How mRNA Could Revolutionize Medicine

In November 2020, messenger RNA catapulted into the public consciousness when the first COVID-19 vaccines were authorized for emergency use. Around the same time, an equally groundbreaking yet relatively unheralded application of mRNA technology was taking place at a London hospital.
Over the past two decades, there's been increasing interest in harnessing mRNA — molecules present in all of our cells that act like digital tape recorders, copying instructions from DNA in the cell nucleus and carrying them to the protein-making structures — to create a whole new class of therapeutics.
Scientists realized that artificial mRNA, designed in the lab, could be used to instruct our cells to produce certain antibodies, turning our bodies into vaccine-making factories, or to recognize and attack tumors. More recently, researchers recognized that mRNA could also be used to make another groundbreaking technology far more accessible to more patients: gene editing. The gene-editing tool CRISPR has generated plenty of hype for its potential to cure inherited diseases. But delivering CRISPR to the body is complicated and costly.
"Most gene editing involves taking cells out of the patient, treating them and then giving them back, which is an extremely expensive process," explains Drew Weissman, professor of medicine at the University of Pennsylvania, who was involved in developing the mRNA technology behind the COVID-19 vaccines.
But last November, a Massachusetts-based biotech company called Intellia Therapeutics showed it was possible to use mRNA to make the CRISPR system inside the body, eliminating the need to extract cells out of the body and edit them in a lab. Just as mRNA can instruct our cells to produce antibodies against a viral infection, it can also teach them to produce one of the two components that make up CRISPR — a cutting protein that snips out a problem gene.
"The pandemic has really shown that not only are mRNA approaches viable, they could in certain circumstances be vastly superior to more traditional technologies."
In Intellia's London-based clinical trial, the company applied this for the first time in a patient with a rare inherited liver disease known as hereditary transthyretin amyloidosis with polyneuropathy. The disease causes a toxic protein to build up in a person's organs and is typically fatal. In a company press release, Intellia's president and CEO John Leonard swiftly declared that its mRNA-based CRISPR therapy could usher in a "new era of potential genome editing cures."
Weissman predicts that turning CRISPR into an affordable therapy will become the next major frontier for mRNA over the coming decade. His lab is currently working on an mRNA-based CRISPR treatment for sickle cell disease. More than 300,000 babies are born with sickle cell every year, mainly in lower income nations.
"There is a FDA-approved cure, but it involves taking the bone marrow out of the person, and then giving it back which is prohibitively expensive," he says. It also requires a patient to have a matched bone marrow done. "We give an intravenous injection of mRNA lipid nanoparticles that target CRISPR to the bone marrow stem cells in the patient, which is easy, and much less expensive."
Cancer Immunotherapy
Meanwhile, the overwhelming success of the COVID-19 vaccines has focused attention on other ways of using mRNA to bolster the immune system against threats ranging from other infectious diseases to cancer.
The practicality of mRNA vaccines – relatively small quantities are required to induce an antibody response – coupled with their adaptable design, mean companies like Moderna are now targeting pathogens like Zika, chikungunya and cytomegalovirus, or CMV, which previously considered commercially unviable for vaccine developers. This is because outbreaks have been relatively sporadic, and these viruses mainly affect people in low-income nations who can't afford to pay premium prices for a vaccine. But mRNA technology means that jabs could be produced on a flexible basis, when required, at relatively low cost.
Other scientists suggest that mRNA could even provide a means of developing a universal influenza vaccine, a goal that's long been the Holy Grail for vaccinologists around the world.
"The mRNA technology allows you to pick out bits of the virus that you want to induce immunity to," says Michael Mulqueen, vice president of business development at eTheRNA, a Belgium-based biotech that's developing mRNA-based vaccines for malaria and HIV, as well as various forms of cancer. "This means you can get the immune system primed to the bits of the virus that don't vary so much between strains. So you could actually have a single vaccine that protects against a whole raft of different variants of the same virus, offering more universal coverage."
Before mRNA became synonymous with vaccines, its biggest potential was for cancer treatments. BioNTech, the German biotech company that collaborated with Pfizer to develop the first authorized COVID-19 vaccine, was initially founded to utilize mRNA for personalized cancer treatments, and the company remains interested in cancers ranging from melanoma to breast cancer.
