The Inside Story of Two Young Scientists Who Helped Make Moderna's Covid Vaccine Possible

Scientists Jason Schrum and Kerry Benenato solved crucial challenges in mRNA vaccine development.
In early 2020, Moderna Inc. was a barely-known biotechnology company with an unproven approach. It wanted to produce messenger RNA molecules to carry instructions into the body, teaching it to ward off disease. Experts doubted the Boston-based company would meet success.
Today, Moderna is a pharmaceutical power thanks to its success developing an effective Covid-19 vaccine. The company is worth $124 billion, more than giants including GlaxoSmithKline and Sanofi, and evidence has emerged that Moderna's shots are more protective than those produced by Pfizer-BioNTech and other vaccine makers. Pressure is building on the company to deliver more of its doses to people around the world, especially in poorer countries, and Moderna is working on vaccines against other pathogens, including Zika, influenza and cytomegalovirus.
But Moderna encountered such difficulties over the course of its eleven-year history that some executives worried it wouldn't survive. Two unlikely scientists helped save the company. Their breakthroughs paved the way for Moderna's Covid-19 shots but their work has never been publicized nor have their contributions been properly appreciated.
Derrick Rossi, a scientist at MIT, and Noubar Afeyan, a Cambridge-based investor, launched Moderna in September 2010. Their idea was to create mRNA molecules capable of delivering instructions to the body's cells, directing them to make proteins to heal ailments and cure disease. Need a statin, immunosuppressive, or other drug or vaccine? Just use mRNA to send a message to the body's cells to produce it. Rossi and Afeyan were convinced injecting mRNA into the body could turn it into its own laboratory, generating specific medications or vaccines as needed.
At the time, the notion that one might be able to teach the body to make proteins bordered on heresy. Everyone knew mRNA was unstable and set off the body's immune system on its way into cells. But in the late 2000's, two scientists at the University of Pennsylvania, Katalin Karikó and Drew Weissman, had figured out how to modify mRNA's chemical building blocks so the molecule could escape the notice of the immune system and enter the cell. Rossi and Afeyan couldn't convince the University of Pennsylvania to license Karikó and Weissman's patent, however, stymying Moderna's early ambitions. At the same time, the Penn scientists' technique seemed more applicable to an academic lab than a biotech company that needed to produce drugs or shots consistently and in bulk. Rossi and Afeyan's new company needed their own solution to help mRNA evade the body's defenses.
Some of Moderna's founders doubted Schrum could find success and they worried if their venture was doomed from the start.
The Scientist Who Modified mRNA: Jason Schrum
In 2010, Afeyan's firm subleased laboratory space in the basement of another Cambridge biotech company to begin scientific work. Afeyan chose a young scientist on his staff, Jason Schrum, to be Moderna's first employee, charging him with getting mRNA into cells without relying on Karikó and Weissman's solutions.
Schrum seemed well suited for the task. Months earlier, he had received a PhD in biological chemistry at Harvard University, where he had focused on nucleotide chemistry. Schrum even had the look of someone who might do big things. The baby-faced twenty-eight-year-old favored a relaxed, start-up look: khakis, button-downs, and Converse All-Stars.
Schrum felt immediate strain, however. He hadn't told anyone, but he was dealing with intense pain in his hands and joints, a condition that later would be diagnosed as degenerative arthritis. Soon Schrum couldn't bend two fingers on his left hand, making lab work difficult. He joined a drug trial, but the medicine proved useless. Schrum tried corticosteroid injections and anti-inflammatory drugs, but his left hand ached, restricting his experiments.
"It just wasn't useful," Schrum says, referring to his tender hand.*
He persisted, nonetheless. Each day in the fall of 2010, Schrum walked through double air-locked doors into a sterile "clean room" before entering a basement laboratory, in the bowels of an office in Cambridge's Kendall Square neighborhood, where he worked deep into the night. Schrum searched for potential modifications of mRNA nucleosides, hoping they might enable the molecule to produce proteins. Like all such rooms, there were no windows, so Schrum had to check a clock to know if it was day or night. A colleague came to visit once in a while, but most of the time, Schrum was alone.
Some of Moderna's founders doubted Schrum could find success and they worried if their venture was doomed from the start. An established MIT scientist turned down a job with the start-up to join pharmaceutical giant Novartis, dubious of Moderna's approach. Colleagues wondered if mRNA could produce proteins, at least on a consistent basis.
