This breath test can detect liver disease earlier

A company in England has made a test that picks out the compounds from breath that reveal if people have liver disease.
Every year, around two million people worldwide die of liver disease. While some people inherit the disease, it’s most commonly caused by hepatitis, obesity and alcoholism. These underlying conditions kill liver cells, causing scar tissue to form until eventually the liver cannot function properly. Since 1979, deaths due to liver disease have increased by 400 percent.
The sooner the disease is detected, the more effective treatment can be. But once symptoms appear, the liver is already damaged. Around 50 percent of cases are diagnosed only after the disease has reached the final stages, when treatment is largely ineffective.
To address this problem, Owlstone Medical, a biotech company in England, has developed a breath test that can detect liver disease earlier than conventional approaches. Human breath contains volatile organic compounds (VOCs) that change in the first stages of liver disease. Owlstone’s breath test can reliably collect, store and detect VOCs, while picking out the specific compounds that reveal liver disease.
“There’s a need to screen more broadly for people with early-stage liver disease,” says Owlstone’s CEO Billy Boyle. “Equally important is having a test that's non-invasive, cost effective and can be deployed in a primary care setting.”
The standard tool for detection is a biopsy. It is invasive and expensive, making it impractical to use for people who aren't yet symptomatic. Meanwhile, blood tests are less invasive, but they can be inaccurate and can’t discriminate between different stages of the disease.
In the past, breath tests have not been widely used because of the difficulties of reliably collecting and storing breath. But Owlstone’s technology could help change that.
The team is testing patients in the early stages of advanced liver disease, or cirrhosis, to identify and detect these biomarkers. In an initial study, Owlstone’s breathalyzer was able to pick out patients who had early cirrhosis with 83 percent sensitivity.
Boyle’s work is personally motivated. His wife died of colorectal cancer after she was diagnosed with a progressed form of the disease. “That was a big impetus for me to see if this technology could work in early detection,” he says. “As a company, Owlstone is interested in early detection across a range of diseases because we think that's a way to save lives and a way to save costs.”
How it works
In the past, breath tests have not been widely used because of the difficulties of reliably collecting and storing breath. But Owlstone’s technology could help change that.
Study participants breathe into a mouthpiece attached to a breath sampler developed by Owlstone. It has cartridges are designed and optimized to collect gases. The sampler specifically targets VOCs, extracting them from atmospheric gases in breath, to ensure that even low levels of these compounds are captured.
The sampler can store compounds stably before they are assessed through a method called mass spectrometry, in which compounds are converted into charged atoms, before electromagnetic fields filter and identify even the tiniest amounts of charged atoms according to their weight and charge.
The top four compounds in our breath
In an initial study, Owlstone captured VOCs in breath to see which ones could help them tell the difference between people with and without liver disease. They tested the breath of 46 patients with liver disease - most of them in the earlier stages of cirrhosis - and 42 healthy people. Using this data, they were able to create a diagnostic model. Individually, compounds like 2-Pentanone and limonene performed well as markers for liver disease. Owlstone achieved even better performance by examining the levels of the top four compounds together, distinguishing between liver disease cases and controls with 95 percent accuracy.
“It was a good proof of principle since it looks like there are breath biomarkers that can discriminate between diseases,” Boyle says. “That was a bit of a stepping stone for us to say, taking those identified, let’s try and dose with specific concentrations of probes. It's part of building the evidence and steering the clinical trials to get to liver disease sensitivity.”
Sabine Szunerits, a professor of chemistry in Institute of Electronics at the University of Lille, sees the potential of Owlstone’s technology.
“Breath analysis is showing real promise as a clinical diagnostic tool,” says Szunerits, who has no ties with the company. “Owlstone Medical’s technology is extremely effective in collecting small volatile organic biomarkers in the breath. In combination with pattern recognition it can give an answer on liver disease severity. I see it as a very promising way to give patients novel chances to be cured.”
Improving the breath sampling process
Challenges remain. With more than one thousand VOCs found in the breath, it can be difficult to identify markers for liver disease that are consistent across many patients.