One of the major hurdles in treating cancer has been the fact that tumors can look very different from one person to the next. It's why conventional approaches, such as chemotherapy or radiation, don't work for every patient. But weaponizing mRNA against cancer primes the immune cells with the tumor's specific genetic sequence, training the patient's body to attack their own unique type of cancer.
"It means you're able to think about personalizing cancer treatments down to specific subgroups of patients," says Mulqueen. "For example, eTheRNA are developing a renal cell carcinoma treatment which will be targeted at around 20% of these patients, who have specific tumor types. We're hoping to take that to human trials next year, but the challenge is trying to identify the right patients for the treatment at an early stage."
Repairing Damaged mRNA
While hopes are high that mRNA could usher in new cancer treatments and make CRISPR more accessible, a growing number of companies are also exploring an alternative to gene editing, known as RNA editing.
In genetic disorders, the mRNA in certain cells is impaired due to a rogue gene defect, and so the body ceases to produce a particular vital protein. Instead of permanently deleting the problem gene with CRISPR, the idea behind RNA editing is to inject small pieces of synthetic mRNA to repair the existing mRNA. Scientists think this approach will allow normal protein production to resume.
Over the past few years, this approach has gathered momentum, as some researchers have recognized that it holds certain key advantages over CRISPR. Companies from Belgium to Japan are now looking at RNA editing to treat all kinds of disorders, from Huntingdon's disease, to amyotrophic lateral sclerosis, or ALS, and certain types of cancer.
"With RNA editing, you don't need to make any changes to the DNA," explains Daniel de Boer, CEO of Dutch biotech ProQR, which is looking to treat rare genetic disorders that cause blindness. "Changes to the DNA are permanent, so if something goes wrong, that may not be desirable. With RNA editing, it's a temporary change, so we dose patients with our drugs once or twice a year."
Last month, ProQR reported a landmark case study, in which a patient with a rare form of blindness called Leber congenital amaurosis, which affects the retina at the back of the eye, recovered vision after three months of treatment.
"We have seen that this RNA therapy restores vision in people that were completely blind for a year or so," says de Boer. "They were able to see again, to read again. We think there are a large number of other genetic diseases we could go after with this technology. There are thousands of different mutations that can lead to blindness, and we think this technology can target approximately 25% of them."
Ultimately, there's likely to be a role for both RNA editing and CRISPR, depending on the disease. "I think CRISPR is ideally suited for illnesses where you would like to permanently correct a genetic defect," says Joshua Rosenthal of the Marine Biology Laboratory in Chicago. "Whereas RNA editing could be used to treat things like pain, where you might want to reset a neural circuit temporarily over a shorter period of time."
Much of this research has been accelerated by the COVID-19 pandemic, which has played a major role in bringing mRNA to the forefront of people's minds as a therapeutic.
"The pandemic has really shown that not only are mRNA approaches viable, they could in certain circumstances be vastly superior to more traditional technologies," says Mulqueen. "In the future, I would not be surprised if many of the top pharma products are mRNA derived."
How One University Is Successfully Tackling COVID-19
Alma Mater, a beloved bronze mother figure on the campus of the University of Illinois, Champaign-Urbana, wears a mask to encourage students to do the same as they return for the fall semester. Students are also expected to take a COVID-19 test twice a week.
China, South Korea and other places controlled the SARS-CoV-2 epidemic with the early use of strict lockdown and aggressive electronic contact tracing, monitoring, and enforcement.
The tussles in America over voluntary social distancing and wearing a mask in public suggest that more stringent enforcement methods adopted elsewhere would not work here. But one American university has emerged as a model of tough love pandemic management.
While many universities have become hot spots of COVID-19 infections this fall when students returned to campus, the University of Illinois was an exception. It has gotten the virus under control, at least for the moment, at a rate that is far below the national average and with minimal social disruption. Can the program they implemented work in our broader society?
