Lab-Grown Mini Kidneys Are Bringing Science Closer to Custom Organs
A model of a human kidney.
Science's dream of creating perfect custom organs on demand as soon as a patient needs one is still a long way off. But tiny versions are already serving as useful research tools and stepping stones toward full-fledged replacements.
Although organoids cannot yet replace kidneys, they are invaluable tools for research.
The Lowdown
Australian researchers have grown hundreds of mini human kidneys in the past few years. Known as organoids, they function much like their full-grown counterparts, minus a few features due to a lack of blood supply.
Cultivated in a petri dish, these kidneys are still a shadow of their human counterparts. They grow no larger than one-sixth of an inch in diameter; fully developed organs are up to five inches in length. They contain no more than a few dozen nephrons, the kidney's individual blood-filtering unit, whereas a fully-grown kidney has about 1 million nephrons. And the dish variety live for just a few weeks.
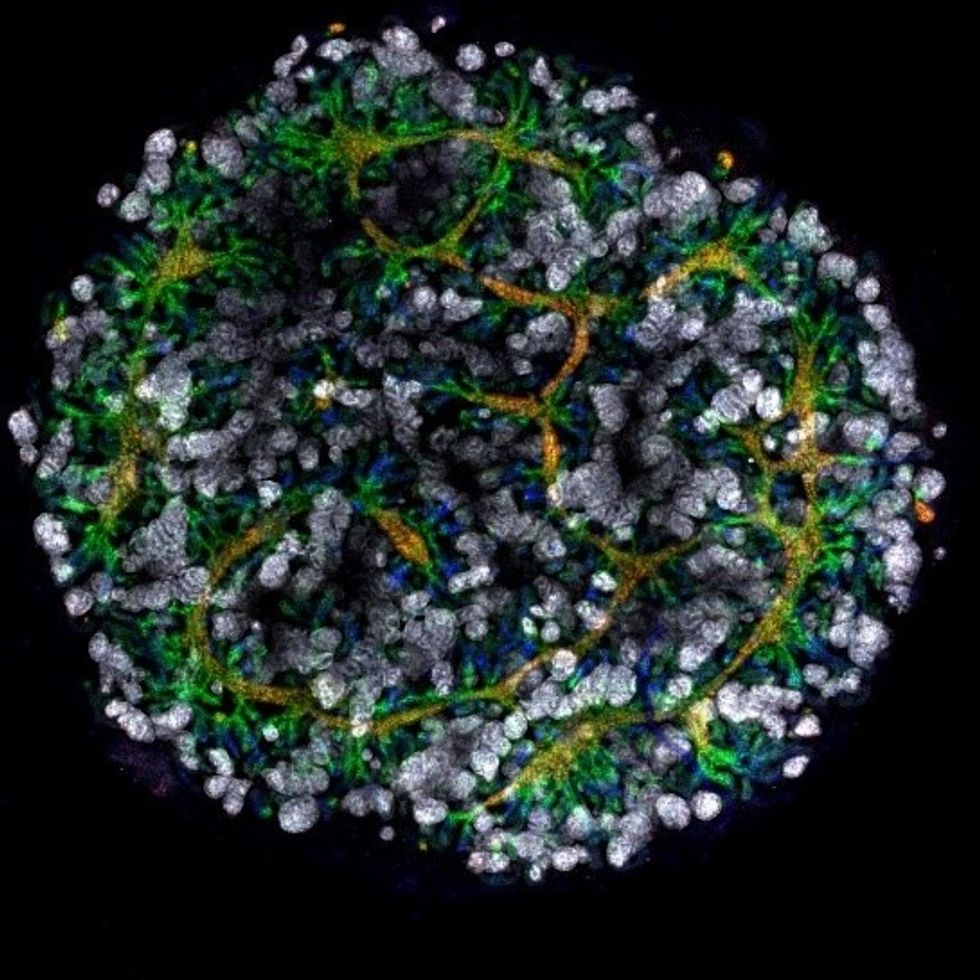
An organoid kidney created by the Murdoch Children's Institute in Melbourne, Australia.
Photo Credit: Shahnaz Khan.
But Melissa Little, head of the kidney research laboratory at the Murdoch Children's Institute in Melbourne, says these organoids are invaluable tools for research. Although renal failure is rare in children, more than half of those who suffer from such a disorder inherited it.
The mini kidneys enable scientists to better understand the progression of such disorders because they can be grown with a patient's specific genetic condition.
Mature stem cells can be extracted from a patient's blood sample and then reprogrammed to become like embryonic cells, able to turn into any type of cell in the body. It's akin to walking back the clock so that the cells regain unlimited potential for development. (The Japanese scientist who pioneered this technique was awarded the Nobel Prize in 2012.) These "induced pluripotent stem cells" can then be chemically coaxed to grow into mini kidneys that have the patient's genetic disorder.
"The (genetic) defects are quite clear in the organoids, and they can be monitored in the dish," Little says. To date, her research team has created organoids from 20 different stem cell lines.
Medication regimens can also be tested on the organoids, allowing specific tailoring for each patient. For now, such testing remains restricted to mice, but Little says it eventually will be done on human organoids so that the results can more accurately reflect how a given patient will respond to particular drugs.
Next Steps
Although these organoids cannot yet replace kidneys, Little says they may plug a huge gap in renal care by assisting in developing new treatments for chronic conditions. Currently, most patients with a serious kidney disorder see their options narrow to dialysis or organ transplantation. The former not only requires multiple sessions a week, but takes a huge toll on patient health.
Ten percent of older patients on dialysis die every year in the U.S. Aside from the physical trauma of organ transplantation, finding a suitable donor outside of a family member can be difficult.
"This is just another great example of the potential of pluripotent stem cells."
Meanwhile, the ongoing creation of organoids is supplying Little and her colleagues with enough information to create larger and more functional organs in the future. According to Little, researchers in the Netherlands, for example, have found that implanting organoids in mice leads to the creation of vascular growth, a potential pathway toward creating bigger and better kidneys.
And while Little acknowledges that creating a fully-formed custom organ is the ultimate goal, the mini organs are an important bridge step.
"This is just another great example of the potential of pluripotent stem cells, and I am just passionate to see it do some good."
Therapies for Healthy Aging with Dr. Alexandra Bause
My guest today is Dr. Alexandra Bause, a biologist who has dedicated her career to advancing health, medicine and healthier human lifespans. Dr. Bause co-founded a company called Apollo Health Ventures in 2017. Currently a venture partner at Apollo, she's immersed in the discoveries underway in Apollo’s Venture Lab while the company focuses on assembling a team of investors to support progress. Dr. Bause and Apollo Health Ventures say that biotech is at “an inflection point” and is set to become a driver of important change and economic value.
Previously, Dr. Bause worked at the Boston Consulting Group in its healthcare practice specializing in biopharma strategy, among other priorities
She did her PhD studies at Harvard Medical School focusing on molecular mechanisms that contribute to cellular aging, and she’s also a trained pharmacist
In the episode, we talk about the present and future of therapeutics that could increase people’s spans of health, the benefits of certain lifestyle practice, the best use of electronic wearables for these purposes, and much more.
Dr. Bause is at the forefront of developing interventions that target the aging process with the aim of ensuring that all of us can have healthier, more productive lifespans.

