This Resistance Fighter Invented Dialysis in Nazi-Occupied Holland
When Willem Johan Kolff invented dialysis, the "father" of artificial organs was just getting started.
One of the Netherlands’ most famous pieces of pop culture is “Soldier of Orange.” It’s the title of the country’s most celebrated war memoir, movie and epic stage musical, all of which detail the exploits of the nation’s resistance fighters during World War II.
Willem Johan Kolff was a member of the Dutch resistance, but he doesn’t rate a mention in the “Solider of Orange” canon. Yet his wartime toils in a rural backwater not only changed medicine, but the world.
Kolff had been a physician less than two years before Germany invaded the Netherlands in May 1940. He had been engaged in post-graduate studies at the University of Gronigen but withdrew because he refused to accommodate the demands of the Nazi occupiers. Kolff’s Jewish supervisor made an even starker choice: He committed suicide.
After his departure from the university, Kolff took a job managing a small hospital in Kampen. Located 50 miles from the heavily populated coastal region, the facility was far enough away from the prying eyes of Germans that not only could Kolff care for patients, he could hide fellow resistance fighters and even Jewish refugees in relative safety. Kolff coached many of them to feign convincing terminal illnesses so the Nazis would allow them to remain in the hospital.
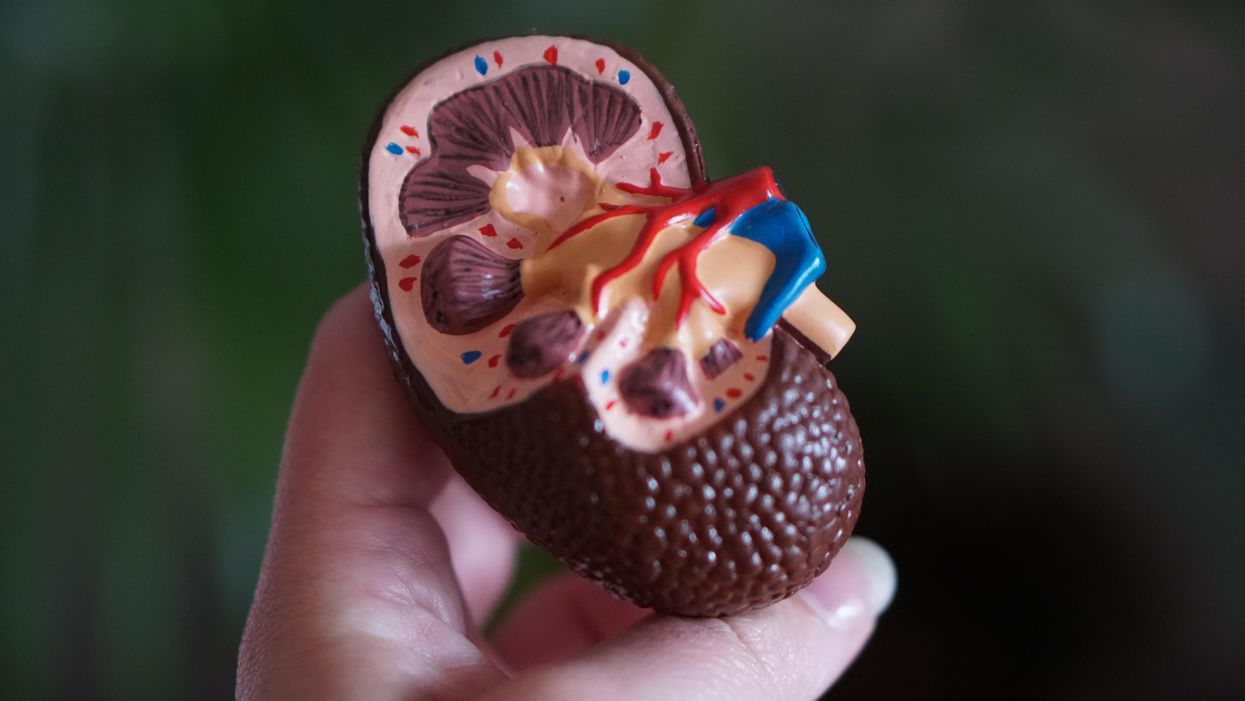
Despite the demands of practicing medicine and resistance work, Kolff still found time to conduct research. He had been haunted and inspired when, not long before the Nazi invasion, one of his patients died in agony from kidney disease. Kolff wanted to find a way to save future patients.