The Illinois model is a comprehensive one which, as elsewhere, includes masking and social distancing, but it also requires a twice-weekly saliva test for SARS-CoV-2. All students and employees are assigned test days when they swipe their ID card and spit in a plastic tube, which is collected hourly and taken to a campus lab.
There a simplified but highly sensitive PCR genetic test goes through many cycles of amplifying the viral RNA. "Tracking three different viral RNA [genes] gives us very high accuracy," explains Martin Burke, the professor who developed the system and is monitoring its implementation at the University of Illinois Urbana-Champaign. They immediately retest any positive sample to confirm the results, "So we think our false positive rate is extremely low. … The goal is to notify the positive person within 30 minutes of a positive test results becoming known."
Testing everyone so frequently, with a sensitive test that can quickly detect small amounts of the virus soon after infection, and isolating those who test positive before the virus can grow to volumes that make it very infectious helps the Illinois system break the chain of transmission.
"The testing we have done is not a silver bullet, it has to be done in combination with other mitigation measures. Our modeling shows that if you have masks, social distancing, and contact tracing you get a very dramatic, in fact synergistic effect with this combination,' says Burke. "So it really has to be a holistic approach with lots of community engagement in order to make this process successful."
The real teeth of enforcement are that people have to display their health status to gain access to campus facilities. A green check mark over their photo on a college ID phone app means they are good to go but a big red X means they are not current on their testing or have tested positive for the virus. Their ID is inactivated and they cannot enter campus facilities until they become compliant. Burke puts it bluntly; "We stop them from going where they want to go, a measure first used successfully with the pandemic in Wuhan, China.
He says they have learned from their experience and evolved their approach. "We never modeled for people who tested positive to ignore that result and go to or host parties, which could spread the infection." But several students did just that, and a few have been suspended for it.
So the university clamped down on enforcing isolation and now requires some higher risk persons to test three times a week to catch any infections earlier. Since more than 95 percent of new infections were among undergraduates, with no crossover from them to the local community, faculty, or graduate students, they have cut back testing of the latter two groups to just once a week.
About a thousand positive tests results have come back so far but no one has been hospitalized. Part of that likely is because the undergraduate population is largely young and healthy with few risk cofactors. But it may also be that with early identification and isolation, about five percent of dorm rooms have been set aside for that purpose, the person adopts healthier patterns of sleeping and eating that allows the immune system to better fight off the virus.
"But when you compare that to the being able to educate our students, perform research, keep our community thriving, our businesses open, if you add it all up, it's a tremendous return on investment."
The logistics are quite impressive for the campus that in ordinary times is home to more than 50,000 students; a lab capable of churning through 20,000 tests a day, with notification of results within hours, not days as is common elsewhere. And the results are equally impressive. The rate of positive test results blipped up to around 3 percent when undergraduates arrived back on campus but that has plummeted to 0.35 percent for the last seven-day period of testing, a tiny fraction of the rate for the nation as a whole. Much of it can be attributed to the closed environment with limited outside contact that might reintroduce the virus.
Still, even while the campus population has dropped by about a third, they are detecting about 250 new infections a week.
The threat of outside contact adding to the risk is why the university amended the undergraduate school calendar to close for Thanksgiving, hold final classes and exams for the semester online, and not return until February.
It doesn't come cheap. Burke estimates it cost $10 million to set up the program and about the same each semester to operate. "But when you compare that to the being able to educate our students, perform research, keep our community thriving, our businesses open, if you add it all up, it's a tremendous return on investment."
Burke acknowledges that they started with some significant advantages. The community is geographically isolated, an electronically linked ID system was already in place for students and employees, they have the ability to control much activity through access to buildings, and they can expel those who do not conform. He believes their system can translate to similar settings but admits, "A big city is very different from a university community." Still, he believes many of those lessons can be translated to different settings.
An alternative story
However, the situation is very different at the University of Colorado, where new infections have surged since undergraduates returned in late August. Administrators recently switched all classes to online only in an attempt to control the virus.
But that wasn't enough for state authorities who cracked down further, just yesterday declaring a two-week lockdown of all students aged 18 to 22, prohibiting gatherings of any size, indoors or out. Students must stay in their rooms except for essential activities, and if any symptoms develop, report for testing. Fraternities and sororities were targeted as past hot spots of infection.
