A vaccine for Lyme disease could be coming. But will patients accept it?

Hiking is one of Marci Flory's favorite activities, but her chronic Lyme disease sometimes renders her unable to walk. Biotech companies Pfizer and Valneva are testing a vaccine for Lyme disease in a Phase III clinical trial.
For more than two decades, Marci Flory, a 40-year-old emergency room nurse from Lawrence, Kan., has battled the recurring symptoms of chronic Lyme disease, an illness which she believes began after being bitten by a tick during her teenage years.
Over the years, Flory has been plagued by an array of mysterious ailments, ranging from fatigue to crippling pain in her eyes, joints and neck, and even postural tachycardia syndrome or PoTS, an abnormal increase in heart rate after sitting up or standing. Ten years ago, she began to experience the onset of neurological symptoms which ranged from brain fog to sudden headaches, and strange episodes of leg weakness which would leave her unable to walk.
“Initially doctors thought I had ALS, or less likely, multiple sclerosis,” she says. “But after repeated MRI scans for a year, they concluded I had a rare neurological condition called acute transverse myelitis.”
But Flory was not convinced. After ordering a variety of private blood tests, she discovered she was infected with a range of bacteria in the genus Borrelia that live in the guts of ticks, the infectious agents responsible for Lyme disease.
“It made sense,” she says. “Looking back, I was bitten in high school and misdiagnosed with mononucleosis. This was probably the start, and my immune system kept it under wraps for a while. The Lyme bacteria can burrow into every tissue in the body, go into cyst form and become dormant before reactivating.”
The reason why cases of Lyme disease are increasing is down to changing weather patterns, triggered by climate change, meaning that ticks are now found across a much wider geographic range than ever before.
When these species of bacteria are transmitted to humans, they can attack the nervous system, joints and even internal organs which can lead to serious health complications such as arthritis, meningitis and even heart failure. While Lyme disease can sometimes be successfully treated with antibiotics if spotted early on, not everyone responds to these drugs, and for patients who have developed chronic symptoms, there is no known cure. Flory says she knows of fellow Lyme disease patients who have spent hundreds of thousands of dollars seeking treatments.
Concerningly, statistics show that Lyme and other tick-borne diseases are on the rise. Recently released estimates based on health insurance records suggest that at least 476,000 Americans are diagnosed with Lyme disease every year, and many experts believe the true figure is far higher.
The reason why the numbers are growing is down to changing weather patterns, triggered by climate change, meaning that ticks are now found across a much wider geographic range than ever before. Health insurance data shows that cases of Lyme disease have increased fourfold in rural parts of the U.S. over the last 15 years, and 65 percent in urban regions.
As a result, many scientists who have studied Lyme disease feel that it is paramount to bring some form of protective vaccine to market which can be offered to people living in the most at-risk areas.
“Even the increased awareness for Lyme disease has not stopped the cases,” says Eva Sapi, professor of cellular and molecular biology at the University of New Haven. “Some of these patients are looking for answers for years, running from one doctor to another, so that is obviously a very big cost for our society at so many levels.”
Emerging vaccines – and backlash
But with the rising case numbers, interest has grown among the pharmaceutical industry and research communities. Vienna-based biotech Valneva have partnered with Pfizer to take their vaccine – a seasonal jab which offers protection against the six most common strains of Lyme disease in the northern hemisphere – into a Phase III clinical trial which began in August. Involving 6,000 participants in a number of U.S. states and northern Europe where Lyme disease is endemic, it could lead to a licensed vaccine by 2025, if it proves successful.
“For many years Lyme was considered a small market vaccine,” explains Monica E. Embers, assistant professor of parasitology at Tulane University in New Orleans. “Now we know that this is a much bigger problem, Pfizer has stepped up to invest in preventing this disease and other pharmaceutical companies may as well.”
Despite innovations, patient communities and their representatives remain ambivalent about the idea of a vaccine. Some of this skepticism dates back to the failed LYMErix vaccine which was developed in the late 1990s before being withdrawn from the market.
At the same time, scientists at Yale University are developing a messenger RNA vaccine which aims to train the immune system to respond to tick bites by exposing it to 19 proteins found in tick saliva. Whereas the Valneva vaccine targets the bacteria within ticks, the Yale vaccine attempts to provoke an instant and aggressive immune response at the site of the bite. This causes the tick to fall off and limits the potential for transmitting dangerous infections.