He broke his problem down to a simple task: If he could remove 20 grams of urea from a patient’s blood in 24 hours, they would survive. He began experimenting with ways to filter blood and return it to a patient’s body. Since the war had ground all non-military manufacturing to a halt, he was mostly forced to make do with material he could find at the hospital and around Kampen. Kolff eventually built a device from a washing machine parts, juice cans, sausage casings, a valve from an old Ford automobile radiator, and even scrap from a downed German aircraft.
The world’s first dialysis machine was hardly imposing; it resembled a rotating drum for a bingo game or raffle. Yet it carried on the highly sophisticated task of moving a patient’s blood through a semi-permeable membrane (about a 50-foot length of sausage casings) into a saline solution that drew out urea while leaving the blood cells untouched.
In emigrating to the U.S. to practice medicine, Kolff's intent was twofold: Advocate for a wider adoption of dialysis, and work on new projects. He wildly succeeded at both.
Kolff began using the machine to treat patients in 1943, most of whom had lapsed into comas due to their kidney failure. But like most groundbreaking medical devices, it was not an immediate success. By the end of the war, Kolff had dialyzed more than a dozen patients, but all had died. He briefly suspended use of the device after the Allied invasion of Europe, but he continued to refine its operation and the administration of blood thinners to patients.
In September 1945, Kolff dialyzed another comatose patient, 67-year-old Sofia Maria Schafstadt. She regained consciousness after 11 hours, and would live well into the 1950s with Kolff’s assistance. Yet this triumph contained a dark irony: At the time of her treatment, Schafstadt had been imprisoned for collaborating with the Germans.
With a tattered Europe struggling to overcome the destruction of the war, Kolff and his family emigrated to the U.S. in 1950, where he began working for the Cleveland Clinic while undergoing the naturalization process so he could practice medicine in the U.S. His intent was twofold: Advocate for a wider adoption of dialysis, and work on new projects. He wildly succeeded at both.
By the mid-1950s, dialysis machines had become reliable and life-saving medical devices, and Kolff had become a U.S. citizen. About that time he invented a membrane oxygenator that could be used in heart bypass surgeries. This was a critical component of the heart-lung machine, which would make heart transplants possible and bypass surgeries routine. He also invented among the very first practical artificial hearts, which in 1957 kept a dog alive for 90 minutes.
Kolff moved to the University of Utah in 1967 to become director of its Institute for Biomedical Engineering. It was a promising time for such a move, as the first successful transplant of a donor heart to a human occurred that year. But he was interested in going a step further and creating an artificial heart for human use.
It took more than a decade of tinkering and research, but in 1982, a team of physicians and engineers led by Kolff succeeded in implanting the first artificial heart in dentist Barney Clark, whose failing health disqualified him from a heart transplant. Although Clark died in March 1983 after 112 days tethered to the device, that it kept him alive generated international headlines. While graduate student Robert Jarvik received the named credit for the heart, he was directly supervised by Kolff, whose various endeavors into artificial organ research at the University of Utah were segmented into numerous teams.
Forty years later, several artificial hearts have been approved for use by the Food and Drug Administration, although all are a “bridge” that allow patients to wait for a transplant.
Kolff continued researching and tinkering with biomedical devices – including artificial eyes and ears – until he retired in 1997 at the age of 86. When he died in 2009, the medical community acknowledged that he was not only a pioneer in biotechnology, but the “father” of artificial organs.
Podcast: Pfizer's Head of Medicine Design Discusses the Newly Authorized Anti-Covid Pill
Charlotte Allerton, a chemist who is Pfizer's Head of Medicine Design, shares why Paxlovid is a significant breakthrough for the treatment of new COVID infections.
The "Making Sense of Science" podcast features interviews with leading medical and scientific experts about the latest developments and the big ethical and societal questions they raise. This monthly podcast is hosted by journalist Kira Peikoff, founding editor of the award-winning science outlet Leaps.org.
This month, Pfizer's Head of Medicine Design shares timely insights on this important breakthrough, including how the pill works, the impressive results of the recent studies, its encouraging profile against Omicron, its expected ability to be effective for both vaccinated and unvaccinated individuals, and why it could alter the trajectory of the pandemic in 2022.
Watch the 60-second trailer
LISTEN TO THE WHOLE EPISODE:
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.
People With This Rare Disease Can Barely Eat Protein. Biotechnology May Change That.