The police will be actively enforcing the lockdown, and violators can face a penalty of up to 90 days in jail and a $1,000 fine.
Skepticism
Public health largely is based upon an appeal to self-interest and altruism, and voluntary compliance with official guidance. Harm reduction often comes into play when an ideal solution meets resistance and coercion plays only a limited role, as when a person with infectious tuberculosis is not compliant with treatment. Many question whether the medical threat of COVID-19 justifies such a sweeping restriction of individual rights of movement and association imposed on everyone simply because of their age and place of residence as is happening in Colorado.
State and federal courts have begun to strike down as an unconstitutional overreach some of the more restrictive decrees to stay at home or close businesses ordered by state and local officials. What was once tolerated as a few weeks or even a few months of restrictions now seems to stretch without an end in sight, and threatens peoples' livelihoods. In this litigious country it seems only a matter of time before someone will challenge some aspects of the Illinois model or similar programs being set up elsewhere as an infringement of their rights.
"I have real concerns about what we have seen over the course of the past several months in terms of going from not enough testing being available to now having more testing [available] because people don't want to be tested, even when they have symptoms," says Michael Osterholm, a noted expert on pandemic preparedness at the University of Minnesota. "We have some college campuses reporting over fifty percent of the students refusing to be tested or refusing to give any of the contacts that might be followed up on."
Often those who have tested positive for the virus "don't want people to know that they're the potential reason there could be an outbreak in their small social circle," says LaQuandra Nesbitt, public health director for Washington, DC. Stigma is one of the main reasons why only 37% of newly infected people have provided names for contact tracing in D.C., and few offer more than a single name.
"We can't test every single person every single day, we would completely go broke, we would be looking at no other health problems. We're not the NFL," says Monica Gandhi. She is a professor of medicine at the University of California San Francisco and works closely with local health officials. "Just because we have a technology doesn't mean that we have to apply it for every purpose that may be indicated. … We would never dream of mass screening the public for influenza."
"Tests don't solve the problem," she argues. Masking is the most crucial piece for Gandhi, along with social distancing, washing hands regularly, and quarantine when testing positive or in contact with someone who is. Those are the actions that break the ongoing spread of transmission. She does support regular testing in high-risk settings such as nursing homes, inpatients in hospitals, and prisons, and periodic surveys in the general population to better understand where the virus is moving.
Drawing from experience with HIV, Gandhi worries that the stigma of a positive result will drive people away from testing. "Low-income persons will be particularly hesitant to get tested, or to share contact information if they do test positive, if they think they may have to quarantine, not work or gain income." That is why San Francisco initially assisted people in isolation with payment of $1285 for two weeks of isolation and other support as part of a right to health program. And this fall, the State of California passed legislation requiring that large businesses continue to pay employees in quarantine.
Tools for self-protection
The American temperament, decentralization, size, administrative complexity, and sheer cost make it highly unlikely that a coercive one-size-fits-all Illinois approach will ever be rolled out from a university campus to the entire nation. People make different decisions in trading off between safety and personal freedom or autonomy, and many are likely to embrace a rapid, inexpensive self-test if one becomes available, much like a home pregnancy test, to proactively monitor their own health.
OraSure Technologies pioneered the first home test for HIV. It is the only over-the-counter saliva test for HIV approved for sale in the U.S. Results show in about 20 minutes. The company went on to develop versions of this test for hepatitis C and Ebola. Thus it came as no surprise when in April the Department of Health and Human Services awarded it a $710 thousand contract to develop a rapid antigen home test for SARS-CoV-2.
Initial optimization studies for the antigen test showed that a nasal sample rather than an oral one generated better results, OraSure president and CEO Stephen Tang told LeapsMag. A test using a nasal swab is expected to be available later this year while work continues to develop an antibody test that uses saliva. He says, "the fundamental challenge is not only to develop the tests but to get it to scale quickly. That's the only way it's really going to matter." The company has manufacturing capacity to produce 35 million tests a year, with about half for SARS-CoV-2, and will double that capacity in steps within the next twelve months, with all of the increased capacity dedicated to COVID-19.
