How mRNA Could Revolutionize Medicine

In November 2020, messenger RNA catapulted into the public consciousness when the first COVID-19 vaccines were authorized for emergency use. Around the same time, an equally groundbreaking yet relatively unheralded application of mRNA technology was taking place at a London hospital.
Over the past two decades, there's been increasing interest in harnessing mRNA — molecules present in all of our cells that act like digital tape recorders, copying instructions from DNA in the cell nucleus and carrying them to the protein-making structures — to create a whole new class of therapeutics.
Scientists realized that artificial mRNA, designed in the lab, could be used to instruct our cells to produce certain antibodies, turning our bodies into vaccine-making factories, or to recognize and attack tumors. More recently, researchers recognized that mRNA could also be used to make another groundbreaking technology far more accessible to more patients: gene editing. The gene-editing tool CRISPR has generated plenty of hype for its potential to cure inherited diseases. But delivering CRISPR to the body is complicated and costly.
"Most gene editing involves taking cells out of the patient, treating them and then giving them back, which is an extremely expensive process," explains Drew Weissman, professor of medicine at the University of Pennsylvania, who was involved in developing the mRNA technology behind the COVID-19 vaccines.
But last November, a Massachusetts-based biotech company called Intellia Therapeutics showed it was possible to use mRNA to make the CRISPR system inside the body, eliminating the need to extract cells out of the body and edit them in a lab. Just as mRNA can instruct our cells to produce antibodies against a viral infection, it can also teach them to produce one of the two components that make up CRISPR — a cutting protein that snips out a problem gene.
"The pandemic has really shown that not only are mRNA approaches viable, they could in certain circumstances be vastly superior to more traditional technologies."
In Intellia's London-based clinical trial, the company applied this for the first time in a patient with a rare inherited liver disease known as hereditary transthyretin amyloidosis with polyneuropathy. The disease causes a toxic protein to build up in a person's organs and is typically fatal. In a company press release, Intellia's president and CEO John Leonard swiftly declared that its mRNA-based CRISPR therapy could usher in a "new era of potential genome editing cures."
Weissman predicts that turning CRISPR into an affordable therapy will become the next major frontier for mRNA over the coming decade. His lab is currently working on an mRNA-based CRISPR treatment for sickle cell disease. More than 300,000 babies are born with sickle cell every year, mainly in lower income nations.
"There is a FDA-approved cure, but it involves taking the bone marrow out of the person, and then giving it back which is prohibitively expensive," he says. It also requires a patient to have a matched bone marrow done. "We give an intravenous injection of mRNA lipid nanoparticles that target CRISPR to the bone marrow stem cells in the patient, which is easy, and much less expensive."
Cancer Immunotherapy
Meanwhile, the overwhelming success of the COVID-19 vaccines has focused attention on other ways of using mRNA to bolster the immune system against threats ranging from other infectious diseases to cancer.
The practicality of mRNA vaccines – relatively small quantities are required to induce an antibody response – coupled with their adaptable design, mean companies like Moderna are now targeting pathogens like Zika, chikungunya and cytomegalovirus, or CMV, which previously considered commercially unviable for vaccine developers. This is because outbreaks have been relatively sporadic, and these viruses mainly affect people in low-income nations who can't afford to pay premium prices for a vaccine. But mRNA technology means that jabs could be produced on a flexible basis, when required, at relatively low cost.
Other scientists suggest that mRNA could even provide a means of developing a universal influenza vaccine, a goal that's long been the Holy Grail for vaccinologists around the world.
"The mRNA technology allows you to pick out bits of the virus that you want to induce immunity to," says Michael Mulqueen, vice president of business development at eTheRNA, a Belgium-based biotech that's developing mRNA-based vaccines for malaria and HIV, as well as various forms of cancer. "This means you can get the immune system primed to the bits of the virus that don't vary so much between strains. So you could actually have a single vaccine that protects against a whole raft of different variants of the same virus, offering more universal coverage."
