Is Finding Out Your Baby’s Genetics A New Responsibility of Parenting?
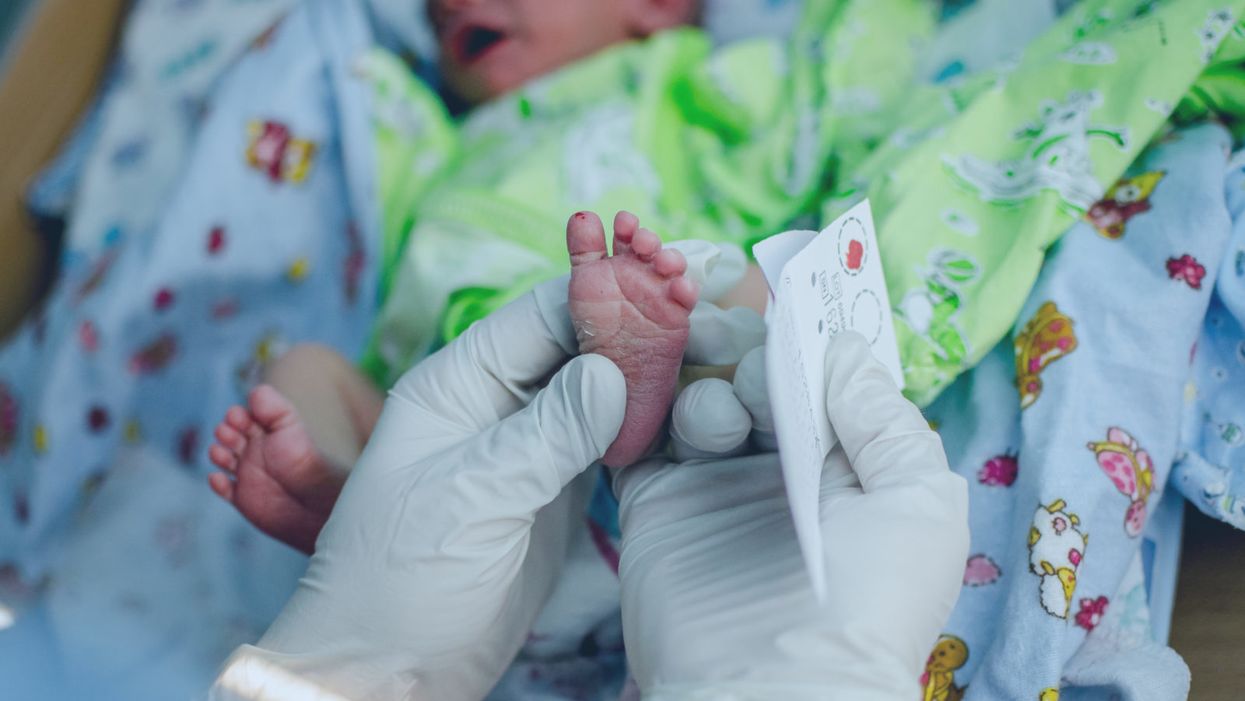
A doctor pricks the heel of a newborn for a blood test.
Hours after a baby is born, its heel is pricked with a lancet. Drops of the infant's blood are collected on a porous card, which is then mailed to a state laboratory. The dried blood spots are screened for around thirty conditions, including phenylketonuria (PKU), the metabolic disorder that kick-started this kind of newborn screening over 60 years ago. In the U.S., parents are not asked for permission to screen their child. Newborn screening programs are public health programs, and the assumption is that no good parent would refuse a screening test that could identify a serious yet treatable condition in their baby.
Learning as much as you can about your child's health might seem like a natural obligation of parenting. But it's an assumption that I think needs to be much more closely examined.
Today, with the introduction of genome sequencing into clinical medicine, some are asking whether newborn screening goes far enough. As the cost of sequencing falls, should parents take a more expansive look at their children's health, learning not just whether they have a rare but treatable childhood condition, but also whether they are at risk for untreatable conditions or for diseases that, if they occur at all, will strike only in adulthood? Should genome sequencing be a part of every newborn's care?
It's an idea that appeals to Anne Wojcicki, the founder and CEO of the direct-to-consumer genetic testing company 23andMe, who in a 2016 interview with The Guardian newspaper predicted that having newborns tested would soon be considered standard practice—"as critical as testing your cholesterol"—and a new responsibility of parenting. Wojcicki isn't the only one excited to see everyone's genes examined at birth. Francis Collins, director of the National Institutes of Health and perhaps the most prominent advocate of genomics in the United States, has written that he is "almost certain … that whole-genome sequencing will become part of new-born screening in the next few years." Whether that would happen through state-mandated screening programs, or as part of routine pediatric care—or perhaps as a direct-to-consumer service that parents purchase at birth or receive as a baby-shower gift—is not clear.
