New Options Are Emerging in the Search for Better Birth Control
A decade ago, Elizabeth Summers' options for birth control suddenly narrowed. Doctors diagnosed her with Factor V Leiden, a rare genetic disorder, after discovering blood clots in her lungs. The condition increases the risk of clotting, so physicians told Summers to stay away from the pill and other hormone-laden contraceptives. "Modern medicine has generally failed to provide me with an effective and convenient option," she says.
But new birth control options are emerging for women like Summers. These alternatives promise to provide more choices to women who can't ingest hormones or don't want to suffer their unpleasant side effects.
These new products have their own pros and cons. Still, doctors are welcoming new contraceptives following a long drought in innovation. "It's been a long time since we've had something new in the world of contraception," says Heather Irobunda, an obstetrician and gynecologist at NYC Health and Hospitals.
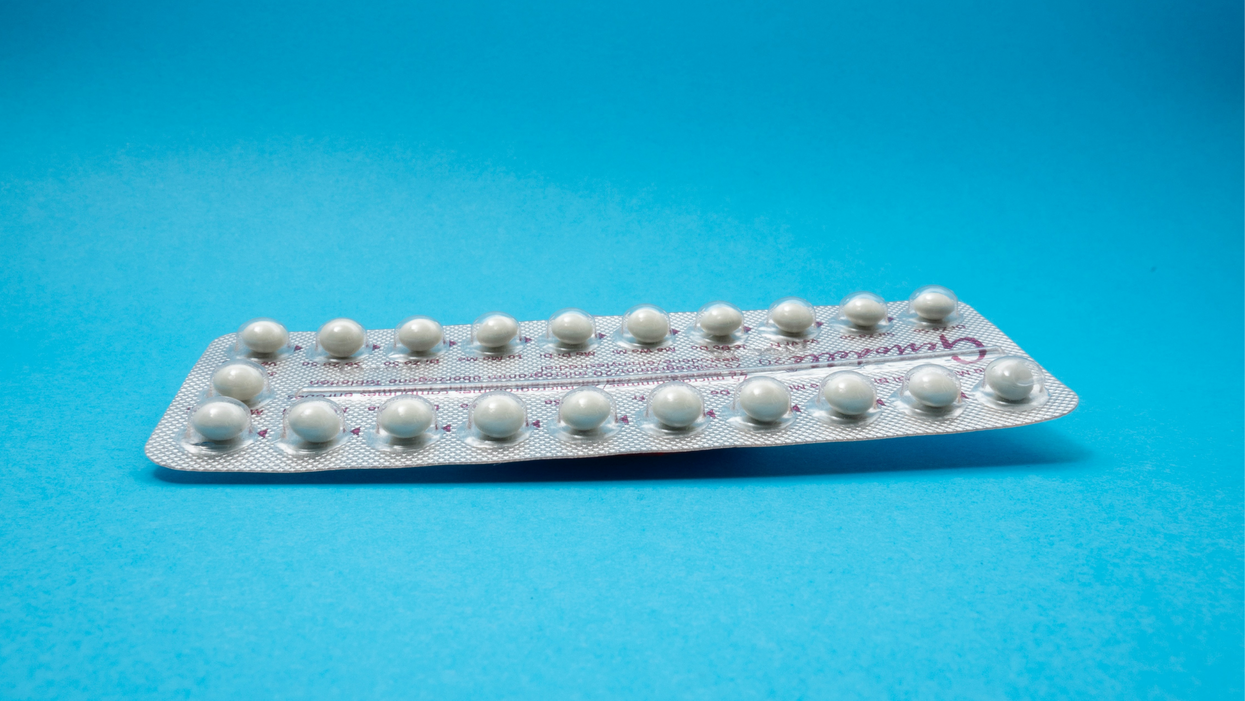
On social media, Irobunda often fields questions about one of these new options, a lubricating gel called Phexxi. San Diego-based Evofem, the company behind Phexxi, has been advertising the product on Hulu and Instagram after the gel was approved by the Food and Drug Administration in May 2020. The company's trendy ads target women who feel like condoms diminish the mood, but who also don't want to mess with an IUD or hormones.