Julian Gardner is a professor of electrical engineering at Warwick University who researches electronic sensing devices. “Everyone’s breath has different levels of VOCs and different ones according to gender, diet, age etc,” Gardner says. “It is indeed very challenging to selectively detect the biomarkers in the breath for liver disease.”
So Owlstone is putting chemicals in the body that they know interact differently with patients with liver disease, and then using the breath sampler to measure these specific VOCs. The chemicals they administer are called Exogenous Volatile Organic Compound) probes, or EVOCs.
Most recently, they used limonene as an EVOC probe, testing 29 patients with early cirrhosis and 29 controls. They gave the limonene to subjects at specific doses to measure how its concentrations change in breath. The aim was to try and see what was happening in their livers.
“They are proposing to use drugs to enhance the signal as they are concerned about the sensitivity and selectivity of their method,” Gardner says. “The approach of EVOC probes is probably necessary as you can then eliminate the person-to-person variation that will be considerable in the soup of VOCs in our breath.”
Through these probes, Owlstone could identify patients with liver disease with 83 percent sensitivity. By targeting what they knew was a disease mechanism, they were able to amplify the signal. The company is starting a larger clinical trial, and the plan is to eventually use a panel of EVOC probes to make sure they can see diverging VOCs more clearly.
“I think the approach of using probes to amplify the VOC signal will ultimately increase the specificity of any VOC breath tests, and improve their practical usability,” says Roger Yazbek, who leads the South Australian Breath Analysis Research (SABAR) laboratory in Flinders University. “Whilst the findings are interesting, it still is only a small cohort of patients in one location.”
The future of breath diagnosis
Owlstone wants to partner with pharmaceutical companies looking to learn if their drugs have an effect on liver disease. They’ve also developed a microchip, a miniaturized version of mass spectrometry instruments, that can be used with the breathalyzer. It is less sensitive but will enable faster detection.
Boyle says the company's mission is for their tests to save 100,000 lives. "There are lots of risks and lots of challenges. I think there's an opportunity to really establish breath as a new diagnostic class.”
Podcast: The Friday Five Weekly Roundup in Health Research
In this week's Friday Five: progress on a pill to prevent lung cancer. Plus, ancient wisdom about Neti pots could pay off against Covid, a breakthrough for precision medicine and obesity, the connection between taking a multivitamin pill and brain health - and much more.
The Friday Five covers five stories in research that you may have missed this week. There are plenty of controversies and troubling ethical issues in science – and we get into many of them in our online magazine – but this news roundup focuses on scientific creativity and progress to give you a therapeutic dose of inspiration headed into the weekend.
Listen on Apple | Listen on Spotify | Listen on Stitcher | Listen on Amazon | Listen on Google
Here are the promising studies covered in this week's Friday Five:
- A pill to prevent lung cancer?
- Ancient wisdom about Neti pots could pay off for Covid
- Breakthrough for precision medicine and obesity
- How to refreeze the north and south poles
- The connection between taking multivitamin pills and brain health
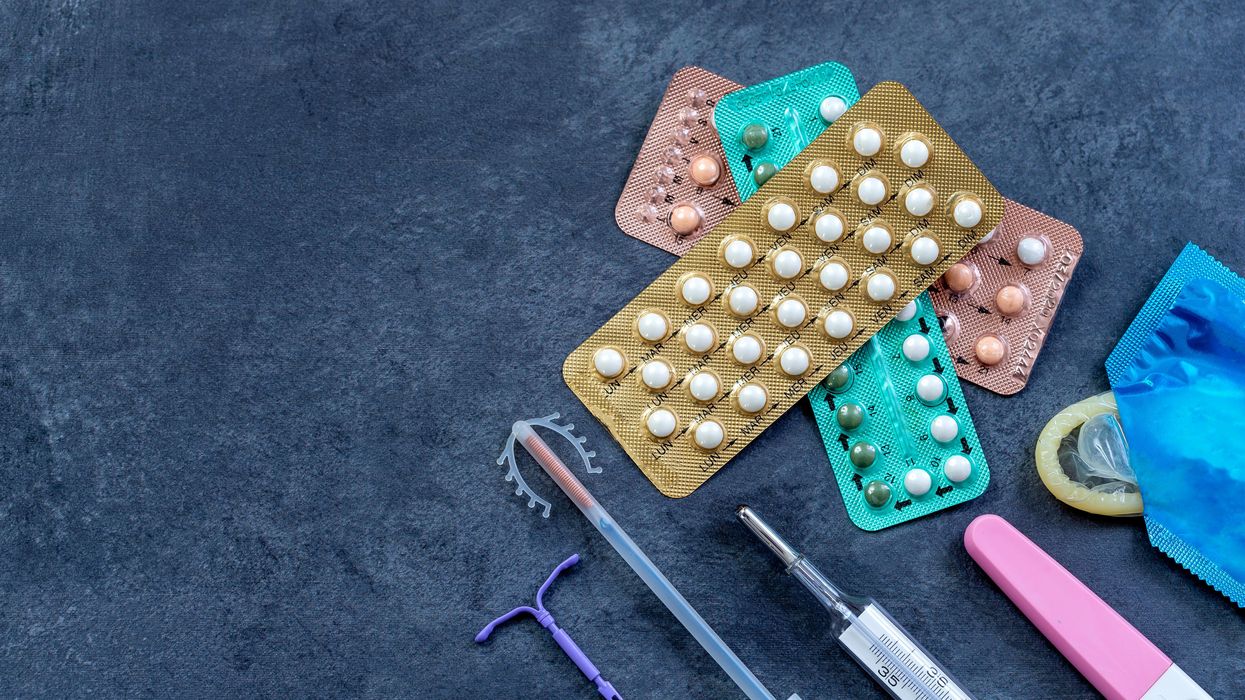
New Options Are Emerging in the Search for Better Birth Control
Photo by JPC-PROD on Adobe Stock
About decade ago, Elizabeth Summers' options for birth control suddenly narrowed. Doctors diagnosed her with Factor V Leiden, a rare genetic disorder, after discovering blood clots in her lungs. The condition increases the risk of clotting, so physicians told Summers to stay away from the pill and other hormone-laden contraceptives. "Modern medicine has generally failed to provide me with an effective and convenient option," she says.
But new birth control options are emerging for women like Summers. These alternatives promise to provide more choices to women who can't ingest hormones or don't want to suffer their unpleasant side effects.
These new products have their own pros and cons. Still, doctors are welcoming new contraceptives following a long drought in innovation. "It's been a long time since we've had something new in the world of contraception," says Heather Irobunda, an obstetrician and gynecologist at NYC Health and Hospitals.
On social media, Irobunda often fields questions about one of these new options, a lubricating gel called Phexxi. San Diego-based Evofem, the company behind Phexxi, has been advertising the product on Hulu and Instagram after the gel was approved by the Food and Drug Administration in May 2020. The company's trendy ads target women who feel like condoms diminish the mood, but who also don't want to mess with an IUD or hormones.
Here's how it works: Phexxi is inserted via a tampon-like device up to an hour before sex. The gel regulates vaginal pH — essentially, the acidity levels — in a range that's inhospitable to sperm. It sounds a lot like spermicide, which is also placed in the vagina prior to sex to prevent pregnancy. But spermicide can damage the vagina's cell walls, which can increase the risk of contracting sexually transmitted diseases.
"Not only is innovation needed, but women want a non-hormonal option."
Phexxi isn't without side effects either. The most common one is vaginal burning, according to a late-stage trial. It's also possible to develop a urinary tract infection while using the product. That same study found that during typical use, Phexxi is about 86 percent effective at preventing pregnancy. The efficacy rate is comparable to condoms but lower than birth control pills (91 percent) and significantly lower than an IUD (99 percent).
Phexxi – which comes in a pack of 12 – represents a tiny but growing part of the birth control market. Pharmacies dispensed more than 14,800 packs from April through June this year, a 65 percent increase over the previous quarter, according to data from Evofem.
"We've been able to demonstrate that not only is innovation needed, but women want a non-hormonal option," says Saundra Pelletier, Evofem's CEO.
Beyond contraception, the company is carrying out late-stage tests to gauge Phexxi's effectiveness at preventing the sexually transmitted infections chlamydia and gonorrhea.

Phexxi is inserted via a tampon-like device up to an hour before sex.