As Schrum began testing the modifications in January 2011, he made an unexpected discovery. Karikó and Weissman saw that by turned one of the building blocks for mRNA, a ribonucleoside called uridine, into a slightly different form called pseudouridine, the cell's immune system ignored the mRNA and the molecule avoided an immune response. After a series of experiments in the basement lab, Schrum discovered that a variant of pseudouridine called N1- methyl-pseudouridine did an even better job reducing the cell's innate immune response. Schrum's nucleoside switch enabled even higher protein production than Karikó and Weissman had generated, and Schrum's mRNAs lasted longer than either unmodified molecules or the modified mRNA the Penn academics had used, startling the young researcher. Working alone in a dreary basement and through intense pain, he had actually improved on the Penn professors' work.
Years later, Karikó and Weissman who would win acclaim. In September 2021, the scientists were awarded the Lasker-DeBakey Clinical Medical Research Award. Some predict they eventually will win a Nobel prize. But it would be Schrum's innovation that would form the backbone of both Moderna and Pfizer-BioNTech's Covid-19 vaccine, not the chemical modifications that Karikó and Weissman developed. For Schrum, necessity had truly been the mother of invention.
The Scientist Who Solved Delivery: Kerry Benenato
For several years, Moderna would make slow progress developing drugs to treat various diseases. Eventually, the company decided that mRNA was likely better suited for vaccines. By 2017, Moderna and the National Institutes of Health were discussing working together to develop mRNA–based vaccines, a partnership that buoyed Moderna's executives. There remained a huge obstacle in Moderna's way, however. It was up to Kerry Benenato to find a solution.
Benenato received an early hint of the hurdle in front of her three years earlier, when the organic chemist was first hired. When a colleague gave her a company tour, she was introduced to Moderna's chief scientific officer, Joseph Bolen, who seemed unusually excited to meet her.
"Oh, great!" Bolen said with a smile. "She's the one who's gonna solve delivery."
Bolen gave a hearty laugh and walked away, but Benenato detected seriousness in his quip.
Solve delivery?
It was a lot to expect from a 37-year-old scientist already dealing withinsecurities and self-doubt. Benenato was an accomplished researcher who most recently had worked at AstraZeneca after completing post-doctoral studies at Harvard University. Despite her impressive credentials, Benenato battled a lack of confidence that sometimes got in her way. Performance reviews from past employers had been positive, but they usually produced similar critiques: Be more vocal. Do a better job advocating for your ideas. Give us more, Kerry.
Benenato was petite and soft-spoken. She sometimes stuttered or relied on "ums" and "ahs" when she became nervous, especially in front of groups, part of why she sometimes didn't feel comfortable speaking up.
"I'm an introvert," she says. "Self-confidence is something that's always been an issue."
To Benenato, Moderna's vaccine approach seemed promising—the team was packaging mRNAs in microscopic fatty-acid compounds called lipid nanoparticles, or LNPs, that protected the molecules on their way into cells. Moderna's shots should have been producing ample and long-lasting proteins. But the company's scientists were alarmed—they were injecting shots deep into the muscle of mice, but their immune systems were mounting spirited responses to the foreign components of the LNPs, which had been developed by a Canadian company.
This toxicity was a huge issue: A vaccine or drug that caused sharp pain and awful fevers wasn't going to prove very popular. The Moderna team was in a bind: Its mRNA had to be wrapped in the fatty nanoparticles to have a chance at producing plentiful proteins, but the body wasn't tolerating the microscopic encasements, especially upon repeated dosing.
The company's scientists had done everything they could to try to make the molecule's swathing material disappear soon after entering the cells, in order to avoid the unfortunate side effects, such as chills and headaches, but they weren't making headway. Frustration mounted. Somehow, the researchers had to find a way to get the encasements—made of little balls of fat, cholesterol, and other substances—to deliver their payload mRNA and then quickly vanish, like a parent dropping a teenager off at a party, to avoid setting off the immune system in unpleasant ways, even as the RNA and the proteins the molecule created stuck around.
Benenato wasn't entirely shocked by the challenges Moderna was facing. One of the reasons she had joined the upstart company was to help develop its delivery technology. She just didn't realize how pressing the issue was, or how stymied the researchers had become. Benenato also didn't know that Moderna board members were among those most discouraged by the delivery issue. In meetings, some of them pointed out that pharmaceutical giants like Roche Holding and Novartis had worked on similar issues and hadn't managed to develop lipid nanoparticles that were both effective and well tolerated by the body. Why would Moderna have any more luck?
