Technology is Redefining the Age of 'Older Mothers'

Scientists are working on technologies that would enable more 70-year-old women to have babies.
In October 2021, a woman from Gujarat, India, stunned the world when it was revealed she had her first child through in vitro fertilization (IVF) at age 70. She had actually been preceded by a compatriot of hers who, two years before, gave birth to twins at the age of 73, again with the help of IVF treatment. The oldest known mother to conceive naturally lived in the UK; in 1997, Dawn Brooke conceived a son at age 59.
These women may seem extreme outliers, almost freaks of nature; in the US, for example, the average age of first-time mothers is 26. A few decades from now, though, the sight of 70-year-old first-time mothers may not even raise eyebrows, say futurists.
“We could absolutely have more 70-year-old mothers because we are learning how to regulate the aging process better,” says Andrew Hessel, a microbiologist and geneticist, who cowrote "The Genesis Machine," a book about “rewriting life in the age of synthetic biology,” with Amy Webb, the futurist who recently wondered why 70-year-old women shouldn’t give birth.
Technically, we're already doing this, says Hessel, pointing to a technique known as in vitro gametogenesis (IVG). IVG refers to turning adult cells into sperm or egg cells. “You can think of it as the upgrade to IVF,” Hessel says. These vanguard stem cell research technologies can take even skin cells and turn them into induced pluripotent stem cells (iPSCs), which are basically master cells capable of maturing into any human cell, be it kidney cells, liver cells, brain cells or gametes, aka eggs and sperm, says Henry T. “Hank” Greely, a Stanford law professor who specializes in ethical, legal, and social issues in biosciences.
Mothers over 70 will be a minor blip, statistically speaking, Greely predicts.
In 2016, Greely wrote "The End of Sex," a book in which he described the science of making gametes out of iPSCs in detail. Greely says science will indeed enable us to see 70-year-old new mums fraternize with mothers several decades younger at kindergartens in the (not far) future. And it won’t be that big of a deal.
“An awful lot of children all around the world have been raised by grandmothers for millennia. To have 70-year-olds and 30-year-olds mingling in maternal roles is not new,” he says. That said, he doubts that many women will want to have a baby in the eighth decade of their life, even if science allows it. “Having a baby and raising a child is hard work. Even if 1% of all mothers are over 65, they aren’t going to change the world,” Greely says. Mothers over 70 will be a minor blip, statistically speaking, he predicts. But one thing is certain: the technology is here.
And more technologies for the same purpose could be on the way. In March 2021, researchers from Monash University in Melbourne, Australia, published research in Nature, where they successfully reprogrammed skin cells into a three-dimensional cellular structure that was morphologically and molecularly similar to a human embryo–the iBlastoid. In compliance with Australian law and international guidelines referencing the “primitive streak rule," which bans the use of embryos older than 14 days in scientific research, Monash scientists stopped growing their iBlastoids in vitro on day 11.
“The research was both cutting-edge and controversial, because it essentially created a new human life, not for the purpose of a patient who's wanting to conceive, but for basic research,” says Lindsay Wu, a senior lecturer in the School of Medical Sciences at the University of New South Wales (UNSW), in Kensington, Australia. If you really want to make sure what you are breeding is an embryo, you need to let it develop into a viable baby. “This is the real proof in the pudding,'' says Wu, who runs UNSW’s Laboratory for Ageing Research. Then you get to a stage where you decide for ethical purposes you have to abort it. “Fiddling here a bit too much?” he asks. Wu believes there are other approaches to tackling declining fertility due to older age that are less morally troubling.
He is actually working on them. Why would it be that women, who are at peak physical health in almost every other regard, in their mid- to late- thirties, have problems conceiving, asked Wu and his team in a research paper published in 2020 in Cell Reports. The simple answer is the egg cell. An average girl in puberty has between 300,000 and 400,000 eggs, while at around age 37, the same woman has only 25,000 eggs left. Things only go downhill from there. So, what torments the egg cells?