Here's how it works: Phexxi is inserted via a tampon-like device up to an hour before sex. The gel regulates vaginal pH — essentially, the acidity levels — in a range that's inhospitable to sperm. It sounds a lot like spermicide, which is also placed in the vagina prior to sex to prevent pregnancy. But spermicide can damage the vagina's cell walls, which can increase the risk of contracting sexually transmitted diseases.
"Not only is innovation needed, but women want a non-hormonal option."
Phexxi isn't without side effects either. The most common one is vaginal burning, according to a late-stage trial. It's also possible to develop a urinary tract infection while using the product. That same study found that during typical use, Phexxi is about 86 percent effective at preventing pregnancy. The efficacy rate is comparable to condoms but lower than birth control pills (91 percent) and significantly lower than an IUD (99 percent).
Phexxi – which comes in a pack of 12 – represents a tiny but growing part of the birth control market. Pharmacies dispensed more than 14,800 packs from April through June this year, a 65 percent increase over the previous quarter, according to data from Evofem.
"We've been able to demonstrate that not only is innovation needed, but women want a non-hormonal option," says Saundra Pelletier, Evofem's CEO.
Beyond contraception, the company is carrying out late-stage tests to gauge Phexxi's effectiveness at preventing the sexually transmitted infections chlamydia and gonorrhea.

Phexxi is inserted via a tampon-like device up to an hour before sex.
Phexxi
A New Pill
The first birth control pill arrived in 1960, combining the hormones estrogen and progestin to stop sperm from joining with an egg, giving women control over their fertility. Subsequent formulations sought to ease side effects, by way of lower amounts of estrogen. But some women still experience headaches and nausea – or more serious complications like blood clots. On social media, women recently noted that birth control pills are much more likely to cause blood clots than Johnson & Johnson's COVID-19 vaccine that was briefly paused to evaluate the risk of clots in women under age 50. What will it take, they wondered, for safer birth control?
Mithra Pharmaceuticals of Belgium sought to create a gentler pill. In April, the FDA approved Mithra's Nextstellis, which includes a naturally occurring estrogen, the first new estrogen in the U.S. in 50 years. Nextstellis selectively acts on tissues lining the uterus, while other birth control pills have a broader target.
A Phase 3 trial showed a 98 percent efficacy rate. Andrew London, an obstetrician and gynecologist, who practices at several Maryland hospitals, says the results are in line with some other birth control pills. But, he added, early studies indicate that Nextstellis has a lower risk of blood clotting, along with other potential benefits, which additional clinical testing must confirm.
"It's not going to be worse than any other pill. We're hoping it's going to be significantly better," says London.
The estrogen in Nexstellis, called estetrol, was skipped over by the pharmaceutical industry after its discovery in the 1960s. Estetrol circulates between the mother and fetus during pregnancy. Decades later, researchers took a new look, after figuring out how to synthesize estetrol in a lab, as well as produce estetrol from plants.
"That allowed us to really start to investigate the properties and do all this stuff you have to do for any new drug," says Michele Gordon, vice president of marketing in women's health at Mayne Pharma, which licensed Nextstellis.
Bonnie Douglas, who followed the development of Nextstellis as part of a search for better birth control, recently switched to the product. "So far, it's much more tolerable," says Douglas. Previously, the Midwesterner was so desperate to find a contraceptive with fewer side effects that she turned to an online pharmacy to obtain a different birth control pill that had been approved in Canada but not in the U.S.
Contraceptive Access
Even if a contraceptive lands FDA approval, access poses a barrier. Getting insurers to cover new contraceptives can be difficult. For the uninsured, state and federal programs can help, and companies should keep prices in a reasonable range, while offering assistance programs. So says Kelly Blanchard, president of the nonprofit Ibis Reproductive Health. "For innovation to have impact, you want to reach as many folks as possible," she says.
In addition, companies developing new contraceptives have struggled to attract venture capital. That's changing, though.
In 2015, Sabrina Johnson founded DARÉ Bioscience around the idea of women's health. She estimated the company would be fully funded in six months, based on her track record in biotech and the demand for novel products.
But it's been difficult to get male investors interested in backing new contraceptives. It took Johnson two and a half years to raise the needed funds, via a reverse merger that took the company public. "There was so much education that was necessary," Johnson says, adding: "The landscape has changed considerably."