Stephen Hoge insisted the company could yet find a solution.
"There's no way the only innovations in LNP are going to come from some academics and a small Canadian company," insisted Hoge, who had convinced the executives that hiring Benenato might help deliver an answer.
Benenato realized that while Moderna might have been a hot Boston-area start- up, it wasn't set up to do the chemistry necessary to solve their LNP problem. Much of its equipment was old or secondhand, and it was the kind used to tinker with mRNAs, not lipids.
"It was scary," she says.
When Benenato saw the company had a nuclear magnetic resonance spectrometer, which allows chemists to see the molecular structure of material, she let out a sigh of relief. Then Benenato inspected the machine and realized it was a jalopy. The hulking, aging instrument had been decommissioned and left behind by a previous tenant, too old and banged up to bring with them.
Benenato began experimenting with different chemical changes for Moderna's LNPs, but without a working spectrometer she and her colleagues had to have samples ready by noon each day, so they could be picked up by an outside company that would perform the necessary analysis. After a few weeks, her superiors received an enormous bill for the outsourced work and decided to pay to get the old spectrometer running again.
After months of futility, Benenato became impatient. An overachiever who could be hard on herself, she was eager to impress her new bosses. Benenato felt pressure outside the office, as well. She was married with a preschool-age daughter and an eighteen-month-old son. In her last job, Benenato's commute had been a twenty-minute trip to Astra-Zeneca's office in Waltham, outside Boston; now she was traveling an hour to Moderna's Cambridge offices. She became anxious—how was she going to devote the long hours she realized were necessary to solve their LNP quandary while providing her children proper care? Joining Moderna was beginning to feel like a possible mistake.
She turned to her husband and father for help. They reminded her of the hard work she had devoted to establishing her career and said it would be a shame if she couldn't take on the new challenge. Benenato's husband said he was happy to stay home with the kids, alleviating some of her concerns.
Back in the office, she got to work. She wanted to make lipids that were easier for the body to chop into smaller pieces, so they could be eliminated by the body's enzymes. Until then, Moderna, like most others, relied on all kinds of complicated chemicals to hold its LNP packaging together. They weren't natural, though, so the body was having a hard time breaking them down, causing the toxicity.
Benenato began experimenting with simpler chemicals. She inserted "ester bonds"—compounds referred to in chemical circles as "handles" because the body easily grabs them and breaks them apart. Ester bonds had two things going for them: They were strong enough to help ensure the LNP remained stable, acting much like a drop of oil in water, but they also gave the body's enzymes something to target and break down as soon as the LNP entered the cell, a way to quickly rid the body of the potentially toxic LNP components. Benenato thought the inclusion of these chemicals might speed the elimination of the LNP delivery material.
This idea, Benenato realized, was nothing more than traditional, medicinal chemistry. Most people didn't use ester bonds because they were pretty unsophisticated. But, hey, the tricky stuff wasn't working, so Benenato thought she'd see if the simple stuff worked.
Benenato also wanted to try to replace a group of unnatural chemicals in the LNP that was contributing to the spirited and unwelcome response from the immune system. Benenato set out to build a new and improved chemical combination. She began with ethanolamine, a colorless, natural chemical, an obvious start for any chemist hoping to build a more complex chemical combination. No one relied on ethanolamine on its own.
Benenato was curious, though. What would happen if she used just these two simple modifications to the LNP: ethanolamine with the ester bonds? Right away, Benenato noticed her new, super-simple compound helped mRNA create some protein in animals. It wasn't much, but it was a surprising and positive sign. Benenato spent over a year refining her solution, testing more than one hundred variations, all using ethanolamine and ester bonds, showing improvements with each new version of LNP. After finishing her 102nd version of the lipid molecule, which she named SM102, Benenato was confident enough in her work to show it to Hoge and others.
They immediately got excited. The team kept tweaking the composition of the lipid encasement. In 2017, they wrapped it around mRNA molecules and injected the new combination in mice and then monkeys. They saw plentiful, potent proteins were being produced and the lipids were quickly being eliminated, just as Benenato and her colleagues had hoped. Moderna had its special sauce.