Initial use will be limited to health care workers and by prescription, but the company hopes to make it available over the counter soon after the FDA finalizes its rules on these types of tests for COVID-19. Importantly, OraSure believes its nasal swab test will be able to meet the current FDA standards for at-home tests. No such tests have yet been approved.
Tang says they envision using a phone app with the test, but that's tied to "the question of our century; who owns the data? If you are an individual buying the test, are you really compelled to report to anybody? If you are an employer and you buy the test and your employees take it, are you then entitled to the information because you're the one administering the test? That's all still being debated as well" by regulators, lawyers, and ethicists.
The price hasn't been set but Tang notes that they have "vast experience" in selling directly to the consumer, physicians, and public health systems in the U.S. and in lower-income companies. "We are very aware of what the economics are and what the need is today. We're trying to make this product as widely available to as many people as possible."
Another tool that may help protect the self-motivated are cell phone apps that alert you to potential exposure to others with the virus. Apple, Google and others have developed versions of the app that all work on the same principle and, miraculously, are compatible between the Apple and Android operating system universes. At first glance they look promising.
The glitch is that where they have been available the longest, only about 15-20 percent of users bother to download it, says Bennett Cyphers, a staff technologist with the Electronic Freedom Foundation (EFF), a nonprofit that advocates for privacy and other concerns in cyberspace. He explains, "If 1 in 10 people have the app installed, then only 1 in 100 interactions between everyone is going to be captured by the app. It scales that way; the fewer people you have, then a really, really small fraction of contacts are actually detected."
It is important to remember that much of public health is not the result of policy but of what people do in their daily lives.
Importantly, about 20 percent of Americans do not own a smart phone with the capacity to handle the app; that percentage is even higher among lower income, less educated, older folks who often are most at risk for suffering a severe case of COVID-19. So the value of this tool is likely to remain largely theoretical.
Divining the future
"It's tough to make predictions, especially about the future," the great baseball sage Yogi Berra is reported to have said. Will the COVID-19 pandemic in the U.S. follow the path of Illinois or Colorado?
The recent past often is no guide to such predictions. France, Spain, and Israel once earned plaudits for early and strict enforcement of lockdowns to control spread of the virus and then eased up on those restrictions. At the same time the world watched with condemnation and fascination as Sweden chose to follow a more laissez faire approach, urging voluntary distancing and masking but no major curtailing of activity.
Today the rates of new infections of COVID-19 in the first three countries have exploded to equal or multiples of the rate in Sweden. Which approach was the correct policy? Most people say it is still too early to tell for sure. The same can be said for the examples of Illinois and Colorado.
And then there is the puzzling example of Manaus, the Brazilian city of 1.8 million in the middle of the Amazon which was slammed with infections as hard as New York City; without the medical infrastructure to cope with the virus, 4000 have died. But then, suddenly, new infections began to taper off, and nobody claims to understand why, it certainly wasn't because official policies changed. One guess is that perhaps the region reached herd immunity, but that is simply speculation.
One can pick and choose examples of tough enforcement of quarantine or none to prove their point for the short term. But draconian measures will not be tolerated for long in a free society, and there is no clear, overwhelming evidence that over the long run one policy approach works better than another.
It is important to remember that much of public health is not the result of policy but of what people do in their daily lives. We have come remarkably far in what is still only months since we first heard the name of the virus. Death rates have fallen dramatically as we have learned how to better manage severe disease, often by adapting treatments for other diseases. And there is reason for optimism with the large number of vaccine candidates already in human trials.
We also have learned that we can control much of our own fate through simple but concerted actions in our daily lives such as social distancing, wearing masks, and washing hands. Let's not only remember those facts, but practice them.