The UNSW team found that the levels of key molecules called NAD+ precursors, which are essential to the metabolism and genome stability of egg cells, decline with age. The team proceeded to add these vitamin-like substances back into the drinking water of reproductively aged, infertile lab mice, which then had babies.
“It's an important proof of concept,” says Wu. He is investigating how safe it is to replicate the experiment with humans in two ongoing studies. The ultimate goal is to restore the quality of egg cells that are left in patients in their late 30s and early- to mid-40s, says Wu. He sees the goal of getting pregnant for this age group as less ethically troubling, compared to 70-year-olds.
But what is ethical, anyway? “It is a tricky word,” says Hessel. He differentiates between ethics, which represent a personal position and may, thus, be more transient, and morality, longer lasting principles embraced across society such as, “Thou shalt not kill.” Unprecedented advances often bring out fear and antagonism until time passes and they just become…ordinary. When IVF pioneer Landrum Shettles tried to perform IVF in 1973, the chairman of Columbia’s College of Physicians and Surgeons interdicted the procedure at the last moment. Almost all countries in the world have IVF clinics today, and the global IVF services market is clearly a growth industry.
Besides, you don’t have a baby at 70 by accident: you really want it, Greely and Hessel agree. And by that age, mothers may be wiser and more financially secure, Hessel says (though he is quick to add that even the pregnancy of his own wife, who had her child at 40, was a high-risk one).
As a research question, figuring out whether older mothers are better than younger ones and vice-versa entails too many confounding variables, says Greely. And why should we focus on who’s the better mother anyway? “We've had 70-year-old and 80-year-old fathers forever–why should people have that much trouble getting used to mothers doing the same?” Greely wonders. For some women having a child at an old(er) age would be comforting; maybe that’s what matters.