The Brown family at the Grand Tetons (2019). Clockwise from left, Christine, Kevin, Keagan, Connor, and Kellen.
Imagine that the protein in bread, eggs, steak, even beans is not the foundation for a healthy diet, but a poison to your brain. That is the reality for people living with Phenylketonuria, or PKU. This cluster of rare genetic variations affects the ability to digest phenylalanine (Phe), one of the chemical building blocks of protein. The toxins can build up in the brain causing severe mental retardation.
Can a probiotic help digest the troublesome proteins before they can enter the bloodstream and travel to the brain? A Boston area biotech start up, Synlogic, believes it can. Their starting point is an E. coli bacterium that has been used as a probiotic for more than a century. The company then screened thousands of gene variants to identify ones that produced enzymes most efficient at slicing and dicing the target proteins and optimized them further through directed evolution. The results have been encouraging.
But Christine Brown knew none of this when the hospital called saying that standard newborn screening of her son Connor had come back positive for PKU. It was urgent that they visit a special metabolic clinic the next day, which was about a three-hour drive away.
“I was told not to go on the Internet,” Christine recalls, “So when somebody tells you not to go on the Internet, what do you do? Even back in 2005, right.” What she saw were the worst examples of retardation, which was a common outcome from PKU before newborn screening became routine. “We were just in complete shell shock, our whole world just kind of shattered and went into a tail spin.”
“I remember feeding him the night before our clinic visit and almost feeling like I was feeding him poison because I knew that breast milk must have protein in it,” she says.
“Some of my first memories are of asking, ‘Mommy, can I eat this? There were yes foods and no foods.'"
Over the next few days the dedicated staff of the metabolic clinic at the Waisman Center at the University of Wisconsin Madison began to walk she and husband Kevin back from that nightmare. They learned that a simple blood test to screen newborns had been developed in the early 1960s to detect PKU and that the condition could be managed with stringent food restrictions and vigilant monitoring of Phe levels.
Everything in Your Mouth Counts
PKU can be successfully managed with a severely restricted diet. That simple statement is factually true, but practically impossible to follow, as it requires slashing protein consumption by about 90 percent. To compensate for the missing protein, several times a day PKU patients take a medical formula – commonly referred to simply as formula – containing forms of proteins that are digestible to their bodies. Several manufacturers now add vitamins and minerals and offer a variety of formats and tastes to make it more consumer friendly, but that wasn't always the case.
“When I was a kid, it tasted horrible, was the consistency of house paint. I didn't think about it, I just drank it. I didn't like it but you get used to it after a while,” recalls Jeff Wolf, the twang of Appalachia still strong in his voice. Now age 50, he grew up in Ashland, Kentucky and was part of the first wave of persons with PKU who were identified at birth as newborn screening was rolled out across the US. He says the options of taste and consistency have improved tremendously over the years.
Some people with PKU are restricted to as little as 8 grams of protein a day from food. That's a handful of almonds or a single hard-boiled egg; a skimpy 4-ounce hamburger and slice of cheese adds up to half of their weekly protein ration. Anything above that daily allowance is more than the body can handle and toxic levels of Phe begin to accumulate in the brain.
“Some of my first memories are of asking, ‘Mommy, can I eat this? There were yes foods and no foods,’” recalls Les Clark. He has never eaten a hamburger, steak, or ribs, practically a sacrilege for someone raised in Stanton, a small town in northeastern Nebraska, a state where the number of cattle and hogs are several-fold those of people.
His grandmother learned how to make low protein bread, but it looked and tasted different. His mom struggled making birthday cakes. “I learned some bad words at a very young age” as mom struggled applying icing that would pull the cake apart or a slice would collapse into a heap of crumbs, Les recalls.

Les Clark with a birthday cake.
Courtesy Clark
Controlling the diet “is not so bad when you are a baby” because that's all you know, says Jerry Vockley, Director of the Center for Rare Disease Therapy at Children's Hospital of Pittsburgh. “But after a while, as you get older and you start tasting other things and you say, Well, gee, this stuff tastes way better than what you're giving me. What's the deal? It becomes harder to maintain the diet.”
First is the lure of forbidden foods as children venture into the community away from the watchful eyes of parents. The support system weakens further when they leave home for college or work. “Pizza was mighty tasty,” Wolf' says of his first slice.