Johnson says she would like to think DARÉ had something to do with the shift, along with companies like Organon, a spinout of pharma company Merck that's focused on reproductive health. In surveying the fertility landscape, DARÉ saw limited non-hormonal options. On-demand options – like condoms – can detract from the moment. Copper IUDs must be inserted by a doctor and removed if a woman wants to return to fertility, and this method can have onerous side effects.
So, DARÉ created Ovaprene, a hormone-free device that's designed to be inserted into the vagina monthly by the user. The mesh product acts as a barrier, while releasing a chemical that immobilizes sperm. In an early study, the company reported that Ovaprene prevented almost all sperm from entering the cervical canal. The results, DARÉ believes, indicate high efficacy.
A late-stage study, slated to kick off next year, will be the true judge. Should Ovaprene eventually win regulatory approval, drug giant Bayer will handle commercializing the device.
Other new forms of birth control in development are further out, and that's assuming they perform well in clinical trials. Among them: a once-a-month birth control pill, along with a male version of the birth control pill. The latter is often brought up among women who say it's high time that men take a more proactive role in birth control.
For Summers, her search for a safe and convenient birth control continues. She tried Phexxi, which caused irritation. Still, she's excited that a non-hormonal option now exists. "I'm sure it will work for others," she says.
Growing Human Organs Inside Pigs Could Save Lives, But the U.S. Won't Fund the Research
Pigs in a barn.
The shortage of organs is a public health menace. Approximately 120,000 people in the U.S. need a lifesaving organ transplant. Of those, approximately 75,000 patients are on the active waiting list. Every day, nearly 20 individuals die from the shortage of organs in the United States.
Ethical concerns about human-animal chimera research might be dramatically overblown.
Scientists worldwide are developing new methods with potential to save countless patients in need of organs. Such approaches have tremendous potential, if only ethical and regulatory challenges could be overcome first.
One way that scientists are proposing to increase the number of transplantable organs is to produce organs from patient stem cells. Owed to their ability to grow limitlessly in the lab and form all tissue types, pluripotent stem cells from patients, in principle, could supply an infinite amount of cells that could potentially be transplanted back into patients. Unfortunately, all efforts to generate organs that can be transplanted into patients from stem cells to date have been unsuccessful.
A different encouraging approach is to generate patient organs inside livestock species, such as pigs. In the latest methods, interspecies chimeras – animals containing cells from both humans and animals – are generated by introducing human stem cells into early-stage animal embryos. Key genes essential for organ formation are disabled, allowing the introduced human stem cells to fill the empty space. In theory, this strategy will produce a human organ inside pigs or sheep.
Creating chimeras is not new in biology. Chimeras, or animals comprised of tissues from two different individuals, have already been deployed in research. Mouse chimeras are routinely used to create genetically engineered mice to study genes. The concept of generating human organs inside pigs or sheep comes from previous studies involving interspecies chimeras generated between mice and rats. Past experiments have demonstrated that it is possible to generate a rat pancreas inside a mouse.
Scientific and Ethical Obstacles
Unfortunately, chimera research has faced hurdles that have impeded progress. Of note, attempts to generate interspecies chimeras by several groups have failed. The results of these studies indicate that human cells appear unable to grow inside mouse embryos. The levels of human chimerism – the number of human cells inside the host animal embryo – appear too low to support any human organ generation.
Another obstacle is that chimera generation is ethically controversial. Some question the moral status of an animal that is comprised of human and animal cells. The most concerning question is whether human cells will contribute to the host animal's brain, potentially altering the cognition of the animal. These issues have prompted scientists to proceed very cautiously with chimera experiments. However, such concerns might be dramatically overblown. This is because the levels of human chimerism are too low to cause any significant change in animal brain function.
The ethical controversy has affected research policy in the United States. In the United States, the National Institutes of Health (NIH), the major funding body of biomedical research, blocked funding for chimera research while ethical questions were considered. Later, it was proposed that a new review process would be instated for chimera research. However, no change in policy has actually happened. The restrictive NIH policy is a major barrier to chimera research progress because laboratories around the United States cannot obtain funding for it. Lifting the restrictions on NIH funding for chimera research would dramatically accelerate chimera research.
Nonetheless, despite the past and current hurdles that chimera research has faced, new advances are changing the landscape of chimera research.