And the technology to enable older women to have children is already here or coming very soon. That, perhaps, matters even more. Researchers have already created mice–and their offspring–entirely from scratch in the lab. “Doing this to produce human eggs is similar," says Hessel. "It is harder to collect tissues, and the inducing cocktails are different, but steady advances are being made." He predicts that the demand for fertility treatments will keep financing research and development in the area. He says that big leaps will be made if ethical concerns don’t block them: it is not far-fetched to believe that the first baby produced from lab-grown eggs will be born within the next decade.
In an op-ed in 2020 with Stat, Greely argued that we’ve already overcome the technical barrier for human cloning, but no one's really talking about it. Likewise, scientists are also working on enabling 70-year-old women to have babies, says Hessel, but most commentators are keeping really quiet about it. At least so far.
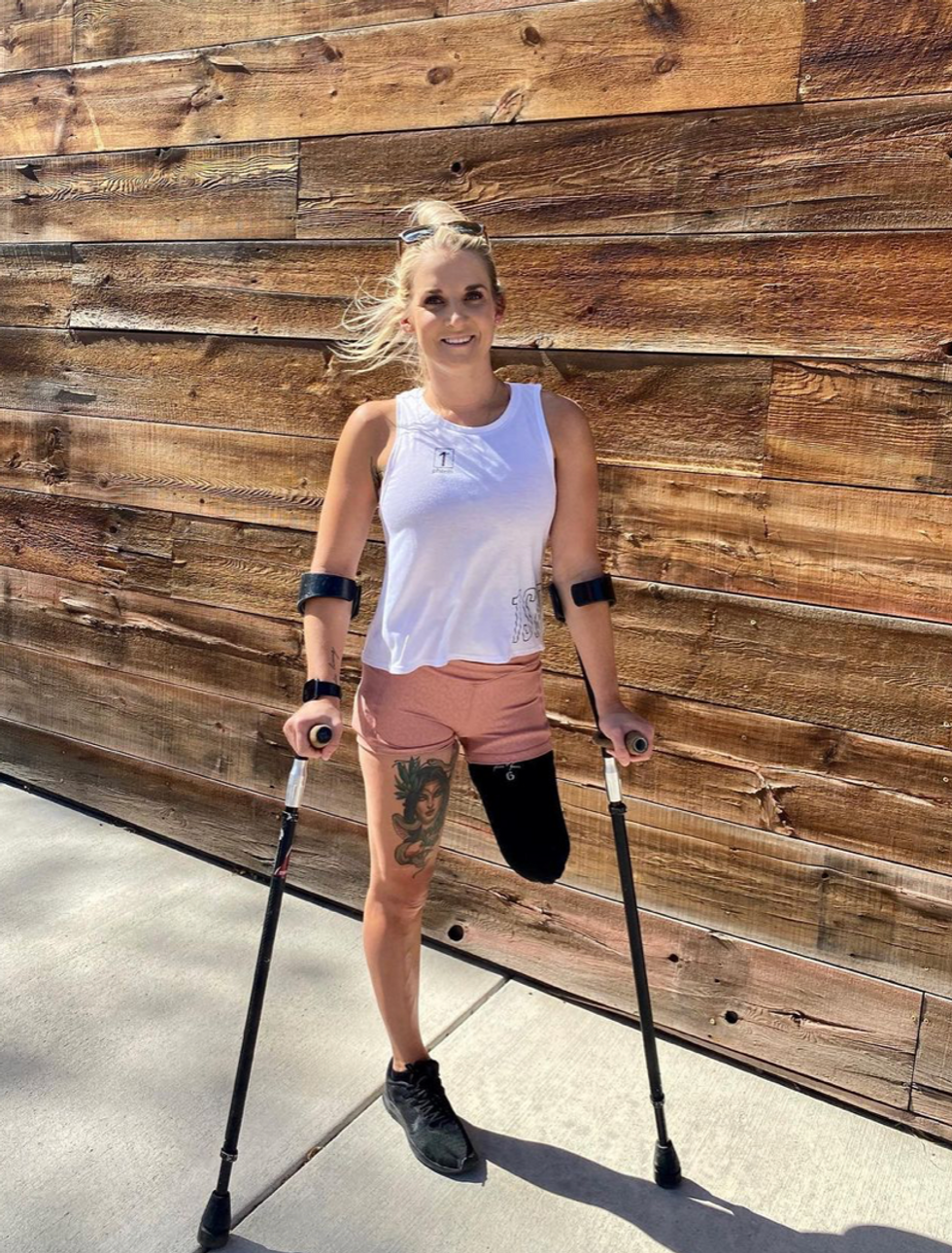
Kirstie Ennis, an Afghanistan veteran who survived a helicopter crash but lost a limb, pictured in May 2021 at Two Rivers Park in Colorado.
In June 2012, Kirstie Ennis was six months into her second deployment to Afghanistan and recently promoted to sergeant. The helicopter gunner and seven others were three hours into a routine mission of combat resupplies and troop transport when their CH-53D helicopter went down hard.
Miraculously, all eight people onboard survived, but Ennis' injuries were many and severe. She had a torn rotator cuff, torn labrum, crushed cervical discs, facial fractures, deep lacerations and traumatic brain injury. Despite a severely fractured ankle, doctors managed to save her foot, for a while at least.
In November 2015, after three years of constant pain and too many surgeries to count, Ennis relented. She elected to undergo a lower leg amputation but only after she completed the 1,000-mile, 72-day Walking with the Wounded journey across the UK.
On Veteran's Day of that year, on the other side of the country, orthopedic surgeon Cato Laurencin announced a moonshot challenge he was setting out to achieve on behalf of wounded warriors like Ennis: the Hartford Engineering A Limb (HEAL) Project.