Before mRNA became synonymous with vaccines, its biggest potential was for cancer treatments. BioNTech, the German biotech company that collaborated with Pfizer to develop the first authorized COVID-19 vaccine, was initially founded to utilize mRNA for personalized cancer treatments, and the company remains interested in cancers ranging from melanoma to breast cancer.
One of the major hurdles in treating cancer has been the fact that tumors can look very different from one person to the next. It's why conventional approaches, such as chemotherapy or radiation, don't work for every patient. But weaponizing mRNA against cancer primes the immune cells with the tumor's specific genetic sequence, training the patient's body to attack their own unique type of cancer.
"It means you're able to think about personalizing cancer treatments down to specific subgroups of patients," says Mulqueen. "For example, eTheRNA are developing a renal cell carcinoma treatment which will be targeted at around 20% of these patients, who have specific tumor types. We're hoping to take that to human trials next year, but the challenge is trying to identify the right patients for the treatment at an early stage."
Repairing Damaged mRNA
While hopes are high that mRNA could usher in new cancer treatments and make CRISPR more accessible, a growing number of companies are also exploring an alternative to gene editing, known as RNA editing.
In genetic disorders, the mRNA in certain cells is impaired due to a rogue gene defect, and so the body ceases to produce a particular vital protein. Instead of permanently deleting the problem gene with CRISPR, the idea behind RNA editing is to inject small pieces of synthetic mRNA to repair the existing mRNA. Scientists think this approach will allow normal protein production to resume.
Over the past few years, this approach has gathered momentum, as some researchers have recognized that it holds certain key advantages over CRISPR. Companies from Belgium to Japan are now looking at RNA editing to treat all kinds of disorders, from Huntingdon's disease, to amyotrophic lateral sclerosis, or ALS, and certain types of cancer.
"With RNA editing, you don't need to make any changes to the DNA," explains Daniel de Boer, CEO of Dutch biotech ProQR, which is looking to treat rare genetic disorders that cause blindness. "Changes to the DNA are permanent, so if something goes wrong, that may not be desirable. With RNA editing, it's a temporary change, so we dose patients with our drugs once or twice a year."
Last month, ProQR reported a landmark case study, in which a patient with a rare form of blindness called Leber congenital amaurosis, which affects the retina at the back of the eye, recovered vision after three months of treatment.
"We have seen that this RNA therapy restores vision in people that were completely blind for a year or so," says de Boer. "They were able to see again, to read again. We think there are a large number of other genetic diseases we could go after with this technology. There are thousands of different mutations that can lead to blindness, and we think this technology can target approximately 25% of them."
Ultimately, there's likely to be a role for both RNA editing and CRISPR, depending on the disease. "I think CRISPR is ideally suited for illnesses where you would like to permanently correct a genetic defect," says Joshua Rosenthal of the Marine Biology Laboratory in Chicago. "Whereas RNA editing could be used to treat things like pain, where you might want to reset a neural circuit temporarily over a shorter period of time."
Much of this research has been accelerated by the COVID-19 pandemic, which has played a major role in bringing mRNA to the forefront of people's minds as a therapeutic.
"The pandemic has really shown that not only are mRNA approaches viable, they could in certain circumstances be vastly superior to more traditional technologies," says Mulqueen. "In the future, I would not be surprised if many of the top pharma products are mRNA derived."