Artificial Wombs Are Getting Closer to Reality for Premature Babies
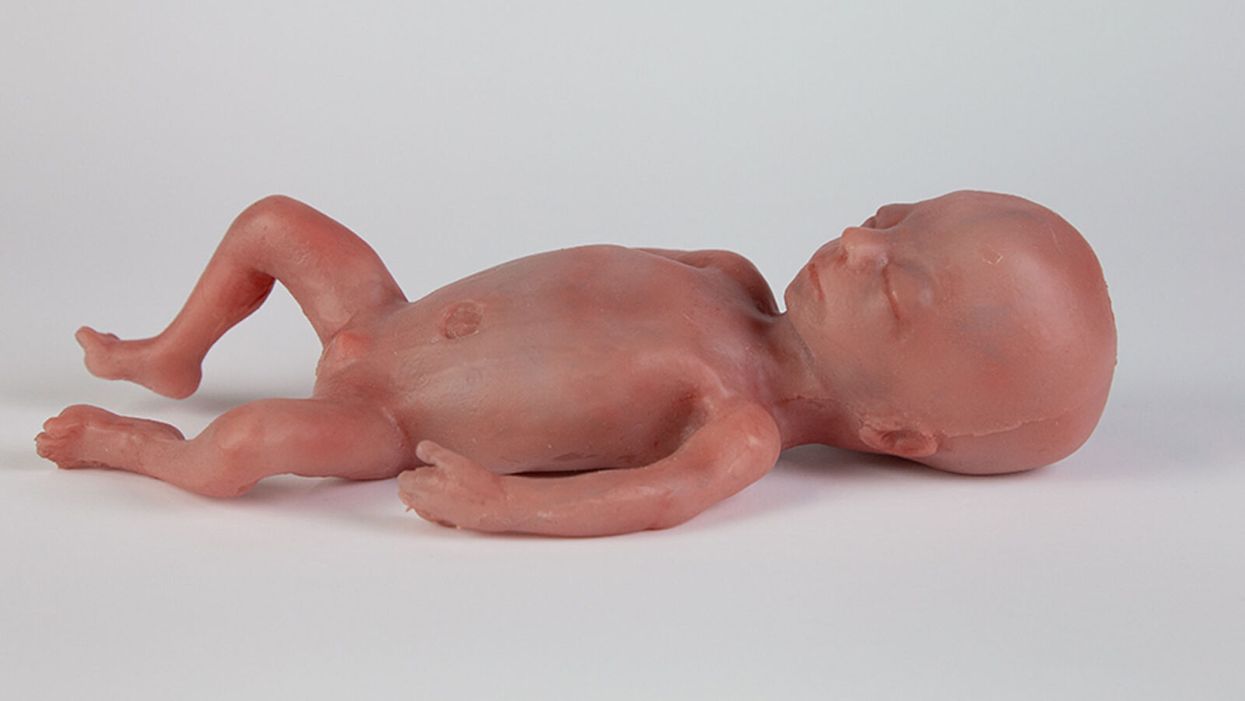
A mannequin of a 24-week-old fetus replicated from MR imaging. Created by: Juliette van Haren, Mark Thielen, Jasper Sterk, Chet Bangaru, and Frank Delbressine, Department of Industrial Design, Eindhoven University of Technology.
In 2017, researchers at the Children's Hospital of Philadelphia grew extremely preterm lambs from hairless to fluffy inside a "biobag," a dark, fluid-filled bag designed to mimic a mother's womb.
"There could be quite a lot of infants that would benefit from artificial womb technologies."
This happened over the course of a month, across a delicate period of fetal development that scientists consider the "edge of viability" for survival at birth.
In 2019, Australian and Japanese scientists repeated the success of keeping extremely premature lambs inside an artificial womb environment until they were ready to survive on their own. Those researchers are now developing a treatment strategy for infants born at "the hard limit of viability," between 20 and 23 weeks of gestation. At the same time, Dutch researchers are going so far as to replicate the sound of a mother's heartbeat inside a biobag. These developments signal exciting times ahead--with a touch of science fiction--for artificial womb technologies. But is there a catch?
"There could be quite a lot of infants that would benefit from artificial womb technologies," says Josephine Johnston, a bioethicist and lawyer at The Hastings Center, an independent bioethics research institute in New York. "These technologies can decrease morbidity and mortality for infants at the edge of viability and help them survive without significant damage to the lungs or other problems," she says.