Learning as much as you can about your child's health might seem like a natural obligation of parenting. But it's an assumption that I think needs to be much more closely examined, both because the results that genome sequencing can return are more complex and more uncertain than one might expect, and because parents are not actually responsible for their child's lifelong health and well-being.
What is a parent supposed to do about such a risk except worry?
Existing newborn screening tests look for the presence of rare conditions that, if identified early in life, before the child shows any symptoms, can be effectively treated. Sequencing could identify many of these same kinds of conditions (and it might be a good tool if it could be targeted to those conditions alone), but it would also identify gene variants that confer an increased risk rather than a certainty of disease. Occasionally that increased risk will be significant. About 12 percent of women in the general population will develop breast cancer during their lives, while those who have a harmful BRCA1 or BRCA2 gene variant have around a 70 percent chance of developing the disease. But for many—perhaps most—conditions, the increased risk associated with a particular gene variant will be very small. Researchers have identified over 600 genes that appear to be associated with schizophrenia, for example, but any one of those confers only a tiny increase in risk for the disorder. What is a parent supposed to do about such a risk except worry?
Sequencing results are uncertain in other important ways as well. While we now have the ability to map the genome—to create a read-out of the pairs of genetic letters that make up a person's DNA—we are still learning what most of it means for a person's health and well-being. Researchers even have a name for gene variants they think might be associated with a disease or disorder, but for which they don't have enough evidence to be sure. They are called "variants of unknown (or uncertain) significance (VUS), and they pop up in most people's sequencing results. In cancer genetics, where much research has been done, about 1 in 5 gene variants are reclassified over time. Most are downgraded, which means that a good number of VUS are eventually designated benign.
While one parent might reasonably decide to learn about their child's risk for a condition about which nothing can be done medically, a different, yet still thoroughly reasonable, parent might prefer to remain ignorant so that they can enjoy the time before their child is afflicted.
Then there's the puzzle of what to do about results that show increased risk or even certainty for a condition that we have no idea how to prevent. Some genomics advocates argue that even if a result is not "medically actionable," it might have "personal utility" because it allows parents to plan for their child's future needs, to enroll them in research, or to connect with other families whose children carry the same genetic marker.
Finding a certain gene variant in one child might inform parents' decisions about whether to have another—and if they do, about whether to use reproductive technologies or prenatal testing to select against that variant in a future child. I have no doubt that for some parents these personal utility arguments are persuasive, but notice how far we've now strayed from the serious yet treatable conditions that motivated governments to set up newborn screening programs, and to mandate such testing for all.
Which brings me to the other problem with the call for sequencing newborn babies: the idea that even if it's not what the law requires, it's what good parents should do. That idea is very compelling when we're talking about sequencing results that show a serious threat to the child's health, especially when interventions are available to prevent or treat that condition. But as I have shown, many sequencing results are not of this type.
While one parent might reasonably decide to learn about their child's risk for a condition about which nothing can be done medically, a different, yet still thoroughly reasonable, parent might prefer to remain ignorant so that they can enjoy the time before their child is afflicted. This parent might decide that the worry—and the hypervigilence it could inspire in them—is not in their child's best interest, or indeed in their own. This parent might also think that it should be up to the child, when he or she is older, to decide whether to learn about his or her risk for adult-onset conditions, especially given that many adults at high familial risk for conditions like Alzheimer's or Huntington's disease choose never to be tested. This parent will value the child's future autonomy and right not to know more than they value the chance to prepare for a health risk that won't strike the child until 40 or 50 years in the future.
Parents are not obligated to learn about their children's risk for a condition that cannot be prevented, has a small risk of occurring, or that would appear only in adulthood.
Contemporary understandings of parenting are famously demanding. We are asked to do everything within our power to advance our children's health and well-being—to act always in our children's best interests. Against that backdrop, the need to sequence every newborn baby's genome might seem obvious. But we should be skeptical. Many sequencing results are complex and uncertain. Parents are not obligated to learn about their children's risk for a condition that cannot be prevented, has a small risk of occurring, or that would appear only in adulthood. To suggest otherwise is to stretch parental responsibilities beyond the realm of childhood and beyond factors that parents can control.
Men and Women Experience Pain Differently. Learning Why Could Lead to Better Drugs.
According to the CDC, one fifth of American adults live with chronic pain, and women are affected more than men.
It's been more than a decade since Jeannette Rotondi has been pain-free. A licensed social worker, she lives with five chronic pain diagnoses, including migraines. After years of exploring treatment options, doctors found one that lessened the pain enough to allow her to "at least get up."
"With all that we know now about genetics and the immune system, I think the future of pain medicine is more precision-based."