As We Wait for a Vaccine, Scientists Work to Scale Up the Best COVID-19 Antibodies to Give New Patients
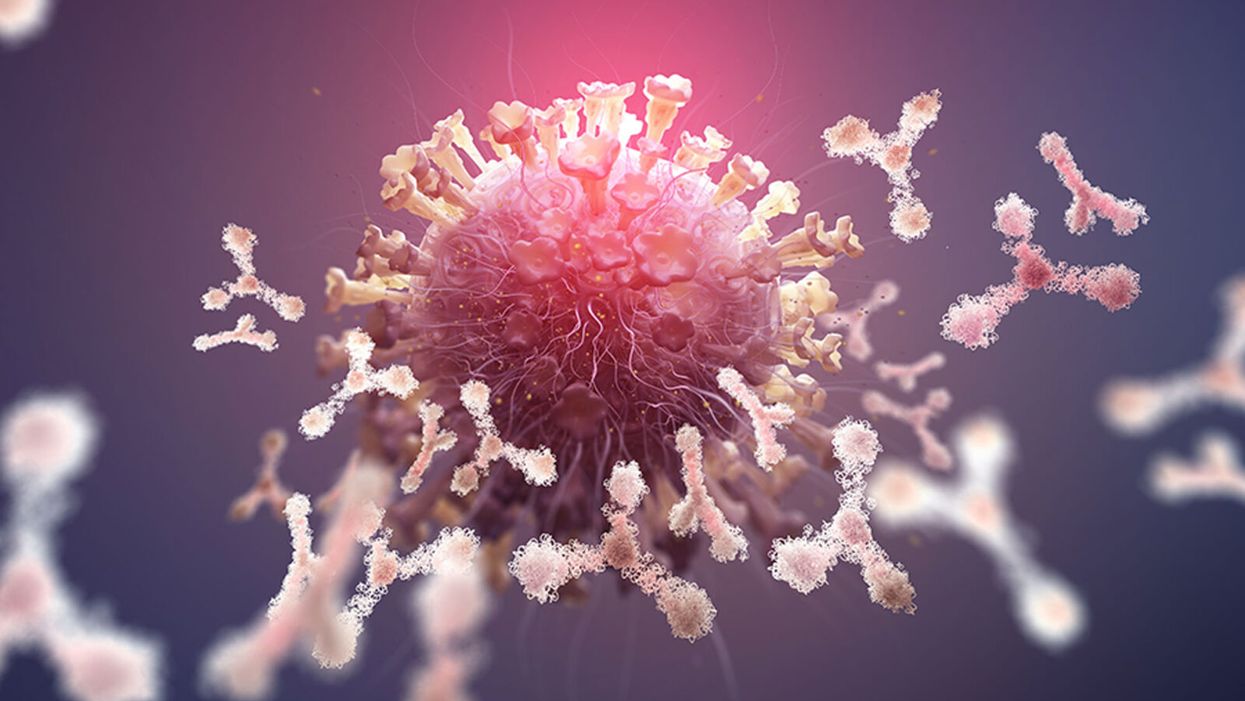
In this illustration of immune defense, Y-shaped proteins called antibodies attack a virus.
When we get sick, our immune system sends its soldier cells to the battlefield. Called B-cells, they "examine" the foreign particles that shouldn't be in our bloodstream—and start producing the antibodies, the proteins to neutralize the invaders.
To screen the antibodies, scientists have developed a proprietary way to make the effective ones glow – like a literal "lightbulb" moment.
The better these antibodies are at neutralizing the pathogen, the faster we recover.
The antibodies acquired from COVID-19 survivors already showed promise in treating other patients, but because they must be obtained from people, generating a regular supply is not feasible. To close the gap, researchers are trying to identify the B-cells that make the best antibodies—and then farm them in laboratories at scale.
Scientists at Berkley Lights, a biotechnology company in California, have been screening B-cells from recovered patients and testing the antibodies they release for virus-neutralizing abilities. To screen the antibodies, scientists there have developed a proprietary way to make the effective ones glow – like a literal "lightbulb" moment.
So how does it work? First, the individual B-cells are placed into microscopic chambers called nano-pens, where they secrete the antibodies. Once released, the antibodies are flushed over tiny beads that have bits of the viral particles attached to them, along with special molecules that can emit fluorescent light.
"When an antibody binds to the bead, we see a bright light on the bead," explains John Proctor, the company's senior vice president of antibody therapeutics. "So we can identify which cells are making the antibodies."
Then the antibodies are tested for their ability to counteract the coronavirus's spike proteins, which the virus uses to break into our cells. Not all antibodies are equally good at this crucial defense move—some can block only parts of the virus's machinery, while others can neutralize it completely. Proctor and his colleagues are looking for the latter.
Once scientists identify the best performing B-cells, they crack the cells open—or in scientific terms "lyse" them—and extract the genetic instructions for making the antibodies. As it turns out, B-cells aren't very efficient at pumping out massive amounts of antibodies, so scientists insert these genetic instructions into a different, more prolific type of cell.