That year, Benenato was asked to deliver a presentation to Stephane Bancel, Moderna's chief executive, Afeyan, and Moderna's executive committee to explain why it made sense to use the new, simpler LNP formulation for all its mRNA vaccines. She still needed approval from the executives to make the change. Ahead of the meeting, she was apprehensive, as some of her earlier anxieties returned. But an unusual calm came over her as she began speaking to the group. Benenato explained how experimenting with basic, overlooked chemicals had led to her discovery.
She said she had merely stumbled onto the company's solution, though her bosses understood the efforts that had been necessary for the breakthrough. The board complimented her work and agreed with the idea of switching to the new LNP. Benenato beamed with pride.
"As a scientist, serendipity has been my best friend," she told the executives.
Over the next few years, Benenato and her colleagues would improve on their methods and develop even more tolerable and potent LNP encasement for mRNA molecules. Their work enabled Moderna to include higher doses of vaccine in its shots. In early 2020, Moderna developed Covid-19 shots that included 100 micrograms of vaccine, compared with 30 micrograms in the Pfizer-BioNTech vaccine. That difference appears to help the Moderna vaccine generate higher titers and provide more protection.
"You set out in a career in drug discovery to want to make a difference," Benenato says. "Seeing it come to reality has been surreal and emotional."
Editor's Note: This essay is excerpted from A SHOT TO SAVE THE WORLD: The Inside Story of the Life-or-Death Race for a COVID-19 Vaccine by Gregory Zuckerman, now on sale from Portfolio/Penguin.
*Jason Schrum's arthritis is now in complete remission, thanks to Humira (adalimumab), a TNF-alpha blocker.

Thanks to safety cautions from the COVID-19 pandemic, a strain of influenza has been completely eliminated.
If you were one of the millions who masked up, washed your hands thoroughly and socially distanced, pat yourself on the back—you may have helped change the course of human history.
Scientists say that thanks to these safety precautions, which were introduced in early 2020 as a way to stop transmission of the novel COVID-19 virus, a strain of influenza has been completely eliminated. This marks the first time in human history that a virus has been wiped out through non-pharmaceutical interventions, such as vaccines.
The flu shot, explained
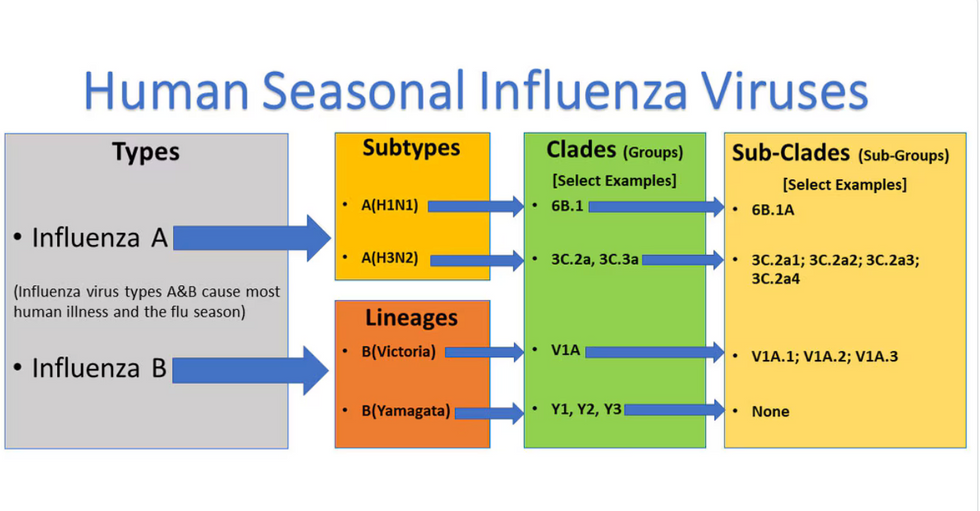
Influenza viruses type A and B are responsible for the majority of human illnesses and the flu season.
Centers for Disease Control
For more than a decade, flu shots have protected against two types of the influenza virus–type A and type B. While there are four different strains of influenza in existence (A, B, C, and D), only strains A, B, and C are capable of infecting humans, and only A and B cause pandemics. In other words, if you catch the flu during flu season, you’re most likely sick with flu type A or B.
Flu vaccines contain inactivated—or dead—influenza virus. These inactivated viruses can’t cause sickness in humans, but when administered as part of a vaccine, they teach a person’s immune system to recognize and kill those viruses when they’re encountered in the wild.