Before she says, "It was completely debilitating. I was spending time in dark rooms. I got laid off from my job." Doctors advised against pregnancy; she and her husband put off starting a family for almost a decade.
"Chronic pain is very unpredictable," she says. "You cannot schedule when you'll be in debilitative pain or cannot function. You don't know when you'll be hit with a flare. It's constantly in your mind. You have to plan for every possibly scenario. You need to carry water, medications. But you can't plan for everything." Even odors can serve as a trigger.
According to the CDC, one fifth of American adults live with chronic pain, and women are affected more than men. Do men and women simply vary in how much pain they can handle? Or is there some deeper biological explanation? The short answer is it's a little of both. But understanding the biological differences can enable researchers to develop more effective treatments.
While studies in animals are straightforward (they either respond to pain or they don't), humans are more complex. Social and psychological factors can affect the outcome. For example, one Florida study found that gender role expectations influenced pain sensitivity.
"If you are a young male and you believe very strongly that men are tougher than women, you will have a much higher threshold and will be less sensitive to pain," says Robert Sorge, an associate professor at the University of Alabama at Birmingham whose lab researches the immune system's involvement in pain and addiction.
He also notes, "We looked at transgender women and their pain sensitivity in comparison to cis men and women. They show very similar pain sensitivity to cis women, so that may reduce the impact of genetic sex in terms of what underlies that sensitivity."
But the difference goes deeper than gender expectations. There are biological differences as well. In 2015, Sorge and his team discovered that pain stimuli activated different immune cells in male and female rodents and that the presence of testosterone seemed to be a factor in the response.
More recently, Ted Price, professor of neuroscience at University of Texas, Dallas, examined pain at a genetic level, specifically looking at the patterns of RNA, which are single-stranded molecules that act as a messenger for DNA. Price noted that there were differences in these patterns that coincided with whether an individual experienced pain.
Price explains, "Every cell in your body has DNA, but the RNA that is in the cells is different for every cell type. The RNA in any particular cell type, like a neuron, can change as a result of some environmental influence like an injury. We found a number of genes that are potentially causative factors for neuropathic pain. Those, interestingly, seemed to be different between men and women."
Differences in treatment also affect pain response. Sorge says, "Women are experiencing more pain dismissal and more hostility when they report chronic pain. Women are more likely to have their pain associated with psychological issues." He adds that this dismissal may require women to exaggerate symptoms in order to be believed.
This can impact pain management. "Women are more likely to be prescribed and to use opioids," says Dr. Roger B. Fillingim, Director of Pain Research and Intervention Center of Excellence at the University of Florida. Yet, when self-administering pain meds, "women used significantly less opioids after surgery than did men." He also points out that "men are at greater risk for dose escalation and for opioid-related death than are women. So even though more women are using opioids, men are more likely to die from opioid-related causes."
Price acknowledges that other drugs treat pain, but "unfortunately, for chronic pain, none of these drugs work very well. We haven't yet made classes of drugs that really target the underlying mechanism that causes people to have chronic pain."
New drugs are now being developed that "might be particularly efficacious in women's chronic pain."
Sorge points out that there are many variables in pain conditions, so drugs that work for one may be ineffective for another. "With all that we know now about genetics and the immune system, I think the future of pain medicine is more precision-based, where based on your genetics, your immune status, your history, we may eventually get to the point where we can say [certain] drugs have a much bigger chance of working for you."
It will take some time for these new discoveries to translate into effective treatments, but Price says, "I'm excited about the opportunities. DNA and RNA sequencing totally changes our ability to make these therapeutics. I'm very hopeful." New drugs are now being developed that "might be particularly efficacious in women's chronic pain," he says, because they target specific receptors that seem to be involved when only women experience pain.
Earlier this year, three such drugs were approved to treat migraines; Rotondi recently began taking one. For Rotondi, improved treatments would allow her to "show up for life. For me," she says, "it would mean freedom."
Deaf Scientists Just Created Over 1000 New Signs to Dramatically Improve Ability to Communicate
Sarah Latchney, the first deaf PhD student at the University of Rochester School of Medicine & Dentistry, pictured here at work at a lab in the department of environmental sciences.
For the deaf, talent and hard work may not be enough to succeed in the sciences. According to the National Science Foundation, deaf Americans are vastly underrepresented in the STEM fields, a discrepancy that has profound economic implications.
The problem with STEM careers for the deaf and hard-of-hearing is that there are not enough ASL signs available.
Deaf and hard-of-hearing professionals in the sciences earn 31 percent more than those employed in other careers, according to a 2010 study by the National Technical Institute for the Deaf (NTID) in Rochester, N.Y., the largest technical college for deaf and hard-of-hearing students. But at the same time, in 2017, U.S. students with hearing disabilities earned only 1.1 percent of the 39,435 doctoral degrees awarded in science and engineering.