Named Chinese Hamster Ovary Cells or CHO, these cells are commonly used in the pharmaceutical industry because they can generate therapeutic proteins en masse. Under the right nutrient conditions, which include a lot of sugar, CHO cells can keep making the antibodies at commercial levels. "They are engineered to operate in very large bioreactors," Proctor explains.
While traditional antibody screening can take three months, the Beacon System can do it in less than 24 hours.
Berkeley Lights' technology has already been used to screen the antibodies of recovered patients from Vanderbilt University Medical Center. In another example, a biotech company GenScript ProBio used the platform to screen mice engineered to have human antibodies for the coronavirus.
In addition to its small, lab-on-a-chip size, Berkeley Lights' system allows scientists to greatly speed up the screening process. While traditional antibody screening can take three months, the Beacon System can do it in less than 24 hours. "We only need one B-cell per pen and a couple of beads to see that fluorescent signal," Proctor says. "It is a more advanced way to process and analyze cells, and that level of sensitivity is unique to our technology."

B-cells secreting antibodies inside the Berkeley Lights Beacon System Nano-Pens.
While vaccines are likely to take months to develop and test, antibodies might arrive to the battleground sooner. With the extremely limited treatment options for COVID-19, antibody-based therapeutics can potentially bridge this gap.
Lina Zeldovich has written about science, medicine and technology for Popular Science, Smithsonian, National Geographic, Scientific American, Reader’s Digest, the New York Times and other major national and international publications. A Columbia J-School alumna, she has won several awards for her stories, including the ASJA Crisis Coverage Award for Covid reporting, and has been a contributing editor at Nautilus Magazine. In 2021, Zeldovich released her first book, The Other Dark Matter, published by the University of Chicago Press, about the science and business of turning waste into wealth and health. You can find her on http://linazeldovich.com/ and @linazeldovich.
A map of cumulative known cases of COVID-19 in the U.S., as of June 12th, 2020.
Have you felt a bit like an armchair epidemiologist lately? Maybe you've been poring over coronavirus statistics on your county health department's website or on the pages of your local newspaper.
If the percentage of positive tests steadily stays under 8 percent, that's generally a good sign.
You're likely to find numbers and charts but little guidance about how to interpret them, let alone use them to make day-to-day decisions about pandemic safety precautions.
Enter the gurus. We asked several experts to provide guidance for laypeople about how to navigate the numbers. Here's a look at several common COVID-19 statistics along with tips about how to understand them.
Case Counts: Consider the Context
The number of confirmed COVID-19 cases in American counties is widely available. Local and state health departments should provide them online, or you can easily look them up at The New York Times' coronavirus database. However, you need to be cautious about interpreting them.
"Case counts are the obvious numbers to look at. But they're probably the hardest thing to sort out," said Dr. Jeff Martin, an epidemiologist at the University of California at San Francisco.
That's because case counts by themselves aren't a good window into how the coronavirus is affecting your community since they rely on testing. And testing itself varies widely from day to day and community to community.
"The more testing that's done, the more infections you'll pick up," explained Dr. F. Perry Wilson, a physician at Yale University. The numbers can also be thrown off when tests are limited to certain groups of people.
"If the tests are being mostly given to people with a high probability of having been infected -- for example, they have had symptoms or work in a high-risk setting -- then we expect lots of the tests to be positive. But that doesn't tell us what proportion of the general public is likely to have been infected," said Eleanor Murray, an epidemiologist at Boston University.
These Stats Are More Meaningful
According to Dr. Wilson, it's more useful to keep two other statistics in mind: the number of COVID tests that are being performed in your community and the percentage that turn up positive, showing that people have the disease. (These numbers may or may not be available locally. Check the websites of your community's health department and local news media outlets.)
If the number of people being tested is going up, but the percentage of positive tests is going down, Dr. Wilson said, that's a good sign. But if both numbers are going up – the number of people tested and the percentage of positive results – then "that's a sign that there are more infections burning in the community."
It's especially worrisome if the percentage of positive cases is growing compared to previous days or weeks, he said. According to him, that's a warning of a "high-risk situation."