But despite these innovations, patient communities and their representatives remain ambivalent about the idea of a vaccine. Some of this skepticism dates back to the failed LYMErix vaccine which was developed in the late 1990s before being withdrawn from the market in 2002 after concerns were raised that it might induce autoimmune reactions in humans.
While this theory was ultimately disproved, the lingering stigma attached to LYMErix meant that most vaccine manufacturers chose to stay away from the disease for many years, something which Gregory Poland, head of the Mayo Clinic’s Vaccine Research Group in Minnesota, describes as a tragedy.
“Since 2002, we have not had a human Lyme vaccine in the U.S. despite the increasing number of cases,” says Poland. “Pretty much everyone in the field thinks they’re ten times higher than the official numbers, so you’re probably talking at least 400,000 each year. It’s an incredible burden but because of concerns about anti-vax protestors, until very recently, no manufacturer has wanted to touch this.”
Such was the backlash surrounding the failed LYMErix program that scientists have even explored the most creative of workarounds for protecting people in tick-populated regions, without needing to actually vaccinate them. One research program at the University of Tennessee came up with the idea of leaving food pellets containing a vaccine in woodland areas with the idea that rodents would eat the pellets, and the vaccine would then kill Borrelia bacteria within any ticks which subsequently fed on the animals.
Even the Pfizer-Valneva vaccine has been cautiously designed to try and allay any lingering concerns, two decades after LYMErix. “The concept is the same as the original LYMErix vaccine, but it has been made safer by removing regions that had the potential to induce autoimmunity,” says Embers. “There will always be individuals who oppose vaccines, Lyme or otherwise, but it will be a tremendous boost to public health to have the option.”
Vaccine alternatives
Researchers are also considering alternative immunization approaches in case sufficiently large numbers of people choose to reject any Lyme vaccine which gets approved. Researchers at UMass Chan Medical School have developed an artificially generated antibody, administered via an annual injection, which is capable of killing Borrelia bacteria in the guts of ticks before they can get into the human host.
So far animal studies have shown it to be 100 percent effective, while the scientists have completed a Phase I trial in which they tested it for safety on 48 volunteers in Nebraska. Because this approach provides the antibody directly, rather than triggering the human immune system to produce the antibody like a vaccine would, Embers predicts that it could be a viable alternative for the vaccine hesitant as well as providing an option for immunocompromised individuals who cannot produce enough of their own antibodies.
At the same time, many patient groups still raise concerns over the fact that numerous diagnostic tests for Lyme disease have been reported to have a poor accuracy. Without this, they argue that it is difficult to prove whether vaccines or any other form of immunization actually work. “If the disease is not understood enough to create a more accurate test and a universally accepted treatment protocol, particularly for those who weren’t treated promptly, how can we be sure about the efficacy of a vaccine?” says Natasha Metcalf, co-founder of the organization Lyme Disease UK.
Flory points out that there are so many different types of Borrelia bacteria which cause Lyme disease, that the immunizations being developed may only stop a proportion of cases. In addition, she says that chronic Lyme patients often report a whole myriad of co-infections which remain poorly understood and are likely to also be involved in the disease process.