Vockley estimates that about 90 percent of adults with PKU are off of treatment. Moving might mean finding a new metabolic clinic that treats PKU. A lapse in insurance coverage can be a factor. Finally there is plain fatigue from multiple daily dosing of barely tolerable formula, monitoring protein intake, and simply being different in terms of food restrictions. Most people want to fit in and not be defined by their medical condition.
Jeff Wolf was one of those who dropped out in his twenties and thirties. He stopped going to clinic, monitoring his Phe levels, and counting protein. But the earlier experience of living with PKU never completely left the back of his mind; he listened to his body whenever eating too much protein left him with the “fuzzy brain" of a protein hangover. About a decade ago he reconnected with a metabolic clinic, began taking formula and watching his protein intake. He still may go over his allotment for a single day but he tries to compensate on subsequent days so that his Phe levels come back into balance.

Jeff Wolf on a boat.
Courtesy Wolf
One of the trickiest parts of trying to manage phenylalanine intake is the artificial sweetener aspartame. The chemical is ubiquitous in diet and lite foods and drinks. Gum too, you don't even have to swallow to receive a toxic dose of Phe. Most PKUers say it is easier to simply avoid these products entirely rather than try to count their Phe content.
Treatments
Most rare diseases have no treatment. There are two drugs for PKU that provide some benefit to some portion of patients but those drugs often have their own burdens.
KUVAN® (sapropterin dihydrochloride) is a pill or powder that helps correct a protein folding error so that food proteins can be digested. It is approved for most types of PKU in adults and children one month and older, and often is used along with a protein-restricted diet.
“The problem is that it doesn't work for every [patient's genetic] mutation, and there are hundreds of mutations that have been identified with PKU. Two to three percent of patients will have a very dramatic response and if you're one of those small number of patients, it's great,” says Vockley. “If you have one of the other mutation, chances are pretty good you still are going to end up on a restricted diet.”
PALYNZIQ® (pegvaliase-pqpz) “has the potential to lower the Phe to normal levels, it's a real breakthrough in the field,” says Vockley. “But is a very hard drug to use. Most folks have to take either one or two 2ml injections a day of something that is basically a gel, and some individuals have to take three.”
Many PKUers have reactions at the site of the injection and some develop anaphylaxis, a severe potentially life-threatening allergic reaction that can happen within seconds and can occur at any time, even after long term use. Many patients using Palynziq carry an EpiPen, a self-injection devise containing a form of adrenaline that can reverse some of the symptoms of anaphylaxis.
Then there is the cost. With the Kuvan dosing for an adult, “you're talking between $100,000 and $200,000 a year. And Palynziq is three times that,” says Vockley. Insurance coverage through a private plan or a state program is essential. Some state programs provide generous coverage while others are skimpy. Most large insurance company plans cover the drugs, sometimes with significant copays, but companies that are self-insured are under no legal obligation to provide coverage.
Les Clark found that out the hard way when the company he worked for was sold. The new owner was self-insured and declined to continue covering his drugs. Almost immediately he was out of pocket an additional $1500 a month for formula, and that was with a substantial discount through the manufacturer's patient support program. He says, “If you don't have an insurance policy that will cover the formula, it's completely unaffordable.” He quickly began to look for a new job.
Hope
It's easy to see why PKUers are eager for advances that will make managing their condition more effect, easier, and perhaps more affordable. Synlogic's efforts have drawn their attention and raised hopes.
Just before Thanksgiving Jerry Vockley presented the latest data to a metabolism conference meeting in Australia. There were only 8 patients in this group of a phase 2 trial using the original version of the company's lead E. coli product, SYNB1618, but they were intensely studied. Each was given the probiotic and then a challenge meal. Vockley saw a 40% reduction in Phe absorption and later a 20% reduction in mean fasting Phe levels in the blood. The product was easy to use and tolerate.
The company also presented early results for SYNB1934, a follow on version that further genetically tweaked the E. coli to roughly double the capacity to chop up the target proteins. Synlogic is recruiting patients for studies to determine the best dosing, which they are planning for next year.
“It's an exciting approach,” says Lex Cowsert, Director of Research Development at the National PKU Alliance, a nonprofit that supports the patient, family, and research communities involved with PKU. “Every patient is different, every patient has a different tolerance for the type of therapy that they are willing to pursue,” and if it pans out, it will be a welcome addition, either alone or in combination with other approaches, to living with PKU.
Author's Note: Reporting this story was made possible by generous support from the National Press Foundation and the Fondation Ipsen. Thanks to the people who so generously shared their time and stories in speaking with me.

