I'm a Transplant Surgeon Who Designed the Breakthrough That Saved My Own Life
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.
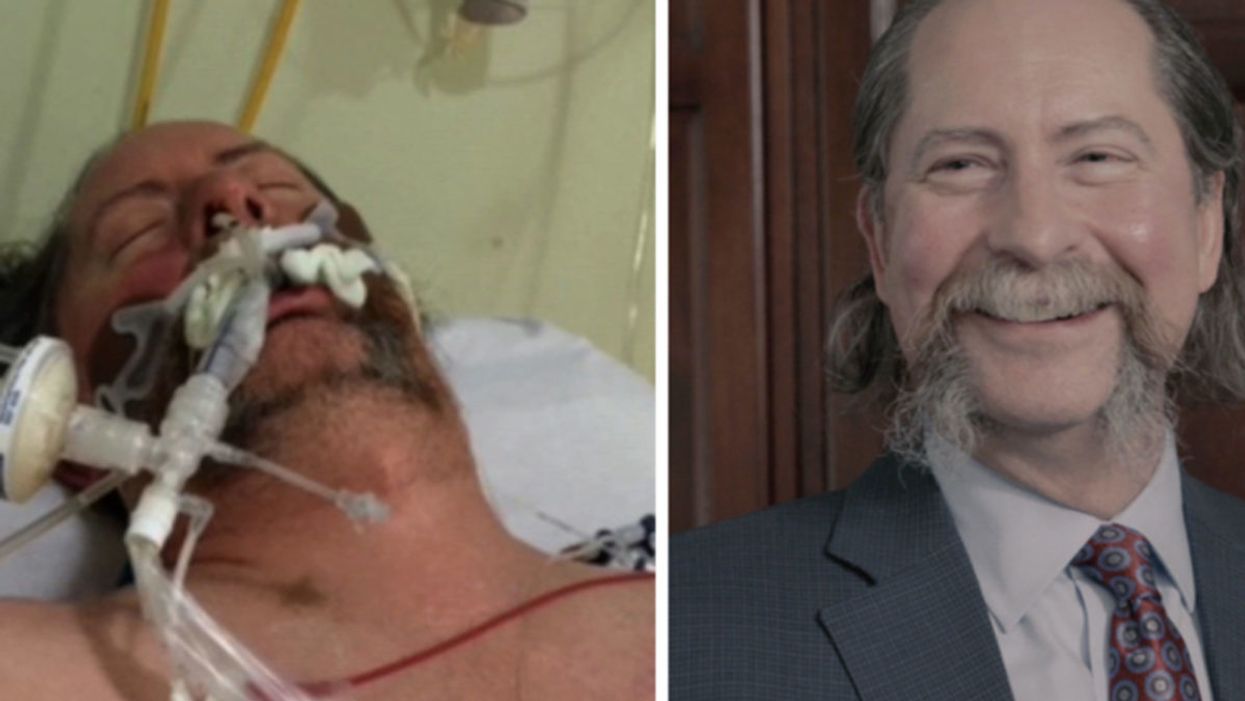
Dr. Robert Montgomery, the director of NYU Langone's Transplant Institute, is ironically the recipient of a heart transplant.
[Ed. Note: This is the fourth episode in our Moonshot series, which explores four cutting-edge scientific developments that stand to fundamentally transform our world.]
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.
How One Doctor Single-Handedly Saved Countless Babies from Birth Defects
President John F. Kennedy gave Dr. Frances Oldham Kelsey the nation's highest federal civilian service award in 1962, saying she had "prevented a major tragedy of birth deformities."
In July 1956, a new drug hit the European market for the first time. The drug was called thalidomide – a sedative that was considered so safe it was available without a prescription.
Sedatives were in high demand in post-war Europe – but barbiturates, the most widely-used sedative at the time, caused overdoses and death when consumers took more than the recommended amount. Thalidomide, on the other hand, didn't appear to cause any side effects at all: Chemie Grünenthal, thalidomide's manufacturer, dosed laboratory rodents with over 600 times the normal dosage during clinical testing and had observed no evidence of toxicity.
The drug therefore was considered universally safe, and Grünenthal supplied thousands of doctors with samples to give to their patients. Doctors were encouraged to recommend thalidomide to their pregnant patients specifically because it was so safe, in order to relieve the nausea and insomnia associated with the first trimester of pregnancy.
By 1960, Thalidomide was being sold in countries throughout the world, and the United States was expected to soon follow suit. Dr. Frances Oldham Kelsey, a pharmacologist and physician, was hired by the Food and Drug Administration (FDA) in September of that year to review and approve drugs for the administration. Immediately, Kelsey was tasked with approving thalidomide for commercial use in the United States under the name Kevadon. Kelsey's approval was supposed to be a formality, since the drug was so widely used in other countries.