It is a viewpoint shared by Frans van de Vosse, leader of the Cardiovascular Biomechanics research group at Eindhoven University of Technology in the Netherlands. He participates in a university project that recently received more than $3 million in funding from the E.U. to produce a prototype artificial womb for preterm babies between 24 and 28 weeks of gestation by 2024.
The Eindhoven design comes with a fluid-based environment, just like that of the natural womb, where the baby receives oxygen and nutrients through an artificial placenta that is connected to the baby's umbilical cord. "With current incubators, when a respiratory device delivers oxygen into the lungs in order for the baby to breathe, you may harm preterm babies because their lungs are not yet mature for that," says van de Vosse. "But when the lungs are under water, then they can develop, they can mature, and the baby will receive the oxygen through the umbilical cord, just like in the natural womb," he says.
His research team is working to achieve the "perfectly natural" artificial womb based on strict mathematical models and calculations, van de Vosse says. They are even employing 3D printing technology to develop the wombs and artificial babies to test in them--the mannequins, as van de Vosse calls them. These mannequins are being outfitted with sensors that can replicate the environment a fetus experiences inside a mother's womb, including the soothing sound of her heartbeat.
"The Dutch study's artificial womb design is slightly different from everything else we have seen as it encourages a gestateling to experience the kind of intimacy that a fetus does in pregnancy," says Elizabeth Chloe Romanis, an assistant professor in biolaw at Durham Law School in the U.K. But what is a "gestateling" anyway? It's a term Romanis has coined to describe neither a fetus nor a newborn, but an in-between artificial stage.
"Because they aren't born, they are not neonates," Romanis explains. "But also, they are not inside a pregnant person's body, so they are not fetuses. In an artificial womb the fetus is still gestating, hence why I call it gestateling."
The terminology is not just a semantic exercise to lend a name to what medical dictionaries haven't yet defined. "Gestatelings might have a slightly different psychology," says Romanis. "A fetus inside a mother's womb interacts with the mother. A neonate has some kind of self-sufficiency in terms of physiology. But the gestateling doesn't do either of those things," she says, urging us to be mindful of the still-obscure effects that experiencing early life as a gestateling might have on future humans. Psychology aside, there are also legal repercussions.
The Universal Declaration of Human Rights proclaims the "inalienable rights which everyone is entitled to as a human being," with "everyone" including neonates. However, such a legal umbrella is absent when it comes to fetuses, which have no rights under the same declaration. "We might need a new legal category for a gestateling," concludes Romanis.
But not everyone agrees. "However well-meaning, a new legal category would almost certainly be used to further erode the legality of abortion in countries like the U.S.," says Johnston.
The "abortion war" in the U.S. has risen to a crescendo since 2019, when states like Missouri, Mississippi, Kentucky, Louisiana and Georgia passed so-called "fetal heartbeat bills," which render an abortion illegal once a fetal heartbeat is detected. The situation is only bound to intensify now that Justice Ruth Bader Ginsburg, one of the Supreme Court's fiercest champions for abortion rights, has passed away. If President Trump appoints Ginsburg's replacement, he will probably grant conservatives on the Court the votes needed to revoke or weaken Roe v. Wade, the milestone decision of 1973 that established women's legal right to an abortion.
"A gestateling with intermediate status would almost certainly be considered by some in the U.S. (including some judges) to have at least certain legal rights, likely including right-to-life," says Johnston. This would enable a fetus on the edge of viability to make claims on the mother, and lead either to a shortening of the window in which abortion is legal—or a practice of denying abortion altogether. Instead, Johnston predicts, doctors might offer to transfer the fetus to an artificial womb for external gestation as a new standard of care.
But the legal conundrum does not stop there. The viability threshold is an estimate decided by medical professionals based on the clinical evidence and the technology available. It is anything but static. In the 1970s when Roe v. Wade was decided, for example, a fetus was considered legally viable starting at 28 weeks. Now, with improved technology and medical management, "the hard limit today is probably 20 or 21 weeks," says Matthew Kemp, associate professor at the University of Western Australia and one of the Australian-Japanese artificial womb project's senior researchers.