This man spent over 70 years in an iron lung. What he was able to accomplish is amazing.
Paul Alexander spent more than 70 years confined to an iron lung after a polio infection left him paralyzed at age 6. Here, Alexander uses a mirror attached to the top of his iron lung to view his surroundings.
It’s a sight we don’t normally see these days: A man lying prone in a big, metal tube with his head sticking out of one end. But it wasn’t so long ago that this sight was unfortunately much more common.
In the first half of the 20th century, tens of thousands of people each year were infected by polio—a highly contagious virus that attacks nerves in the spinal cord and brainstem. Many people survived polio, but a small percentage of people who did were left permanently paralyzed from the virus, requiring support to help them breathe. This support, known as an “iron lung,” manually pulled oxygen in and out of a person’s lungs by changing the pressure inside the machine.
Paul Alexander was one of several thousand who were infected and paralyzed by polio in 1952. That year, a polio epidemic swept the United States, forcing businesses to close and polio wards in hospitals all over the country to fill up with sick children. When Paul caught polio in the summer of 1952, doctors urged his parents to let him rest and recover at home, since the hospital in his home suburb of Dallas, Texas was already overrun with polio patients.
Paul rested in bed for a few days with aching limbs and a fever. But his condition quickly got worse. Within a week, Paul could no longer speak or swallow, and his parents rushed him to the local hospital where the doctors performed an emergency procedure to help him breathe. Paul woke from the surgery three days later, and found himself unable to move and lying inside an iron lung in the polio ward, surrounded by rows of other paralyzed children.

But against all odds, Paul lived. And with help from a physical therapist, Paul was able to thrive—sometimes for small periods outside the iron lung.
The way Paul did this was to practice glossopharyngeal breathing (or as Paul called it, “frog breathing”), where he would trap air in his mouth and force it down his throat and into his lungs by flattening his tongue. This breathing technique, taught to him by his physical therapist, would allow Paul to leave the iron lung for increasing periods of time.
With help from his iron lung (and for small periods of time without it), Paul managed to live a full, happy, and sometimes record-breaking life. At 21, Paul became the first person in Dallas, Texas to graduate high school without attending class in person, owing his success to memorization rather than taking notes. After high school, Paul received a scholarship to Southern Methodist University and pursued his dream of becoming a trial lawyer and successfully represented clients in court.

Throughout his long life, Paul was also able to fly on a plane, visit the beach, adopt a dog, fall in love, and write a memoir using a plastic stick to tap out a draft on a keyboard. In recent years, Paul joined TikTok and became a viral sensation with more than 330,000 followers. In one of his first videos, Paul advocated for vaccination and warned against another polio epidemic.
Paul was reportedly hospitalized with COVID-19 at the end of February and died on March 11th, 2024. He currently holds the Guiness World Record for longest survival inside an iron lung—71 years.
Polio thankfully no longer circulates in the United States, or in most of the world, thanks to vaccines. But Paul continues to serve as a reminder of the importance of vaccination—and the power of the human spirit.
““I’ve got some big dreams. I’m not going to accept from anybody their limitations,” he said in a 2022 interview with CNN. “My life is incredible.”