But Kelsey did something that few people expected – she paused. Rather than approving the drug offhand as she was expected to do, Kelsey asked the manufacturer – William S. Merrell Co., who was manufacturing thalidomide under license from Chemie Grünenthal – to supply her with more safety data, noting that Merrell's application for approval relied mostly on anecdotal testimony. Kelsey – along with her husband who worked as a pharmacologist at the National Institutes of Health (NIH) — was highly suspicious of a drug that had no lethal dose and no side effects. "It was just too positive," Kelsey said later. "This couldn't be the perfect drug with no risk."
At the same time, rumors were starting to swirl across Europe that thalidomide was not as safe as everyone had initially thought: Physicians were starting to notice an "unusual increase" in the birth of severely deformed babies, and they were beginning to suspect thalidomide as the cause. The babies, whose mothers had all taken thalidomide during pregnancy, were born with conditions like deafness, blindness, congenital heart problems, and even phocomelia, a malformation of the arms and legs. Doctors and midwives were also starting to notice a sharp rise in miscarriages and stillbirths among their patients as well.
Kelsey's skepticism was rewarded in November 1961 when thalidomide was yanked abruptly off the market, following a growing outcry that it was responsible for hundreds of stillbirths and deformities.
Kelsey had heard none of these rumors, but she did know from her post-doctoral research that adults could metabolize drugs differently than fetuses – in other words, a drug that was perfectly safe for adults could be detrimental to a patient's unborn child. Noting that thalidomide could cross the placental barrier, she asked for safety data, such as clinical trials, that showed specifically the drug was non-toxic for fetuses. Merrell supplied Kelsey with anecdotal data – in other words, accounts from patients who attested to the fact that they took thalidomide with no adverse effects – but she rejected it, needing stronger data: clinical studies with pregnant women included.
The drug company was annoyed at what they considered Kelsey's needless bureaucracy. After all, Germans were consuming around 1 million doses of thalidomide every day in 1960, with lots of anecdotal evidence that it was safe, even among pregnant women. As the holidays approached – the most lucrative time of year for sedative sales – Merrell executives started hounding Kelsey to approve thalidomide, even phoning her superior and paying her visits at work. But Kelsey was unmovable. Kelsey's skepticism was solidified in December 1960, when she read a letter published in the British Medical Journal from a physician. In the letter, the author warned that his long-term thalidomide patients were starting to report pain in their arms and legs.
"The burden of proof that the drug is safe … lies with the applicant," Kelsey wrote in a letter to Merrell executive Joseph F. Murray in May of 1961. Despite increasing pressure, Kelsey held fast to her insistence that more safety data – particularly for fetuses – was needed.
Kelsey's skepticism was rewarded in November 1961 when Chemie Grünenthal yanked thalidomide off the market overseas, following a growing outcry that it was responsible for hundreds of stillbirths and deformities. In early 1962, Merrell conceded that the drug's safety was unproven in fetuses and formally withdrew its application at the FDA.
Thanks to Kelsey, the United States was spared the effects of thalidomide – although countries like Europe and Canada were not so lucky. Thalidomide remained in people's homes under different names long after it was pulled from the market, and so women unfortunately continued to take thalidomide during their pregnancies, unaware of its effects. All told, thalidomide is thought to have caused around 10,000 birth defects and anywhere from 5,000 to 7,000 miscarriages. Many so-called "thalidomide babies" are now adults living with disabilities.

Niko von Glasow, born in 1960, is a German film director and producer who was born disabled due to the side effects of thalidomide.
Wikimedia Commons
Just two years after joining the FDA, Kelsey was presented with the President's Award for Distinguished Federal Civilian Service and was appointed as the head of the Investigational Drug Branch at the FDA. Not only did Kelsey save the U.S. public from the horrific effects of thalidomide, but she forever changed the way drugs were developed and approved for use in the United States: Drugs now need to not only be proven safe and effective, but adverse drug reactions need to be reported to the FDA and informed consent must be obtained by all participants before they volunteer for clinical trials. Today, the United States is safer because of Frances Kelsey's bravery.
Angry Citizens Pressure the World Health Organization to Fully Recognize COVID’s Airborne Spread
Aerosol scientists say that the evidence points to airborne transmission of COVID-19 "beyond any reasonable doubt."
A new citizen movement is gathering steam to try to convince the influential World Health Organization to change its messaging about how the coronavirus is transmitted.
The new petition "COVID is Airborne" (www.covidisairborne.org) started in early November and has approximately 3,000 signatures. During this particularly dangerous acceleration of the pandemic, the petition's backers allege that the WHO is failing the public with mixed messaging and thus inadvertently fueling the wildfire of transmission.
"Early on in the pandemic, [WHO Director General Dr. Tedros Adhanom Ghebreyesus] said that coronavirus is airborne, but then in March, WHO tweeted that COVID-19 is not airborne, saying that it is primarily transmitted via droplets that are too heavy to hang in the air," says petition co-creator Jessica Bassett Allen.
The organization's late March messaging, still available on social media, is a digital graphic saying, "FACT CHECK: COVID-19 is NOT Airborne".

