Hyperbaric oxygen therapy could treat Long COVID, new study shows

Hyperbaric oxygen therapy has been used in the past to help people with traumatic brain injury, stroke and other conditions involving wounds to the brain. Now, researchers at Shamir Medical Center in Tel Aviv are studying how it could treat Long Covid.
Long COVID is not a single disease, it is a syndrome or cluster of symptoms that can arise from exposure to SARS-CoV-2, a virus that affects an unusually large number of different tissue types. That's because the ACE2 receptor it uses to enter cells is common throughout the body, and inflammation from the immune response fighting that infection can damage surrounding tissue.
One of the most widely shared groups of symptoms is fatigue and what has come to be called “brain fog,” a difficulty focusing and an amorphous feeling of slowed mental functioning and capacity. Researchers have tied these COVID-related symptoms to tissue damage in specific sections of the brain and actual shrinkage in its size.
When Shai Efrati, medical director of the Sagol Center for Hyperbaric Medicine and Research in Tel Aviv, first looked at functional magnetic resonance images (fMRIs) of patients with what is now called long COVID, he saw “micro infarcts along the brain.” It reminded him of similar lesions in other conditions he had treated with hyperbaric oxygen therapy (HBOT). “Once we saw that, we said, this is the type of wound we can treat. It doesn't matter if the primary cause is mechanical injury like TBI [traumatic brain injury] or stroke … we know how to oxidize them.”Efrati came to HBOT almost by accident. The physician had seen how it had helped heal diabetic ulcers and improved the lives of other patients, but he was busy with his own research. Then the director of his Tel Aviv hospital threatened to shut down the small HBOT chamber unless Efrati took on administrative responsibility for it. He reluctantly agreed, a decision that shifted the entire focus of his research.
“The main difference between wounds in the leg and wounds in the brain is that one is something we can see, it's tangible, and the wound in the brain is hidden,” says Efrati. With fMRIs, he can measure how a limited supply of oxygen in blood is shuttled around to fuel activity in various parts of the brain. Years of research have mapped how specific areas of the brain control activity ranging from thinking to moving. An fMRI captures the brain area as it’s activated by supplies of oxygen; lack of activity after the same stimuli suggests damage has occurred in that tissue. Suddenly, what was hidden became visible to researchers using fMRI. It helped to make a diagnosis and measure response to treatment.
HBOT is not a single thing but rather a tool, a process or approach with variations depending on the condition being treated. It aims to increase the amount of oxygen that gets to damaged tissue and speed up healing. Regular air is about 21 percent oxygen. But inside the HBOT chamber the atmospheric pressure can be increased to up to three times normal pressure at sea level and the patient breathes pure oxygen through a mask; blood becomes saturated with much higher levels of oxygen. This can defuse through the damaged capillaries of a wound and promote healing.
The trial
Efrati’s clinical trials started in December 2020, barely a year after SARS-CoV-2 had first appeared in Israel. Patients who’d experienced cognitive issues after having COVID received 40 sessions in the chamber over a period of 60 days. In each session, they spent 90 minutes breathing through a mask at two atmospheres of pressure. While inside, they performed mental exercises to train the brain. The only difference between the two groups of patients was that one breathed pure oxygen while the other group breathed normal air. No one knew who was receiving which level of oxygen.
The results were striking. Before and after fMRIs showed significant repair of damaged tissue in the brain and functional cognition tests improved substantially among those who received pure oxygen. Importantly, 80 percent of patients said they felt back to “normal,” but Efrati says they didn't include patient evaluation in the paper because there was no baseline data to show how they functioned before COVID. After the study was completed, the placebo group was offered a new round of treatments using 100 percent oxygen, and the team saw similar results.