Laurencin, who is a University of Connecticut professor of chemical, materials and biomedical engineering, teamed up with experts in tissue bioengineering and regenerative medicine from Harvard, Columbia, UC Irvine and SASTRA University in India. Laurencin and his colleagues at the Connecticut Convergence Institute for Translation in Regenerative Engineering made a bold commitment to regenerate an entire limb within 15 years – by the year 2030.

Dr. Cato Laurencin pictured in his office at UConn.
Photo Credit: UConn
Regenerative Engineering -- A Whole New Field
Limb regeneration in humans has been a medical and scientific fascination for decades, with little to show for the effort. However, Laurencin believes that if we are to reach the next level of 21st century medical advances, this puzzle must be solved.
An estimated 185,000 people undergo upper or lower limb amputation every year. Despite the significant advances in electromechanical prosthetics, these individuals still lack the ability to perform complex functions such as sensation for tactile input, normal gait and movement feedback. As far as Laurencin is concerned, the only clinical answer that makes sense is to regenerate a whole functional limb.
Laurencin feels other regeneration efforts were hampered by their siloed research methods with chemists, surgeons, engineers all working separately. Success, he argues, requires a paradigm shift to a trans-disciplinary approach that brings together cutting-edge technologies from disparate fields such as biology, material sciences, physical, chemical and engineering sciences.
As the only surgeon ever inducted into the academies of Science, Medicine and Innovation, Laurencin is uniquely suited for the challenge. He is regarded as the founder of Regenerative Engineering, defined as the convergence of advanced materials sciences, stem cell sciences, physics, developmental biology and clinical translation for the regeneration of complex tissues and organ systems.
But none of this is achievable without early clinician participation across scientific fields to develop new technologies and a deeper understanding of how to harness the body's innate regenerative capabilities. "When I perform a surgical procedure or something is torn or needs to be repaired, I count on the body being involved in regenerating tissue," he says. "So, understanding how the body works to regenerate itself and harnessing that ability is an important factor for the regeneration process."
The Birth of the Vision
Laurencin's passion for regeneration began when he was a sports medicine fellow at Cornell University Medical Center in the early 1990s. There he saw a significant number of injuries to the anterior cruciate ligament (ACL), the major ligament that stabilizes the knee. He believed he could develop a better way to address those injuries using biomaterials to regenerate the ligament. He sketched out a preliminary drawing on a napkin one night over dinner. He has spent the next 30 years regenerating tissues, including the patented L-C ligament.
As chair of Orthopaedic Surgery at the University of Virginia during the peak of the wars in Iraq and Afghanistan, Laurencin treated military personnel who survived because of improved helmets, body armor and battlefield medicine but were left with more devastating injuries, including traumatic brain injuries and limb loss.
"I was so honored to care for them and I so admired their steadfast courage that I became determined to do something big for them," says Laurencin.
When he tells people about his plans to regrow a limb, he gets a lot of eye rolls, which he finds amusing but not discouraging. Growing bone cells was relatively new when he was first focused on regenerating bone in 1987 at MIT; in 2007 he was well on his way to regenerating ligaments at UVA when many still doubted that ligaments could even be reconstructed. He and his team have already regenerated torn rotator cuff tendons and ACL ligaments using a nano-textured fabric seeded with stem cells.
Even as a finalist for the $4 million NIH Pioneer Award for high-risk/high-reward research, he faced a skeptical scientific audience in 2014. "They said, 'Well what do you plan to do?' I said 'I plan to regenerate a whole limb in people.' There was a lot of incredulousness. They stared at me and asked a lot of questions. About three days later, I received probably the best score I've ever gotten on an NIH grant."
In the Thick of the Science
Humans are born with regenerative abilities--two-year-olds have regrown fingertips--but lose that ability with age. Salamanders are the only vertebrates that can regenerate lost body parts as adults; axolotl, the rare Mexican salamander, can grow extra limbs.