Phexxi
A New Pill
The first birth control pill arrived in 1960, combining the hormones estrogen and progestin to stop sperm from joining with an egg, giving women control over their fertility. Subsequent formulations sought to ease side effects, by way of lower amounts of estrogen. But some women still experience headaches and nausea – or more serious complications like blood clots. On social media, women noted that birth control pills are much more likely to cause blood clots than Johnson & Johnson's COVID-19 vaccine that was briefly paused to evaluate the risk of clots in women under age 50. What will it take, they wondered, for safer birth control?
Mithra Pharmaceuticals of Belgium sought to create a gentler pill. In April 2021, the FDA approved Mithra's Nextstellis, which includes a naturally occurring estrogen, the first new estrogen in the U.S. in 50 years. Nextstellis selectively acts on tissues lining the uterus, while other birth control pills have a broader target.
A Phase 3 trial showed a 98 percent efficacy rate. Andrew London, an obstetrician and gynecologist, who practices at several Maryland hospitals, says the results are in line with some other birth control pills. But, he added, early studies indicate that Nextstellis has a lower risk of blood clotting, along with other potential benefits, which additional clinical testing must confirm.
"It's not going to be worse than any other pill. We're hoping it's going to be significantly better," says London.
The estrogen in Nexstellis, called estetrol, was skipped over by the pharmaceutical industry after its discovery in the 1960s. Estetrol circulates between the mother and fetus during pregnancy. Decades later, researchers took a new look, after figuring out how to synthesize estetrol in a lab, as well as produce estetrol from plants.
"That allowed us to really start to investigate the properties and do all this stuff you have to do for any new drug," says Michele Gordon, vice president of marketing in women's health at Mayne Pharma, which licensed Nextstellis.
Bonnie Douglas, who followed the development of Nextstellis as part of a search for better birth control, recently switched to the product. "So far, it's much more tolerable," says Douglas. Previously, the Midwesterner was so desperate to find a contraceptive with fewer side effects that she turned to an online pharmacy to obtain a different birth control pill that had been approved in Canada but not in the U.S.
Contraceptive Access
Even if a contraceptive lands FDA approval, access poses a barrier. Getting insurers to cover new contraceptives can be difficult. For the uninsured, state and federal programs can help, and companies should keep prices in a reasonable range, while offering assistance programs. So says Kelly Blanchard, president of the nonprofit Ibis Reproductive Health. "For innovation to have impact, you want to reach as many folks as possible," she says.
In addition, companies developing new contraceptives have struggled to attract venture capital. That's changing, though.
In 2015, Sabrina Johnson founded DARÉ Bioscience around the idea of women's health. She estimated the company would be fully funded in six months, based on her track record in biotech and the demand for novel products.
But it's been difficult to get male investors interested in backing new contraceptives. It took Johnson two and a half years to raise the needed funds, via a reverse merger that took the company public. "There was so much education that was necessary," Johnson says, adding: "The landscape has changed considerably."
Johnson says she would like to think DARÉ had something to do with the shift, along with companies like Organon, a spinout of pharma company Merck that's focused on reproductive health. In surveying the fertility landscape, DARÉ saw limited non-hormonal options. On-demand options – like condoms – can detract from the moment. Copper IUDs must be inserted by a doctor and removed if a woman wants to return to fertility, and this method can have onerous side effects.
So, DARÉ created Ovaprene, a hormone-free device that's designed to be inserted into the vagina monthly by the user. The mesh product acts as a barrier, while releasing a chemical that immobilizes sperm. In an early study, the company reported that Ovaprene prevented almost all sperm from entering the cervical canal. The results, DARÉ believes, indicate high efficacy. Should Ovaprene eventually win regulatory approval, drug giant Bayer will handle commercializing the device.
Other new forms of birth control in development are further out, and that's assuming they perform well in clinical trials. Among them: a once-a-month birth control pill, along with a male version of the birth control pill. The latter is often brought up among women who say it's high time that men take a more proactive role in birth control.
For Summers, her search for a safe and convenient birth control continues. She tried Phexxi, which caused irritation. Still, she's excited that a non-hormonal option now exists. "I'm sure it will work for others," she says.
This article was first published by Leaps.org on August 31, 2021.