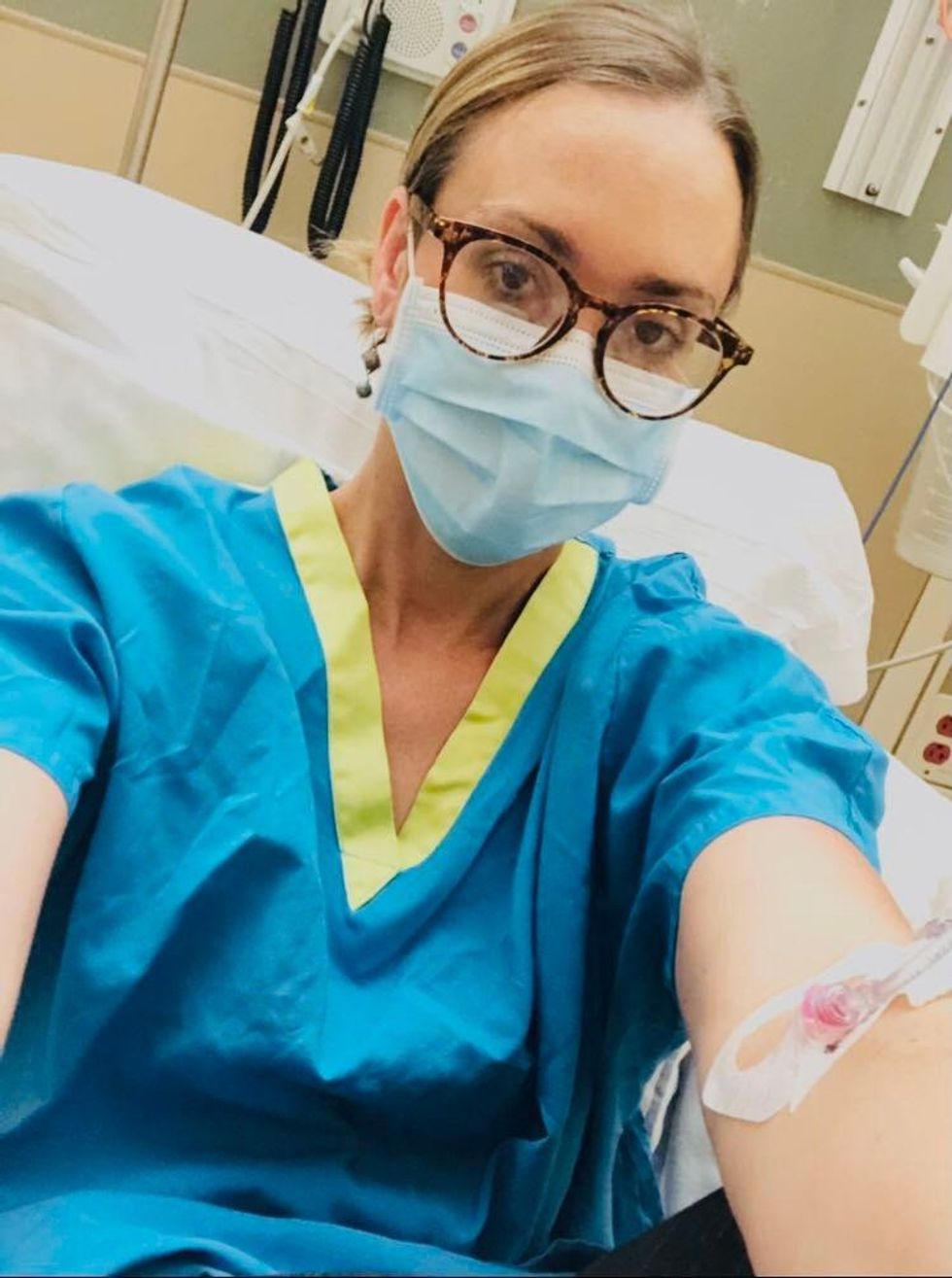
Marci Flory undergoes an infusion in an attempt to treat her Lyme disease symptoms.
Marci Flory
“I would love to see an effective Lyme vaccine but I have my reservations,” she says. “I am infected with four types of Borrelia bacteria, plus many co-infections – Babesia, Bartonella, Erlichiosis, Rickettsia, and Mycoplasma – all from a single Douglas County Kansas tick bite. Lyme never travels alone and the vaccine won’t protect against all the many strains of Borrelia and co-infections.”
Valneva CEO Thomas Lingelbach admits that the Pfizer-Valneva vaccine is not perfect, but predicts that it will still have significant impact if approved.
“We expect the vaccine to have 75 percent plus efficacy,” he says. “There is this legacy around the old Lyme vaccines, but the world is very, very different today. The number of clinical manifestations known to be caused by infection with Lyme Borreliosis has significantly increased, and the understanding around severity has certainly increased.”
Embers agrees that while it will still be important for doctors to monitor for other tick-borne infections which are not necessarily covered by the vaccine, having any clinically approved jab would still represent a major step forward in the fight against the disease.
“I think that any vaccine must be properly vetted, and these companies are performing extensive clinical trials to do just that,” she says. “Lyme is the most common tick-borne disease in the U.S. so the public health impact could be significant. However, clinicians and the general public must remain aware of all of the other tick-borne diseases such as Babesia and Anaplasma, and continue to screen for those when a tick bite is suspected.”