The axolotl is important as a model organism because it is a four-footed vertebrate with a similar body plan to humans. Mapping the axolotl genome in 2018 enhanced scientists' genetic understanding of their evolution, development, and regeneration. Being easy to breed in captivity allowed the HEAL team to closely study these amphibians and discover a new cell type they believe may shed light on how to mimic the process in humans.
"Whenever limb regeneration takes place in the salamander, there is a huge amount of something called heparan sulfate around that area," explains Laurencin. "We thought, 'What if this heparan sulfate is the key ingredient to allowing regeneration to take place?' We found these groups of cells that were interspersed in tissues during the time of regeneration that seemed to have connections to each other that expressed this heparan sulfate."
Called GRID (Groups that are Regenerative, Interspersed and Dendritic), these cells were also recently discovered in mice. While GRID cells don't regenerate as well in mice as in salamanders, finding them in mammals was significant.
"If they're found in mice. we might be able to find these in humans in some form," Laurencin says. "We think maybe it will help us figure out regeneration or we can create cells that mimic what grid cells do and create an artificial grid cell."
What Comes Next?
Laurencin and his team have individually engineered and made every single tissue in the lower limb, including bone, cartilage, ligament, skin, nerve, blood vessels. Regenerating joints and joint tissue is the next big mile marker, which Laurencin sees as essential to regenerating a limb that functions and performs in the way he envisions.
"Using stem cells and amnion tissue, we can regenerate joints that are damaged, and have severe arthritis," he says. "We're making progress on all fronts, and making discoveries we believe are going to be helping people along the way."
That focus and advancement is vital to Ennis. After laboring over the decision to have her leg amputated below the knee, she contracted MRSA two weeks post-surgery. In less than a month, she went from a below-the-knee-amputee to a through-the-knee amputee to an above-the-knee amputee.
"A below-the-knee amputation is night-and-day from above-the-knee," she said. "You have to relearn everything. You're basically a toddler."
Kirstie Ennis pictured in July 2020.
Photo Credit: Ennis' Instagram
The clock is ticking on the timeline Laurencin set for himself. Nine years might seem like forever if you're doing time but it might appear fleeting when you're trying to create something that's never been done before. But Laurencin isn't worried. He's convinced time is on his side.
"Every week, I receive an email or a call from someone, maybe a mother whose child has lost a finger or I'm in communication with a disabled American veteran who wants to know how the progress is going. That energizes me to continue to work hard to try to create these sorts of solutions because we're talking about people and their lives."
He devotes about 60 hours a week to the project and the roughly 100 students, faculty and staff who make up the HEAL team at the Convergence Institute seem acutely aware of what's at stake and appear equally dedicated.
"We're in the thick of the science in terms of making this happen," says Laurencin. "We've moved from making the impossible possible to making the possible a reality. That's what science is all about."
7 Reasons Why We Should Not Need Boosters for COVID-19
A top infectious disease physician explains why immunity derived from natural infection and the vaccines should be long-lasting.
There are at least 7 reasons why immunity after vaccination or infection with COVID-19 should likely be long-lived. If durable, I do not think boosters will be necessary in the future, despite CEOs of pharmaceutical companies (who stand to profit from boosters) messaging that they may and readying such boosters. To explain these reasons, let's orient ourselves to the main components of the immune system.