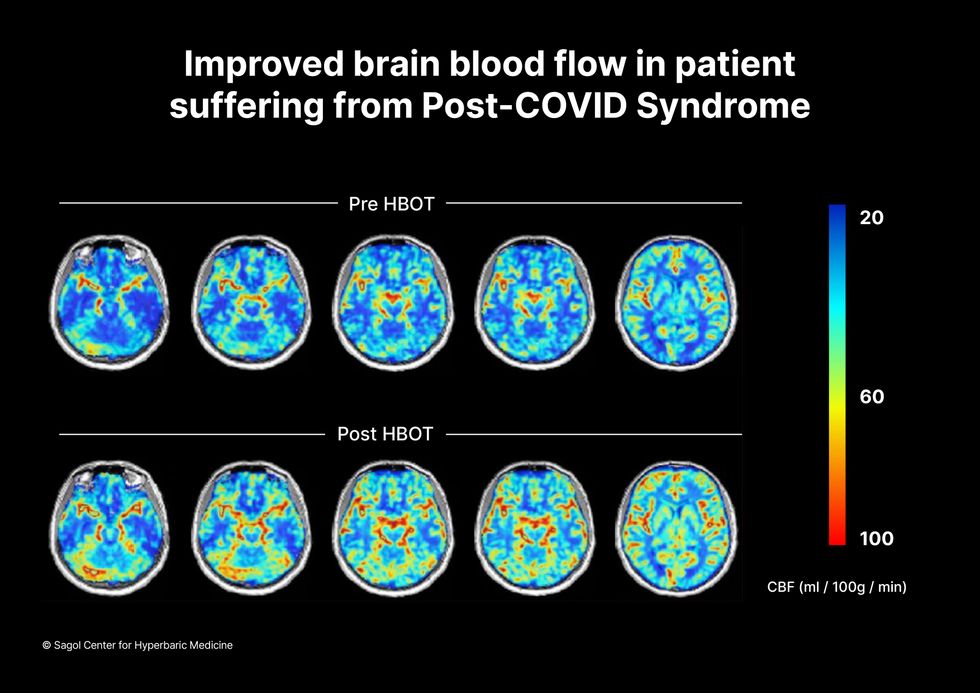
Scans show improved blood flow in a patient suffering from Long Covid.
Sagol Center for Hyperbaric Medicine
Efrati's use of HBOT is part of an emerging geroscience approach to diseases associated with aging. These researchers see systems dysfunctions that are common to several diseases, such as inflammation, which has been shown to play a role in micro infarcts, heart disease and Alzheimer’s disease. Preliminary research suggests that HBOT can retard some underlying mechanisms of aging, which might address several medical conditions. However, the drug approval process is set up to regulate individual disease, not conditions as broad as aging, and so they concentrate on treating the low hanging fruit: disorders where effective treatments currently are limited and success might be demonstrated.
The key to HBOT's effectiveness is something called the hyperoxic-hypoxic paradox where a body does not react to an increase in available oxygen, only to a decrease, regardless of the starting point. That danger signal has a powerful effect on gene expression, resulting in changes in metabolism, and the proliferation of stem cells. That occurs with each cycle of 20 minutes of pure oxygen followed by 5 minutes of regular air circulating through the masks, while the chamber remains pressurized. The high levels of oxygen in the blood provide the fuel necessary for tissue regeneration.
The hyperbaric chamber that Efrati has built can hold a dozen patients and attending medical staff. Think of it as a pressurized airplane cabin, only with much more space than even in first class. In the U.S., people think of HBOT as “a sack of air or some tube that you can buy on Amazon” or find at a health spa. “That is total bullshit,” Efrati says. “It has to be a medical class center where a physician can lose their license if they are not operating it properly.”

Shai Efrati
Alexander Charney, a research psychiatrist at the Icahn School of Medicine at Mount Sinai in New York City, calls Efrati’s study thoughtful and well designed. But it demands a lot from patients with its intense number of sessions. Those types of regimens have proven difficult to roll out to large numbers of patients. Still, the results are intriguing enough to merit additional trials.
John J. Miller, a physician and editor in chief of Psychiatric Times, has seen “many physicians that use hyperbaric oxygen for various brain disorders such as TBI.” He is intrigued by Efrati's work and believes the approach “has great potential to help patients with long COVID whose symptoms are related to brain tissue changes.”
Efrati believes so much in the power of the hyperoxic-hypoxic paradox to heal a variety of tissue injuries that he is leading the medical advisory board at Aviv Clinic, an international network of clinics that are delivering HBOT treatments based on research conducted in Israel. His goal is to silence doubters by quickly opening about 50 such clinics worldwide, based on the model of standalone dialysis clinics in the United States. Sagol Center is treating 300 patients per day, and clinics have opened in Florida and Dubai. There are plans to open another in Manhattan.
Avalanche rescue dogs train to find and dig out people buried in snow slides
Two-and-a-half year-old Huckleberry, a blue merle Australian shepherd, pulls hard at her leash; her yelps can be heard by skiers and boarders high above on the chairlift that carries them over the ski patrol hut to the top of the mountain. Huckleberry is an avalanche rescue dog — or avy dog, for short. She lives and works with her owner and handler, a ski patroller at Breckenridge Ski Resort in Colorado. As she watches the trainer play a game of hide-and-seek with six-month-old Lume, a golden retriever and avy dog-in-training, Huckleberry continues to strain on her leash; she loves the game. Hide-and-seek is one of the key training methods for teaching avy dogs the rescue skills they need to find someone caught in an avalanche — skier, snowmobiler, hiker, climber.