The changing threshold can result in situations where lots of people invested in the decision disagree. "Those can be hard decisions, but they are case-by-case decisions that families make or parents make with the key providers to determine when to proceed and when to let the infant die. Usually, it's a shared decision where the parents have the final say," says Johnston. But this isn't always the case.
On May 9th 2016, a boy named Alfie Evans was born in Liverpool, UK. Suffering seizures a few months after his birth, Alfie was diagnosed with an unknown neurodegenerative disorder and soon went into a semi-vegetative state, which lasted for more than a year. Alfie's medical team decided to withdraw his ventilation support, suggesting further treatment was unlawful and inhumane, but his parents wanted permission to fly him to a hospital in Rome and attempt to prolong his life there. In the end, the case went all the way up to the Supreme Court, which ruled that doctors could stop providing life support for Alfie, saying that the child required "peace, quiet and privacy." What happened to little Alfie raised huge publicity in the UK and pointedly highlighted the dilemma of whether parents or doctors should have the final say in the fate of a terminally-ill child in life-support treatment.
"In a few years from now, women who cannot get pregnant because of uterine infertility will be able to have a fully functional uterus made from their own tissue."
Alfie was born and, thus had legal rights, yet legal and ethical mayhem arose out of his case. When it comes to gestatelings, the scenarios will be even more complicated, says Romanis. "I think there's a really big question about who has parental rights and who doesn't," she says. "The assisted reproductive technology (ART) law in the U.K. hasn't been updated since 2008....It certainly needs an update when you think about all the things we have done since [then]."
This June, for instance, scientists from the Wake Forest Institute for Regenerative Medicine in North Carolina published research showing that they could take a small sample of tissue from a rabbit's uterus and create a bioengineered uterus, which then supported both fertilization and normal pregnancy like a natural uterus does.
"In [a number of] years from now, women who cannot get pregnant because of uterine infertility will be able to have a fully functional uterus made from their own tissue," says Dr. Anthony Atala, the Institute's director and a pioneer in regenerative medicine. These bioengineered uteri will eventually be covered by insurance, Atala expects. But when it comes to artificial wombs that externally gestate premature infants, will all mothers have equal access?
Medical reports have already shown racial and ethnic disparities in infertility treatments and access to assisted reproductive technologies. Costs on average total $12,400 per cycle of treatment and may require several cycles to achieve a live birth. "There's no indication that artificial wombs would be treated any differently. That's what we see with almost every expensive new medical technology," says Johnston. In a much more dystopian future, there is even a possibility that inequity in healthcare might create disturbing chasms in how women of various class levels bear children. Romanis asks us to picture the following scenario:
We live in a world where artificial wombs have become mainstream. Most women choose to end their pregnancies early and transfer their gestatelings to the care of machines. After a while, insurers deem full-term pregnancy and childbirth a risky non-necessity, and are lobbying to stop covering them altogether. Wealthy white women continue opting out of their third trimesters (at a high cost), since natural pregnancy has become a substandard route for poorer women. Those women are strongly judged for any behaviors that could risk their fetus's health, in contrast with the machine's controlled environment. "Why are you having a coffee during your pregnancy?" critics might ask. "Why are you having a glass of red wine? If you can't be perfect, why don't you have it the artificial way?"
Problem is, even if they want to, they won't be able to afford it.
In a more sanguine version, however, the artificial wombs are only used in cases of prematurity as a life-saving medical intervention rather than as a lifestyle accommodation. The 15 million babies who are born prematurely each year and may face serious respiratory, cardiovascular, visual and hearing problems, as well as learning disabilities, instead continue their normal development in artificial wombs. After lots of deliberation, insurers agree to bear the cost of external wombs because they are cheaper than a lifetime of medical care for a disabled or diseased person. This enables racial and ethnic minority women, who make up the majority of women giving premature birth, to access the technology.
Even extremely premature babies, those babies (far) below the threshold of 28 weeks of gestation, half of which die, could now discover this thing called life. In this scenario, as the Australian researcher Kemp says, we are simply giving a good shot at healthy, long-term survival to those who were unfortunate enough to start too soon.