Screenshot of WHO's Tweet from March 28, 2020 that is still published.
The petition asks for a course correct: "We, citizens of the world, request that the World Health Organization (WHO) recognize the compelling scientific evidence that SARS-CoV-2 spreads by aerosol transmission ("airborne") and urge the WHO to immediately develop and initiate clear recommendations to enable people to protect themselves."
In the vacuum of the WHO's inaction, aerosol scientists around the world scrambled to raise awareness of what they saw as a grave error.
"Almost immediately after that [March 28] announcement, we formed a group of 239 scientists from many countries and disciplines to convince them that they should acknowledge that there is airborne transmission, but we find that they are totally dead set against it," says Dr. Jose Jimenez, a chemistry professor at the University of Colorado at Boulder who has studied aerosols for 20 years. He supports the citizen petition.
In a letter to the WHO back in July, he and his colleagues wrote: "Studies by the signatories and other scientists have demonstrated beyond any reasonable doubt that viruses are released during exhalation, talking, and coughing in microdroplets small enough to remain aloft in air and pose a risk of exposure at distances beyond 1–2 m from an infected individual."
The scientists have also gone direct to the public with their findings: They published a comprehensive Google doc with detailed answers to many people's frequently asked questions about how to protect themselves, addressing issues ranging from the best masks and air filters to how to deal with passing someone outdoors and much more.
It's worth noting that the CDC has now modified its COVID FAQ to include airborne transmission as a "less common way" for the virus to spread. This update took place after the CDC stated in September that it is "possible" the virus spreads via airborne transmission – only to reverse course and remove the language from its website several days later. The CDC's website now states that some viruses, including SARS-Cov-2, "may be able to infect people who are further than 6 feet away from the person who is infected or after that person has left the space."
Basset Allen notes that after the scientists' open letter, the WHO "added ventilation to public communications about how to prevent infection, but they haven't explained why."
When contacted, a WHO representative had no specific comment and shared its late March announcement as well as its latest guidelines on transmission. In part, its statement says, "Current evidence suggests that the main way the virus spreads is by respiratory droplets among people who are in close contact with each other. Aerosol transmission can occur in specific settings, particularly in indoor, crowded and inadequately ventilated spaces, where infected person(s) spend long periods of time with others, such as restaurants, choir practices, fitness classes, nightclubs, offices and/or places of worship. More studies are underway to better understand the conditions in which aerosol transmission is occurring outside of medical facilities where specific medical procedures, called aerosol-generating procedures, are conducted."
A forceful and clear message acknowledging the evidence could make it easier to standardize school and office ventilation, petitioners argue.
Aerosol scientist Jimenez was dismayed by the WHO's response.
"The first part is an error in my opinion," he says. "Current evidence suggests that the main way the virus spreads is inhalation of aerosols.…WHO is way behind, unfortunately.
"The second part is incomplete," Jimenez continues. "Aerosol transmission can happen in those indoor crowded low-ventilation spaces. But if aerosols can accumulate under those conditions and cause infection, they must be extremely infective in close proximity when talking, since they are much more concentrated there. Just like talking close to a smoker you would inhale much more smoke (which is an aerosol) than if you were in the same room, but let's say 10 or 15 feet away."
He adds, "The WHO and others are making the assumption that if this goes through the air, then everyone who is infected is putting a lot of virus into the air at all times, but we know that's wrong: People are infectious for a short period of time before and during their symptoms. In China, they have measured how much virus comes out of people, and they see that the emission is sporadic: The virus can come out in millions of viral [particles] per hour, but it doesn't happen all the time."
The petition's co-creator, Basset Allen, says that her life experience showed her the best way to make a change. "My involvement with this effort is entirely personal," she says. "I was first introduced to HIV treatment activism as a college student and what I learned about campaigning and power has been relevant in almost every other project I've worked on since then. HIV activism taught me that everyday people can win big, life-saving policy changes if they build expertise and work strategically to push decision makers."
The petition and its advocates argue that the WHO's mixed messaging is causing real harm. For instance, a forceful and clear message acknowledging the evidence could make it easier to standardize school and office ventilation, they argue. Anecdotally, some schools have refused to install HEPA filtration in their classrooms due to a lack of specific guidance from health agencies. (Note: The CDC now recommends improving central air filtration and considering the use of portable HEPA filters in classrooms.)
As the holidays approach, a clear and unified message from all influential health agencies would also help people understand why it is still important to wear masks while physical distancing, especially indoors.
"Personally, I cheered when I heard President-Elect Biden mention ventilation upgrades in schools during the first 10 minutes of his October town hall event, and again in the second debate," Basset Allen says. "Unfortunately, we're still more than two months away from the Biden administration taking over the U.S. COVID-19 response and we have to do absolutely everything we can right now to save as many lives as possible. Increasing awareness of airborne transmission and mitigation strategies can't wait. WHO can use its power to help close that gap, here and around the world."