Lume’s owner waves a T-shirt in front of the puppy. While another patroller holds him back, Lume’s owner runs away and hides. About a minute later — after a lot of barking — Lume is released and commanded to “search.” He springs free, running around the hut to find his owner who reacts with a great amount of excitement and fanfare. Lume’s scent training will continue for the rest of the ski season (Breckenridge plans operating through May or as long as weather permits) and through the off-season. “We make this game progressively harder by not allowing the dog watch the victim run away,” explains Dave Leffler, Breckenridge's ski patroller and head of the avy dog program, who has owned, trained and raised many of them. Eventually, the trainers “dig an open hole in the snow to duck out of sight and gradually turn the hole into a cave where the dog has to dig to get the victim,” explains Leffler.
By the time he is three, Lume, like Huckleberry, will be a fully trained avy pup and will join seven other avy dogs on Breckenridge ski patrol team. Some of the team members, both human and canine, are also certified to work with Colorado Rapid Avalanche Deployment, a coordinated response team that works with the Summit County Sheriff’s office for avalanche emergencies outside of the ski slopes’ boundaries.
There have been 19 avalanche deaths in the U.S. this season, according to avalanche.org, which tracks slides; eight in Colorado. During the entirety of last season there were 17. Avalanche season runs from November through June, but avalanches can occur year-round.
High tech and high stakes
Complementing avy dogs’ ability to smell people buried in a slide, avalanche detection, rescue and recovery is becoming increasingly high tech. There are transceivers, signal locators, ground scanners and drones, which are considered “games changers” by many in avalanche rescue and recovery
For a person buried in an avalanche, the chance of survival plummets after 20 minutes, so every moment counts.
A drone can provide thermal imaging of objects caught in a slide; what looks like a rock from far away might be a human with a heat signature. Transceivers, also known as beacons, send a signal from an avalanche victim to a companion. Signal locators, like RECCO reflectors which are often sewn directly into gear, can echo back a radar signal sent by a detector; most ski resorts have RECCO detector units.
Research suggests that Ground Penetrating Radar (GPR), an electromagnetic tool used by geophysicists to pull images from inside the ground, could be used to locate an avalanche victim. A new study from the Department of Energy’s Sandia National Laboratories suggests that a computer program developed to pinpoint the source of a chemical or biological terrorist attack could also be used to find someone submerged in an avalanche. The search algorithm allows for small robots (described as cockroach-sized) to “swarm” a search area. Researchers say that this distributed optimization algorithm can help find avalanche victims four times faster than current search mechanisms. For a person buried in an avalanche, the chance of survival plummets after 20 minutes, so every moment counts.

An avy dog in training is picking up scent
Sarah McLear
While rescue gear has been evolving, predicting when a slab will fall remains an emerging science — kind of where weather forecasting science was in the 1980s. Avalanche forecasting still relies on documenting avalanches by going out and looking,” says Ethan Greene, director of the Colorado Avalanche Information Center (CAIC). “So if there's a big snowstorm, and as you might remember, most avalanches happened during snowstorms, we could have 10,000 avalanches that release and we document 50,” says Greene. “Avalanche forecasting is essentially pattern recognition,” he adds--and understanding the layering structure of snow.
However, determining where the hazards lie can be tricky. While a dense layer of snow over a softer, weaker layer may be a recipe for an avalanche, there’s so much variability in snowpack that no one formula can predict the trigger. Further, observing and measuring snow at a single point may not be representative of all nearby slopes. Finally, there’s not enough historical data to help avalanche scientists create better prediction models.
That, however, may be changing.
Last year, an international group of researchers created computer simulations of snow cover using 16 years of meteorological data to forecast avalanche hazards, publishing their research in Cold Regions Science and Technology. They believe their models, which categorize different kinds of avalanches, can support forecasting and determine whether the avalanche is natural (caused by temperature changes, wind, additional snowfall) or artificial (triggered by a human or animal).
With smell receptors ranging from 800 million for an average dog, to 4 billion for scent hounds, canines remain key to finding people caught in slides.