One reason so few deaf students gravitate to science careers and may struggle to complete doctoral programs is the communication chasm between deaf and hard-of-hearing scientists and their hearing colleagues.
Lorne Farovitch is a doctoral candidate in biomedical science at the University of Rochester of New York. Born deaf and raised by two deaf parents, he communicated solely in American Sign Language (ASL) until reaching graduate school. There, he became frustrated at the large chunk of his workdays spent communicating with hearing lab mates and professors, time he would have preferred spending on his scientific work.
The problem with STEM careers for the deaf and hard-of-hearing is that there are not enough ASL signs available, says Farovitch. Names, words, or phrases that don't exist in ASL must be finger spelled — the signer must form a distinct hand shape to correspond with each letter of the English alphabet, a tedious and time-consuming process. For instance, it requires 12 hand motions to spell out the word M-I-T-O-C-H-O-N-D-R-I-A. Imagine repeating those motions countless times a day.
To bust through this linguistic quagmire, Farovitch, along with a team of deaf STEM professionals, linguists, and interpreters, have been cooking up signs for terms like Anaplasma phagocytophilum, the tick-borne bacterium Farovitch studies. The sign creators are then videotaped performing the new signs. Those videos are posted on two crowd-sourcing sites, ASLcore.org and ASL Clear.
The beauty of ASL is you can express an entire concept in a single sign, rather than by the name of a word.
"If others don't pick it up and use it, a sign goes extinct," says Farovitch. Thus far, more than 1,000 STEM terms have been developed on ASL Clear and 500 vetted and approved by the deaf STEM community, according to Jeanne Reis, project director of the ASL Clear Project, based at The Learning Center for the Deaf in Framingham, Mass.
The beauty of ASL is you can express an entire concept in a single sign, rather than by the name of a word. The signs are generally intuitive and wonderfully creative. To express "DNA" Farovitch uses two fingers of each hand touching the tips of the opposite hand; then he draws both the hands away to suggest the double helix form of the hereditary material present in most organisms.
"If you can show it, you can understand the concept better,'' says the Canadian-born scientist. "I feel I can explain science better now."
The hope is that as ASL science vocabulary expands more, deaf and hard-of-hearing students will be encouraged to pursue the STEM fields. "ASL is not just a tool; it's a language. It's a vital part of our lives," Farovitch explains through his interpreter.
The deaf community is diverse—within and beyond the sciences. Sarah Latchney, PhD, an environmental toxicologist, is among the approximately 90 percent of deaf people born to hearing parents. Hers made sure she learned ASL at an early age but they also sent Latchney to a speech therapist to learn to speak and read lips. Latchney is so adept at both that she can communicate one-on-one with a hearing person without an interpreter.
Like Favoritch, Latchney has developed "conceptually accurate" ASL signs but she has no plans to post them on the crowd-sourcing sites. "I don't want to fix [my signs]; it works for me," she explains.
Young scientists like Farovitch and Latchney stress the need for interpreters who are knowledgeable about science. "When I give a presentation I'm a nervous wreck that I'll have an interpreter who may not have a science background," Latchney explains. "Many times what I've [signed] has been misinterpreted; either my interpreter didn't understand the question or didn't frame it correctly."
To enlarge the pool of science-savvy interpreters, the University of Rochester will offer a new masters degree program: ASL Interpreting in Medicine and Science (AIMS), which will train interpreters who have a strong background in the biological sciences.
Since the Americans with Disabilities Act was enacted in 1990, opportunities in higher education for deaf and hard-of-hearing students have opened up in the form of federally funded financial aid and the creation of student disability services on many college campuses. Still, only 18 percent of deaf adults have graduated from college, compared to 33 percent of the general population, according to a survey by the U.S. Census Bureau in 2015.
The University of Rochester and the Rochester Institute of Technology, home to NTID, have jointly created two programs to increase the representation of deaf and hard-of-hearing professionals in the sciences. The Rochester Bridges to the Doctorate Program, which Farovitch is enrolled in, prepares deaf scholars for biomedical PhD programs. The Rochester Postdoctoral Partnership readies deaf postdoctoral scientists to successfully attain academic research and teaching careers. Both programs are funded by the National Institutes of Science. In the last five years, the University of Rochester has gone from zero deaf postdoctoral and graduate students to nine.
"Deafness is not a problem, it's just a difference."
It makes sense for these two private universities to support strong programs for the deaf: Rochester has the highest per capita population of deaf or hard-of-hearing adults younger than 65 in the nation, according to the U.S. Census. According to the U.S. Department of Education, there are about 136,000 post-secondary level students who are deaf or hard of hearing.
"Deafness is not a problem, it's just a difference," says Farovitch. "We just need a different way to communicate. It doesn't mean we require more work."