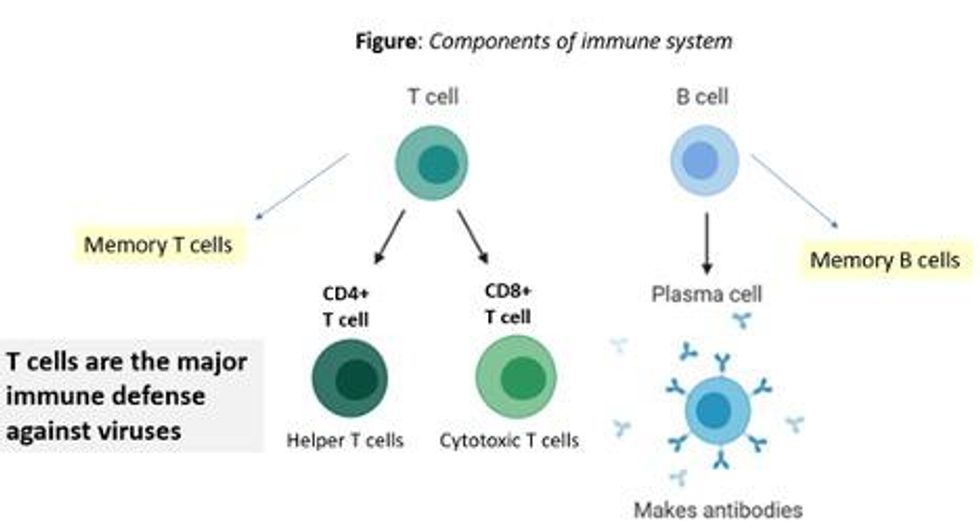
There are two major arms of the immune system: B cells (which produce antibodies) and T cells (which are formed specifically to attack and kill pathogens). T cells are divided into two types, CD4 cells ("helper" T cells) and CD8 cells ("cytotoxic" T cells).
Each arm, once stimulated by infection or vaccine, should hopefully make "memory" banks. So if the body sees the pathogen in the future, these defenses should come roaring back to attack the virus and protect you from getting sick. Plenty of research in COVID-19 indicates a likely long-lasting response to the vaccine or infection. Here are seven of the most compelling reasons:
REASON 1: Memory B Cells Are Produced By Vaccines and Natural Infection
In one study, 12 volunteers who had never had Covid-19--and were fully vaccinated with two Pfizer/BioNTech shots-- underwent biopsies of their lymph nodes. This is where memory B cells are stored in places called "germinal centers". The biopsies were performed three, four, six, and seven weeks after the first mRNA vaccine shot, and were stained to reveal that germinal center memory B cells in the lymph nodes increased in concentration over time.
Natural infection also generates memory B cells. Even after antibody levels wane over time, strong memory B cells were detected in the blood of individuals six and eight months after infection in different studies. Indeed, the half-lives of the memory B cells seen in the study examining patients 8 months after COVID-19 led the authors to conclude that "B cell memory to SARS-CoV-2 was robust and is likely long-lasting." Reason #2 tells us that memory B cells can be active for a very long time indeed.
REASON #2: Memory B Cells Can Produce Neutralizing Antibodies If They See Infection Again Decades Later
Demonstrated production of memory B cells after vaccination or natural infection with COVID-19 is so important because memory B cells, once generated, can be activated to produce high levels of neutralizing antibodies against the pathogen even if encountered many years after the initial exposure. In one amazing study (published in 2008), researchers isolated memory B cells against the 1918 flu strain from the blood of 32 individuals aged 91-101 years. These people had been born on or before 1915 and had survived that pandemic.
Their memory B cells, when exposed to the 1918 flu strain in a test tube, generated high levels of neutralizing antibodies against the virus -- antibodies that then protected mice from lethal infection with this deadly strain. The ability of memory B cells to produce complex antibody responses against an infection nine decades after exposure speaks to their durability.
REASON #3: Vaccines or Natural Infection Trigger Strong Memory T Cell Immunity
All of the trials of the major COVID-19 vaccine candidates measured strong T cell immunity following vaccination, most often assessed by measuring SARS-CoV-2 specific T cells in the phase I/II safety and immunogenicity studies. There are a number of studies that demonstrate the production of strong T cell immunity to COVID-19 after natural infection as well, even when the infection was mild or asymptomatic.
The same study that showed us robust memory B cell production 8 months after natural infection also demonstrated strong and sustained memory T cell production. In fact, the half-lives of the memory T cells in this cohort were long (~125-225 days for CD8+ and ~94-153 days for CD4+ T cells), comparable to the 123-day half-life observed for memory CD8+ T cells after yellow fever immunization (a vaccine usually given once over a lifetime).