With data from two sites in British Columbia and one in Switzerland, researchers built computer simulations of five different avalanche types. “In terms of real time avalanche forecasting, this has potential to fill in a lot of data gaps, where we don't have field observations of what the snow looks like,” says Simon Horton, a postdoctoral fellow with the Simon Fraser University Centre for Natural Hazards Research and a forecaster with Avalanche Canada, who participated in the study. While complex models that simulate snowpack layers have been around for a few decades, they weren’t easy to apply until recently. “It's been difficult to find out how to apply that to actual decision-making and improving safety,” says Horton. If you can derive avalanche problem types from simulated snowpack properties, he says, you’ll learn “a lot about how you want to manage that risk.”
The five categories include “new snow,” which is unstable and slides down the slope, “wet snow,” when rain or heat makes it liquidly, as well as “wind-drifted snow,” “persistent weak layers” and “old snow.” “That's when there's some type of deeply buried weak layer in the snow that releases without any real change in the weather,” Horton explains. “These ones tend to cause the most accidents.” One step by a person on that structurally weak layer of snow will cause a slide. Horton is hopeful that computer simulations of avalanche types can be used by scientists in different snow climates to help predict hazard levels.
Greene is doubtful. “If you have six slopes that are lined up next to each other, and you're going to try to predict which one avalanches and the exact dimensions and what time, that's going to be really hard to do. And I think it's going to be a long time before we're able to do that,” says Greene.
What both researchers do agree on, though, is that what avalanche prediction really needs is better imagery through satellite detection. “Just being able to count the number of avalanches that are out there will have a huge impact on what we do,” Greene says. “[Satellites] will change what we do, dramatically.” In a 2022 paper, scientists at the University of Aberdeen in England used satellites to study two deadly Himalayan avalanches. The imaging helped them determine that sediment from a 2016 ice avalanche plus subsequent snow avalanches contributed to the 2021 avalanche that caused a flash flood, killing over 200 people. The researchers say that understanding the avalanches characteristics through satellite imagery can inform them how one such event increases the magnitude of another in the same area.

Avy dogs trainers hide in dug-out holes in the snow, teaching the dogs to find buried victims
Sarah McLear
Lifesaving combo: human tech and Mother Nature’s gear
Even as avalanche forecasting evolves, dogs with their built-in rescue mechanisms will remain invaluable. With smell receptors ranging from 800 million for an average dog, to 4 billion for scent hounds, canines remain key to finding people caught in slides. (Humans in comparison, have a meager 12 million.) A new study published in the Journal of Neuroscience revealed that in dogs smell and vision are connected in the brain, which has not been found in other animals. “They can detect the smell of their owner's fingerprints on a glass slide six weeks after they touched it,” says Nicholas Dodman, professor emeritus at Cummings School of Veterinary Medicine at Tufts University. “And they can track from a boat where a box filled with meat was buried in the water, 100 feet below,” says Dodman, who is also co-founder and president of the Center for Canine Behavior Studies.
Another recent study from Queens College in Belfast, United Kingdom, further confirms that dogs can smell when humans are stressed. They can also detect the smell of a person’s breath and the smell of the skin cells of a deceased person.
The emerging avalanche-predicting human-made tech and the incredible nature-made tech of dogs’ olfactory talents is the lifesaving “equipment” that Leffler believes in. Even when human-made technology develops further, it will be most efficient when used together with the millions of dogs’ smell receptors, Leffler believes. “It is a combination of technology and the avalanche dog that will always be effective in finding an avalanche victim.”
Living with someone changes your microbiome, new research shows
For the first time, research has shown that bacteria of the microbiome are transmitted between many individuals, not just infants and their mothers, in ways that can’t be explained by having the same diet or geography.
Some roommate frustration can be expected, whether it’s a sink piled high with crusty dishes or crumbs where a clean tabletop should be. Now, research suggests a less familiar issue: person-to-person transmission of shared bacterial strains in our gut and oral microbiomes. For the first time, the lab of Nicola Segata, a professor of genetics and computational biology at the University of Trento, located in Italy, has shown that bacteria of the microbiome are transmitted between many individuals, not just infants and their mothers, in ways that can’t be explained by their shared diet or geography.
It’s a finding with wide-ranging implications, yet frustratingly few predictable outcomes. Our microbiomes are an ever-growing and changing collection of helpful and harmful bacteria that we begin to accumulate the moment we’re born, but experts are still struggling to unravel why and how bacteria from one person’s gut or mouth become established in another person’s microbiome, as opposed to simply passing through.
“If we are looking at the overall species composition of the microbiome, then there is an effect of age of course, and many other factors,” Segata says. “But if we are looking at where our strains are coming from, 99 percent of them are only present in other people’s guts. They need to come from other guts.”