Each spring, a panel of experts gives a recommendation to the US Food and Drug Administration on which strains of each flu type to include in that year’s flu vaccine, depending on what surveillance data says is circulating and what they believe is likely to cause the most illness during the upcoming flu season. For the past decade, Americans have had access to vaccines that provide protection against two strains of influenza A and two lineages of influenza B, known as the Victoria lineage and the Yamagata lineage. But this year, the seasonal flu shot won’t include the Yamagata strain, because the Yamagata strain is no longer circulating among humans.
How Yamagata Disappeared

Flu surveillance data from the Global Initiative on Sharing All Influenza Data (GISAID) shows that the Yamagata lineage of flu type B has not been sequenced since April 2020.
Nature
Experts believe that the Yamagata lineage had already been in decline before the pandemic hit, likely because the strain was naturally less capable of infecting large numbers of people compared to the other strains. When the COVID-19 pandemic hit, the resulting safety precautions such as social distancing, isolating, hand-washing, and masking were enough to drive the virus into extinction completely.
Because the strain hasn’t been circulating since 2020, the FDA elected to remove the Yamagata strain from the seasonal flu vaccine. This will mark the first time since 2012 that the annual flu shot will be trivalent (three-component) rather than quadrivalent (four-component).
Should I still get the flu shot?
The flu shot will protect against fewer strains this year—but that doesn’t mean we should skip it. Influenza places a substantial health burden on the United States every year, responsible for hundreds of thousands of hospitalizations and tens of thousands of deaths. The flu shot has been shown to prevent millions of illnesses each year (more than six million during the 2022-2023 season). And while it’s still possible to catch the flu after getting the flu shot, studies show that people are far less likely to be hospitalized or die when they’re vaccinated.
Another unexpected benefit of dropping the Yamagata strain from the seasonal vaccine? This will possibly make production of the flu vaccine faster, and enable manufacturers to make more vaccines, helping countries who have a flu vaccine shortage and potentially saving millions more lives.
After his grandmother’s dementia diagnosis, one man invented a snack to keep her healthy and hydrated.
Founder Lewis Hornby and his grandmother Pat, sampling Jelly Drops—an edible gummy containing water and life-saving electrolytes.
On a visit to his grandmother’s nursing home in 2016, college student Lewis Hornby made a shocking discovery: Dehydration is a common (and dangerous) problem among seniors—especially those that are diagnosed with dementia.
Hornby’s grandmother, Pat, had always had difficulty keeping up her water intake as she got older, a common issue with seniors. As we age, our body composition changes, and we naturally hold less water than younger adults or children, so it’s easier to become dehydrated quickly if those fluids aren’t replenished. What’s more, our thirst signals diminish naturally as we age as well—meaning our body is not as good as it once was in letting us know that we need to rehydrate. This often creates a perfect storm that commonly leads to dehydration. In Pat’s case, her dehydration was so severe she nearly died.
When Lewis Hornby visited his grandmother at her nursing home afterward, he learned that dehydration especially affects people with dementia, as they often don’t feel thirst cues at all, or may not recognize how to use cups correctly. But while dementia patients often don’t remember to drink water, it seemed to Hornby that they had less problem remembering to eat, particularly candy.

Hornby wanted to create a solution for elderly people who struggled keeping their fluid intake up. He spent the next eighteen months researching and designing a solution and securing funding for his project. In 2019, Hornby won a sizable grant from the Alzheimer’s Society, a UK-based care and research charity for people with dementia and their caregivers. Together, through the charity’s Accelerator Program, they created a bite-sized, sugar-free, edible jelly drop that looked and tasted like candy. The candy, called Jelly Drops, contained 95% water and electrolytes—important minerals that are often lost during dehydration. The final product launched in 2020—and was an immediate success. The drops were able to provide extra hydration to the elderly, as well as help keep dementia patients safe, since dehydration commonly leads to confusion, hospitalization, and sometimes even death.
Not only did Jelly Drops quickly become a favorite snack among dementia patients in the UK, but they were able to provide an additional boost of hydration to hospital workers during the pandemic. In NHS coronavirus hospital wards, patients infected with the virus were regularly given Jelly Drops to keep their fluid levels normal—and staff members snacked on them as well, since long shifts and personal protective equipment (PPE) they were required to wear often left them feeling parched.
In April 2022, Jelly Drops launched in the United States. The company continues to donate 1% of its profits to help fund Alzheimer’s research.

