
Mature stem cells can be extracted from a patient's blood sample and then reprogrammed to become like embryonic cells, able to turn into any type of cell in the body. It's akin to walking back the clock so that the cells regain unlimited potential for development. (The Japanese scientist who pioneered this technique was awarded the Nobel Prize in 2012.) These "induced pluripotent stem cells" can then be chemically coaxed to grow into mini kidneys that have the patient's genetic disorder.
"The (genetic) defects are quite clear in the organoids, and they can be monitored in the dish," Little says. To date, her research team has created organoids from 20 different stem cell lines.
Medication regimens can also be tested on the organoids, allowing specific tailoring for each patient. For now, such testing remains restricted to mice, but Little says it eventually will be done on human organoids so that the results can more accurately reflect how a given patient will respond to particular drugs.
Next Steps
Although these organoids cannot yet replace kidneys, Little says they may plug a huge gap in renal care by assisting in developing new treatments for chronic conditions. Currently, most patients with a serious kidney disorder see their options narrow to dialysis or organ transplantation. The former not only requires multiple sessions a week, but takes a huge toll on patient health.
Ten percent of older patients on dialysis die every year in the U.S. Aside from the physical trauma of organ transplantation, finding a suitable donor outside of a family member can be difficult.
"This is just another great example of the potential of pluripotent stem cells."
Meanwhile, the ongoing creation of organoids is supplying Little and her colleagues with enough information to create larger and more functional organs in the future. According to Little, researchers in the Netherlands, for example, have found that implanting organoids in mice leads to the creation of vascular growth, a potential pathway toward creating bigger and better kidneys.
And while Little acknowledges that creating a fully-formed custom organ is the ultimate goal, the mini organs are an important bridge step.
"This is just another great example of the potential of pluripotent stem cells, and I am just passionate to see it do some good."
Phil Gutis, an Alzheimer's patient who participated in a failed clinical trial, poses with his dog Abe.
Phil Gutis never had a stellar memory, but when he reached his early 50s, it became a problem he could no longer ignore. He had trouble calculating how much to tip after a meal, finding things he had just put on his desk, and understanding simple driving directions.
From 1998-2017, industry sources reported 146 failed attempts at developing Alzheimer's drugs.
So three years ago, at age 54, he answered an ad for a drug trial seeking people experiencing memory issues. He scored so low in the memory testing he was told something was wrong. M.R.I.s and PET scans confirmed that he had early-onset Alzheimer's disease.
Gutis, who is a former New York Times reporter and American Civil Liberties Union spokesman, felt fortunate to get into an advanced clinical trial of a new treatment for Alzheimer's disease. The drug, called aducanumab, had shown promising results in earlier studies.
Four years of data had found that the drug effectively reduced the burden of protein fragments called beta-amyloids, which destroy connections between nerve cells. Amyloid plaques are found in the brains of patients with Alzheimer's disease and are associated with impairments in thinking and memory.
Gutis eagerly participated in the clinical trial and received 35 monthly infusions. "For the first 20 infusions, I did not know whether I was receiving the drug or the placebo," he says. "During the last 15 months, I received aducanumab. But it really didn't matter if I was receiving the drug or the placebo because on March 21, the trial was stopped because [the drug company] Biogen found that the treatments were ineffective."
The news was devastating to the trial participants, but also to the Alzheimer's research community. Earlier this year, another pharmaceutical company, Roche, announced it was discontinuing two of its Alzheimer's clinical trials. From 1998-2017, industry sources reported 146 failed attempts at developing Alzheimer's drugs. There are five prescription drugs approved to treat its symptoms, but a cure remains elusive. The latest failures have left researchers scratching their heads about how to approach attacking the disease.
The failure of aducanumab was also another setback for the estimated 5.8 million people who have Alzheimer's in the United States. Of these, around 5.6 million are older than 65 and 200,000 suffer from the younger-onset form, including Gutis.
Gutis is understandably distraught about the cancellation of the trial. "I really had hopes it would work. So did all the patients."
While drug companies have failed so far, another group is stepping up to expedite the development of a cure: venture philanthropists.
For now, he is exercising every day to keep his blood flowing, which is supposed to delay the progression of the disease, and trying to eat a low-fat diet. "But I know that none of it will make a difference. Alzheimer's is a progressive disease. There are no treatments to delay it, let alone cure it."
But while drug companies have failed so far, another group is stepping up to expedite the development of a cure: venture philanthropists. These are successful titans of industry and dedicated foundations who are donating large sums of money to fill a much-needed void – funding research to look for new biomarkers.
Biomarkers are neurochemical indicators that can be used to detect the presence of a disease and objectively measure its progression. There are currently no validated biomarkers for Alzheimer's, but researchers are actively studying promising candidates. The hope is that they will find a reliable way to identify the disease even before the symptoms of mental decline show up, so that treatments can be directed at a very early stage.
Howard Fillit, Founding Executive Director and Chief Science Officer of the Alzheimer's Drug Discovery Foundation, says, "We need novel biomarkers to diagnose Alzheimer's disease and related dementias. But pharmaceutical companies don't put money into biomarkers research."
One of the venture philanthropists who has recently stepped up to the task is Bill Gates. In January 2018, he announced his father had Alzheimer's disease in an interview on the Today Show with Maria Shriver, whose father Sargent Shriver, died of Alzheimer's disease in 2011. Gates told Ms. Shriver that he had invested $100 million into Alzheimer's research, with $50 million of his donation going to Dementia Discovery Fund, which looks for new cures and treatments.
That August, Gates joined other investors in a new fund called Diagnostics Accelerator. The project aims to supports researchers looking to speed up new ideas for earlier and better diagnosis of the disease.
Gates and other donors committed more than $35 million to help launch it, and this April, Jeff and Mackenzie Bezos joined the coalition, bringing the current program funding to nearly $50 million.
"It makes sense that a challenge this significant would draw the attention of some of the world's leading thinkers."
None of these funders stand to make a profit on their donation, unlike traditional research investments by drug companies. The standard alternatives to such funding have upsides -- and downsides.
As Bill Gates wrote on his blog, "Investments from governments or charitable organizations are fantastic at generating new ideas and cutting-edge research -- but they're not always great at creating usable products, since no one stands to make a profit at the end of the day.
"Venture capital, on the other end of the spectrum, is more likely to develop a test that will reach patients, but its financial model favors projects that will earn big returns for investors. Venture philanthropy splits the difference. It incentivizes a bold, risk-taking approach to research with an end goal of a real product for real patients. If any of the projects backed by Diagnostics Accelerator succeed, our share of the financial windfall goes right back into the fund."
Gutis said he is thankful for any attention given to finding a cure for Alzheimer's.
"Most doctors and scientists will tell you that we're still in the dark ages when it comes to fully understanding how the brain works, let alone figuring out the cause or treatment for Alzheimer's.
"It makes sense that a challenge this significant would draw the attention of some of the world's leading thinkers. I only hope they can be more successful with their entrepreneurial approach to finding a cure than the drug companies have been with their more traditional paths."