If we could better understand this process, we might be able to control and use it; perhaps hospital patients could avoid infections from other patients when their microbiome is depleted by antibiotics and their immune system is weakened, for example. But scientists are just beginning to link human microbiomes with various ailments. Growing evidence shows that our microbiomes steer our long-term health, impacting conditions like obesity, irritable bowel syndrome, type 2 diabetes, and cancer.
Previous work from Segata’s lab and others illuminated the ways bacteria are passed from mothers to infants during the first few months of life during vaginal birth, breastfeeding and other close contact. And scientists have long known that people in close proximity tend to share bacteria. But the factors related to that overlap, such as genetics and diet, were unclear, especially outside the mother-baby dyad.
“If we look at strain sharing between a mother and an infant at five years of age, for example, we cannot really tell which was due to transmission at birth and which is due to continued transmission because of contact,” Segata says. Experts hypothesized that they could be caused by bacterial similarities in the environment itself, genetics, or bacteria from shared foods that colonized the guts of people in close contact.
Strain sharing was highest in mother-child pairs, with 96 percent of them sharing strains, and only slightly lower in members of shared households, at 95 percent.
In Italy, researchers led by Mireia Valles-Colomer, including Segata, hoped to unravel this mystery. They compared data from 9,715 stool and saliva samples in 31 genomic datasets with existing metadata. Scientists zoomed in on variations in each bacterial strain down to the individual level. They examined not only mother-child pairs, but people living in the same household, adult twins, and people living in the same village in a level of detail that wasn’t possible before, due to its high cost and difficulties in retrieving data about interactions between individuals, Segata explained.
“This paper is, with high granularity, quantifying the percent sharing that you expect between different types of social interactions, controlling for things like genetics and diet,” Gibbons says. Strain sharing was highest in mother-child pairs, with 96 percent of them sharing strains, and only slightly lower in members of shared households, at 95 percent. And at least half of the mother-infant pairs shared 30 percent of their strains; the median was 12 percent among people in shared households. Yet, there was no sharing among eight percent of adult twins who lived separately, and 16 percent of people within villages who resided in different households. The results were published in Nature.
It’s not a regional phenomenon. Although the types of bacterial strains varied depending on whether people lived in western and eastern nations — datasets were drawn from 20 countries on five continents — the patterns of sharing were much the same. To establish these links, scientists focused on individual variations in shared bacterial strains, differences that create unique bacterial “fingerprints” in each person, while controlling for variables like diet, demonstrating that the bacteria had been transmitted between people and were not the result of environmental similarities.
The impact of this bacterial sharing isn’t clear, but shouldn’t be viewed with trepidation, according to Sean Gibbons, a microbiome scientist at the nonprofit Institute for Systems Biology.
“The vast majority of these bugs are actually either benign or beneficial to our health, and the fact that we're swapping and sharing them and that we can take someone else's strain and supplement or better diversify our own little garden is not necessarily a bad thing,” he says.
"There are hundreds of billions of dollars of investment capital moving into these microbiome therapeutic companies; bugs as drugs, so to speak,” says Sean Gibbons, a microbiome scientist at the Institute for Systems Biology.
Everyday habits like exercising and eating vegetables promote a healthy, balanced gut microbiome, which is linked to better metabolic and immune function, and fewer illnesses. While many people’s microbiomes contain bacteria like C. diff or E. coli, these bacteria don’t cause diseases in most cases because they’re present in low levels. But a microbiome that’s been wiped out by, say, antibiotics, may no longer keep these bacteria in check, allowing them to proliferate and make us sick.
“A big challenge in the microbiome field is being able to rationally predict whether, if you're exposed to a particular bug, it will stick in the context of your specific microbiome,” Gibbons says.
Gibbons predicts that explorations of microbe-based therapeutics will be “exploding” in the coming decades. “There are hundreds of billions of dollars of investment capital moving into these microbiome therapeutic companies; bugs as drugs, so to speak,” he says. Rather than taking a mass-marketed probiotic, a precise understanding of an individual’s microbiome could help target the introduction of just the right bacteria at just the right time to prevent or treat a particular illness.
Because the current study did not differentiate between different types of contact or relationships among household members sharing bacterial strains or determine the direction of transmission, Segata says his current project is examining children in daycare settings and tracking their microbiomes over time to understand the role genetics and everyday interactions play in the level of transmission that occurs.
This relatively newfound ability to trace bacterial variants to minute levels has unlocked the chance for scientists to untangle when and how bacteria leap from one microbiome to another. As researchers come to better understand the factors that permit a strain to establish itself within a microbiome, they could uncover new strategies to control these microbes, harnessing the makeup of each microbiome to help people to resist life-altering medical conditions.