Dr. Barry Marshall (along with a collaborator) discovered that the bacteria H. Pylori, pictured, caused stomach ulcers.
[Editor's Note: Welcome to Leaps of the Past, a new monthly column that spotlights the fascinating backstory behind a medical or scientific breakthrough from history.]
------
Until about 40 years ago, ulcers were a mysterious – and sometimes deadly – ailment. Found in a person's stomach lining or intestine, ulcers are small sores that cause a variety of painful symptoms, such as vomiting, a burning or aching sensation, internal bleeding and stomach obstruction. Patients with ulcers suffered for years without a cure and sometimes even needed their stomachs completely removed to rid them from pain.
"To gastroenterologists, the concept of a germ causing ulcers was like saying the Earth is flat."
In the early 1980s, the majority of scientists thought that ulcers were caused by stress or poor diet. But a handful of scientists had a different theory: They believed that ulcers were caused by a corkscrew-shaped bacterium called Helicobacter pylori, or H. pylori for short. Robin Warren, a pathologist, and Barry Marshall, an internist, were the two pioneers of this theory, and the two teamed up to study H. pylori at the Royal Perth Hospital in 1981.
The pair started off by trying to culture the bacteria in the stomachs of patients with gastritis, an inflammation of the stomach lining and a precursor to developing an ulcer. Initially, the microbiologists involved in their clinical trial found no trace of the bacteria from patient samples – but after a few weeks, the microbiologists discovered that their lab techs had been throwing away the cultures before H. pylori could grow. "After that, we let the cultures grow longer and found 13 patients with duodenal ulcer," said Marshall in a later interview. "All of them had the bacteria."
Marshall and Warren also cultured H. pylori in the stomachs of patients with stomach cancer. They observed that "everybody with stomach cancer developed it on a background of gastritis. Whenever we found a person without Helicobacter, we couldn't find gastritis either." Marshall and Warren were convinced that H. pylori not only caused gastritis and peptic ulcers, but stomach cancer as well.
But when the team presented their findings at an annual meeting of the Royal Australasian College of Physicians in Perth, they were mostly met with skepticism. "To gastroenterologists, the concept of a germ causing ulcers was like saying the Earth is flat," Marshall said. "The idea was too weird."
Warren started treating his gastritis patients with antibiotics with great success – but other internists remained doubtful, continuing to treat their patients with antacids instead. Making matters more complicated, neither Warren nor Marshall could readily test their theory, since the pair only had lab mice at their disposal and H. pylori infects only humans and non-human primates, such as rhesus monkeys.
So Marshall took an unconventional approach. First, he underwent two tests to get a baseline reading of his stomach, which showed no presence of H. pylori. Then, Marshall took some H. pylori bacteria from a petri dish, mixed it with beef extract to create a broth, and gulped it down. If his theory was correct, a second gastric biopsy would show that his stomach was overrun with H. pylori bacteria, and a second endoscopy would show a painfully inflamed stomach – gastritis.
Less than a week later, Marshall started feeling sick. "I expected to develop an asymptomatic infection," he later said in an interview published in the Canadian Journal of Gastroenterology. "… [but] after five days, I started to have bloating and fullness after the evening meal, and my appetite decreased. My breath was bad and I vomited clear watery liquid, without acid, each morning."
At his wife's urging, Marshall started on a regimen of antibiotics to kill off the burgeoning bacteria, so a follow-up biopsy showed no signs of H. pylori. A follow-up endoscopy, however, showed "severe active gastritis" along with epithelial damage. This was the smoking gun other clinicians needed to believe that H. pylori caused gastritis and stomach cancer. When they began to treat their gastritis patients with antibiotics, the rate of peptic ulcers in the Australian population diminished by 70 percent.
Today, antibiotics are the standard of care for anyone afflicted with gastritis.
In 2005, Marshall and Warren were awarded the Nobel Prize in Physiology or Medicine for their discovery of H. Pylori and its role in developing gastritis and peptic ulcers. "Thanks to the pioneering discovery by Marshall and Warren, peptic ulcer disease is no longer a chronic, frequently disabling condition, but a disease that can be cured by a short regimen of antibiotics and acid secretion inhibitors," the Nobel Prize Committee said.
Today, antibiotics are the standard of care for anyone afflicted with gastritis – and stomach cancer has been significantly reduced in the Western world.