Dr. George Rutherford, an epidemiologist at University of California at San Francisco, offered this tip: If the percentage of positive tests steadily stays under 8 percent, that's generally a good sign.
There's one more caveat about case counts. It takes an average of a week for someone to be infected with COVID-19, develop symptoms, and get tested, Dr. Rutherford said. It can take an additional several days for those test results to be reported to the county health department. This means that case numbers don't represent infections happening right now, but instead are a picture of the state of the pandemic more than a week ago.
Hospitalizations: Focus on Current Statistics
You should be able to find numbers about how many people in your community are currently hospitalized – or have been hospitalized – with diagnoses of COVID-19. But experts say these numbers aren't especially revealing unless you're able to see the number of new hospitalizations over time and track whether they're rising or falling. This number often isn't publicly available, however.
If new hospitalizations are increasing, "you may want to react by being more careful yourself."
And there's an important caveat: "The problem with hospitalizations is that they do lag," UC San Francisco's Dr. Martin said, since it takes time for someone to become ill enough to need to be hospitalized. "They tell you how much virus was being transmitted in your community 2 or 2.5 weeks ago."
Also, he said, people should be cautious about comparing new hospitalization rates between communities unless they're adjusted to account for the number of more-vulnerable older people.
Still, if new hospitalizations are increasing, he said, "you may want to react by being more careful yourself."
Deaths: They're an Even More Delayed Headline
Cable news networks obsessively track the number of coronavirus deaths nationwide, and death counts for every county in the country are available online. Local health departments and media websites may provide charts tracking the growth in deaths over time in your community.
But while death rates offer insight into the disease's horrific toll, they're not useful as an instant snapshot of the pandemic in your community because severely ill patients are typically sick for weeks. Instead, think of them as a delayed headline.
"These numbers don't tell you what's happening today. They tell you how much virus was being transmitted 3-4 weeks ago," Dr. Martin said.
'Reproduction Value': It May Be Revealing
You're not likely to find an available "reproduction value" for your community, but it is available for your state and may be useful.
A reproduction value, also known as R0 or R-naught, "tells us how many people on average we expect will be infected from a single case if we don't take any measures to intervene and if no one has been infected before," said Boston University's Murray.
As The New York Times explained, "R0 is messier than it might look. It is built on hard science, forensic investigation, complex mathematical models — and often a good deal of guesswork. It can vary radically from place to place and day to day, pushed up or down by local conditions and human behavior."
It may be impossible to find the R0 for your community. However, a website created by data specialists is providing updated estimates of a related number -- effective reproduction number, or Rt – for each state. (The R0 refers to how infectious the disease is in general and if precautions aren't taken. The Rt measures its infectiousness at a specific time – the "t" in Rt.) The site is at rt.live.
"The main thing to look at is whether the number is bigger than 1, meaning the outbreak is currently growing in your area, or smaller than 1, meaning the outbreak is currently decreasing in your area," Murray said. "It's also important to remember that this number depends on the prevention measures your community is taking. If the Rt is estimated to be 0.9 in your area and you are currently under lockdown, then to keep it below 1 you may need to remain under lockdown. Relaxing the lockdown could mean that Rt increases above 1 again."
"Whether they're on the upswing or downswing, no state is safe enough to ignore the precautions about mask wearing and social distancing."
Keep in mind that you can still become infected even if an outbreak in your community appears to be slowing. Low risk doesn't mean no risk.
Putting It All Together: Why the Numbers Matter
So you've reviewed COVID-19 statistics in your community. Now what?
Dr. Wilson suggests using the data to remind yourself that the coronavirus pandemic "is still out there. You need to take it seriously and continue precautions," he said. "Whether they're on the upswing or downswing, no state is safe enough to ignore the precautions about mask wearing and social distancing. 'My state is doing well, no one I know is sick, is it time to have a dinner party?' No."
He also recommends that laypeople avoid tracking COVID-19 statistics every day. "Check in once a week or twice a month to see how things are going," he suggested. "Don't stress too much. Just let it remind you to put that mask on before you get out of your car [and are around others]."