A recent study of individuals recovered from COVID-19 show that the initial T cells generated by natural infection mature and differentiate over time into memory T cells that will be "put in the bank" for sustained periods.
REASON #4: T Cell Immunity Following Vaccinations for Other Infections Is Long-Lasting
Last year, we were fortunate to be able to measure how T cell immunity is generated by COVID-19 vaccines, which was not possible in earlier eras when vaccine trials were done for other infections (such as measles, mumps, rubella, pertussis, diphtheria). Antibodies are just the "tip of the iceberg" when assessing the response to vaccination, but were the only arm of the immune response that could be measured following vaccination in the past.
Measuring pathogen-specific T cell responses takes sophisticated technology. However, T cell responses, when assessed years after vaccination for other pathogens, has been shown to be long-lasting. For example, in one study of 56 volunteers who had undergone measles vaccination when they were much younger, strong CD8 and CD4 cell responses to vaccination could be detected up to 34 years later.
REASON #5: T Cell Immunity to Related Coronaviruses That Caused Severe Disease is Long-Lasting
SARS-CoV-2 is a coronavirus that causes severe disease, unlike coronaviruses that cause the common cold. Two other coronaviruses in the recent past caused severe disease, specifically Severely Acute Respiratory Distress Syndrome (SARS) in late 2002-2003 and Middle East Respiratory Syndrome (MERS) in 2011.
A study performed in 2020 demonstrated that the blood of 23 recovered SARS patients possess long-lasting memory T cells that were still reactive to SARS 17 years after the outbreak in 2003. Many scientists expect that T cell immunity to SARS-CoV-2 will be equally durable to that of its cousin.
REASON #6: T Cell Responses from Vaccination and Natural Infection With the Ancestral Strain of COVID-19 Are Robust Against Variants
Even though antibody responses from vaccination may be slightly lower against various COVID-19 variants of concern that have emerged in recent months, T cell immunity after vaccination has been shown to be unperturbed by mutations in the spike protein (in the variants). For instance, T cell responses after mRNA vaccines maintained strong activity against different variants (including P.1 Brazil variant, B.1.1.7 UK variant, B.1.351 South Africa variant and the CA.20.C California variant) in a recent study.
Another study showed that the vaccines generated robust T cell immunity that was unfazed by different variants, including B.1.351 and B.1.1.7. The CD4 and CD8 responses generated after natural infection are equally robust, showing activity against multiple "epitopes" (little segments) of the spike protein of the virus. For instance, CD8 cells responds to 52 epitopes and CD4 cells respond to 57 epitopes across the spike protein, so that a few mutations in the variants cannot knock out such a robust and in-breadth T cell response. Indeed, a recent paper showed that mRNA vaccines were 97.4 percent effective against severe COVID-19 disease in Qatar, even when the majority of circulating virus there was from variants of concern (B.1.351 and B.1.1.7).
REASON #7: Coronaviruses Don't Mutate Quickly Like Influenza, Which Requires Annual Booster Shots
Coronaviruses are RNA viruses, like influenza and HIV (which is actually a retrovirus), but do not mutate as quickly as either one. The reason that coronaviruses don't mutate very rapidly is that their replicating mechanism (polymerase) has a strong proofreading mechanism: If the virus mutates, it usually goes back and self-corrects. Mutations can arise with high rates of replication when transmission is very frequent -- as has been seen in recent months with the emergence of SARS-CoV-2 variants during surges. However, the COVID-19 virus will not be mutating like this when we tamp down transmission with mass vaccination.
In conclusion, I and many of my infectious disease colleagues expect the immunity from natural infection or vaccination to COVID-19 to be durable. Let's put discussion of boosters aside and work hard on global vaccine equity and distribution since the pandemic is not over until it is over for us all.