Would a Broad-Spectrum Antiviral Drug Stop the Pandemic?
An illustration of the novel coronavirus infecting human tissue.
The refocusing of medical research to COVID-19 is unprecedented in human history. Seven months ago, we barely were aware that the virus existed, and now a torrent of new information greets us each day online.
There are many unanswered questions about COVID-19, but perhaps the most fascinating is whether we even need to directly go after the virus itself.
Clinicaltrials.gov, the most commonly used registry for worldwide medical research, listed 1358 clinical trials on the disease, including using scores of different potential drugs and multiple combinations, when I first wrote this sentence. The following day that number of trials had increased to 1409. Laboratory work to prepare for trials presents an even broader and untabulated scope of activity.
Most trials will fail or not be as good as what has been discovered in the interim, but the hope is that a handful of them will yield vaccines for prevention and treatments to attenuate and ultimately cure the deadly infection.
The first impulse is to grab whatever drugs are on the shelf and see if any work against the new foe. We know their safety profiles and they have passed some regulatory hurdles. Remdesivir is the first to register some success against SARS-CoV-2, the virus behind the disease. The FDA has granted it expedited-use status, pending presentation of data that may lead to full approval of the drug.
Most observers see it as a treatment that might help, but not one that by itself is likely to break the back of the pandemic. Part of that is because it is delivered though IV infusion, which requires hospitalization, and as with most antiviral drugs, appears to be most beneficial when started early in disease. "The most effective products are going to be that ones that are developed by actually understanding more about this coronavirus," says Margaret "Peggy" Hamburg, who once led the New York City public health department and later the U.S. Food and Drug Administration.
Combination therapy that uses different drugs to hit a virus at different places in its life cycle have proven to work best in treating HIV and hepatitis C, and likely will be needed with this virus as well. Most viruses are simply too facile at evolving resistance to a single drug, and so require multiple hits to keep them down.
Laboratory work suggests that other drugs, both off-the-shelf and in development, particularly those to treat HIV and hepatitis, might also be of some benefit against SARS-CoV-2. But the number of possible drug combinations is mind-bogglingly large and the capacity to test them all right now is limited.
Broad-Spectrum Antivirals
Viruses are simple quasi-life forms. Effective treatments are more likely to be specific to a given virus, or at best its close relatives. That is unlike bacteria, where broad-spectrum antibiotics often can be used against common elements like the bacterial cell wall, or can disrupt quorum sensing signals that bacteria use to function as biofilms.
More than a decade ago, virologist Benhur Lee's lab at UCLA (now at Mt. Sinai in New York City) stumbled upon a broad-spectrum antiviral approach that seemed to work against all enveloped viruses they tested. The list ranged from the common flu to HIV to Ebola.
Other researchers grabbed this lead to develop a compound that worked quite well in cell cultures, but when they tried it in animals, a frustrating snag emerged; the compound needed to be activated by light. As the greatest medical need is to counter viruses deep inside the body, the research was put on the shelf. So Lee was surprised to learn recently that a company has inquired about rights to develop the compound not as a treatment but as a possible disinfectant. The tale illustrates both the unanticipated difficulties of drug development and that one never knows how knowledge ultimately might be put to use.
Remdesivir is a failed drug for Ebola that has found new life with SARS-CoV-2. It targets polymerase, an enzyme that the virus produces to use host cell machinery to replicate itself, and since the genetic sequence of polymerase is very similar among all of the different coronaviruses, scientists hope that the drug might be useful against known members of the family and others that might emerge in the future.
But nature isn't always that simple. Viral RNA is not a two-dimensional assemblage of genes in a flat line on a table; rather it is a three-dimensional matrix of twists and turns where a single atom change within the polymerase gene or another gene close by might change the orientation of the RNA or a molecular arm within it and block a drug from accessing the targeted binding site on the virus. One drug might need to bind to a large flat surface, while another might be able to slip a dagger-like molecular arm through a space in the matrix to reach its binding target.
That is why a broad-spectrum antiviral is so hard to develop, and why researchers continue to work on a wide variety of compounds that target polymerase as a binding site.
Additionally, it has taken us decades to begin to recognize the unintended consequences of broad-spectrum rather than narrowly targeted antibiotics on the gut microbiome and our overall health. Will a similar issue potentially arise in using a broad-spectrum antiviral?
"Off-target side effects are always of concern with drugs, and antivirals are no exception," says Yale University microbiologist Ben Chen. He believes that "most" bacteriophages, the viruses that infect bacteria and likely help to maintain stability in the gut microbial ecosystem, will shrug off such a drug. However, a few families of phages share polymerases that are similar to those found in coronaviruses. While the immediate need for treatment is great, we will have to keep a sharp eye out for unanticipated activity in the body's ecosystem from new drugs.
Is an Antiviral Needed?
There are many unanswered questions about COVID-19, but perhaps the most fascinating is whether we even need to directly go after the virus itself. Mounting evidence indicates that up to half the people who contract the infection don't seem to experience significant symptoms and their immune system seems to clear the virus.
The most severe cases of COVID-19 appear to result from an overactive immune response that damages surrounding tissue. Perhaps downregulating that response will be sufficient to reduce the disease burden. Several studies are underway using approved antibodies that modulate an overly active immune response.
One of the most surprising findings to date involves the monoclonal antibody leronlimab. It was originally developed to treat HIV infection and works modestly well there, but other drugs are better and its future likely will be mainly to treat patients who have developed resistance to those other drugs.
The response has been amazingly different in patients in the U.S. with COVID-19 who were given emergency access to leronlimab – two injections a week apart, though the company believes that four might be better. The immune response and inflammatory cytokines declined significantly, T cell counts were maintained, and surprisingly the amount of virus in the blood declined too. Data from the first ten patients is available in a preprint while the paper undergoes peer review for publication. Data from an additional fifty patients will be added.
"We got lucky and hit the bulls' eye from a mile away," says Jay Lalezari, the chief science officer of Cytodyn, the company behind leronlimab. Dr. Jay, as he is widely known in San Francisco, built an adoring fan base running many of the early-phase drug studies for treating HIV. While touting leronlimab, Lalezari suspects it might best be used as part of a combination therapy.
The small, under-capitalized firm is struggling for attention in the vast pool of therapies proposed to treat COVID-19. It faces the added challenge of gaining acceptance because it is based on a different approach and mechanism of action, which involves a signaling molecule important to immune cell migration, than what most researchers and the FDA anticipate as being relevant to counter SARS-CoV-2.
Common Issues
All of the therapeutics under development will face some common sets of issues. One is the pressure to have results yesterday, because people are dying. The rush to disseminate information "make me worry that certain things will become entrenched as truth, even in the scientific community, without the actual scientific documentation that ordinarily scientists would demand," says Hamburg.
"It is becoming increasingly clear that the biggest problem for drug and vaccine makers is not which therapeutics or vaccine platform to pursue."
Lack of standardization in assays and laboratory operations makes it difficult to compare results between labs studying SARS-CoV-2. In the long run, this will slow down the iterative process of research that builds upon what has gone before. And the shut down of supply chains, from chemicals to cell lines to animals to air shipment, has the potential to further hobble research.
Almost all researchers consult with the FDA in putting together their clinical trials. But the agency is overwhelmed with the surge of activity in the field, and is even less capable of handling novel approaches that fall outside of its standard guidance.
"It is becoming increasingly clear that the biggest problem for drug and vaccine makers is not which therapeutics or vaccine platform to pursue. It is that conventional clinical development paths are far too lengthy and cumbersome to address the current public health threat," John Hodgson wrote in Nature Biotechnology.
Another complicating factor with this virus is the broad range of organ and tissue types it can infect. That has implications for potential therapies, which often vary in their ability to enter different tissues. At a minimum, it complicates the drug development process.
Remdesivir has become the de facto standard of care. Ideally, clinical trials are conducted using the existing standard of care rather than a placebo as the control group. But shortages of the drug make that difficult and further inhibit learning what is the best treatment regimen for regular clinical care.
"Understandably, we all really want to respond to COVID-19 in a much, much more accelerated fashion," says Hamburg. But ultimately that depends upon "the reality of understanding the nature of the disease. And that is going to take a bit more time than we might like or wish."
[This article was originally published on June 8th, 2020 as part of a standalone magazine called GOOD10: The Pandemic Issue. Produced as a partnership among LeapsMag, The Aspen Institute, and GOOD, the magazine is available for free online.]