It is time to lift restrictions on chimera research so that its promise can be fully realized.
Progress on the Horizon
Scientists are developing improved strategies to increase the numbers of cells in animal embryos to the point where it might be possible to generate a human organ in an animal. For example, it has been suggested that the human stem cells researchers have been using cannot grow in animals. Scientists have made advances in generating new types of human stem cells that might have an improved ability to form chimeras.
Additionally, scientists have identified some barriers responsible for the failure to generate chimeras. For example, preventing cell death and enhancing the ability of stem cells to compete with host animal tissues also improves the numbers of human cells to the point where human organs can be generated inside an animal.
Finally, a relaxation of regulatory hurdles in other countries has created a more permissive environment for human-animal interspecies chimera research. In March, the Japanese government approved the first such experiments that could comprise a new way of generating organs from patients for transplantation.
Additionally, in spite of the somewhat negative attention that chimera generation has received, the International Society for Stem Cell Research (ISSCR) supports the new Japanese policies allowing chimera experiments. The ISSCR maintains that research involving the generation of chimeras should be permitted, as long as rigorous oversight and ethics review occur.
Chimera research has the potential to transform medicine. Of all the impediments, the NIH restrictions on funding remain the single most significant barrier. It is time to lift restrictions on chimera research so that its promise can be fully realized. One day, it might be possible to grow patient-specific organs inside of livestock animals such as pigs and sheep, saving thousands of human lives. But to change our current policy, the public, scientists, and bioethicists must first agree that this critical cause is worth fighting for.
In China, Prisoners of Conscience Are Being Murdered for Their Organs to Fuel Transplant Tourism
A somber photo of the Great Wall of China.
Organ transplantation can dramatically improve or save lives. A heart transplant can literally give a person a new lease of life, while a kidney transplant frees the recipient from lengthy spells on dialysis.
A people's tribunal in London has recently found that in China, organs are sourced from prisoners of conscience who are killed on demand to fuel the lucrative organ transplantation market.
To protect the integrity of organ transplantation, there are strict ethical guidelines. When organs are sourced from deceased persons, the donation must be voluntary, donors must die naturally before any organs are taken, and death must not be hastened to provide organs. These ethical guidelines protect donors and provide assurance to transplant recipients that their organs have been sourced ethically.
However, not all countries follow these ethical guidelines. A people's tribunal in London has recently found that in China, organs are sourced from prisoners of conscience who are killed on demand to fuel the lucrative organ transplantation market. This conclusion, reported at the United Nations Human Rights Council on September 24, was not reached lightly.
The independent China Tribunal, made up of four human rights lawyers, one surgeon with transplant experience, an academic who specialises in China studies and a businessman with human rights interests, spent over a year looking at written materials and heard evidence from over 50 witnesses in five days of hearings. Their grim conclusion, that prisoners of conscience are murdered for their organs, confirms the findings of earlier investigations.
Questions first arose over China's transplant system when the numbers of transplants rose dramatically after 2000. Transplant capacity rapidly increased; new infrastructure was built and staff were trained. Hospital websites offered livers, hearts and kidneys available in a matter of days or weeks, for a price. Foreigners were encouraged to come to China to avoid lengthy transplant waiting lists in their home countries.
At the time, it was a mystery as to how China had a ready supply of organs, despite having no volunteer donation system. Eventually, in 2006, the Chinese government stated that organs were removed from prisoners who had been found guilty and sentenced to the death penalty. But this explanation did not ring true. Death row prisoners often have poor health, including high rates of infectious diseases, making them poor candidates for donation. By contrast, the organs offered for sale were promised to be healthy.
In 2006, the first clues about the source of the organs emerged. A woman called Annie reported that her surgeon husband had been present during organ removal from Falun Gong practitioners who were still breathing as the scalpels cut into them. A subsequent investigation by two Canadian human rights lawyers examined multiple sources of evidence, concluding that murdered Falun Gong practitioners were indeed the source of the organs.
The evidence included testimony from practitioners who had been imprisoned, tortured, and later released. During imprisonment, many practitioners reported blood and other medical tests examining the health of their organs—tests that were not performed on any other prisoners. Phone calls made to Chinese hospitals by investigators posing as patients were offered rapid access to fresh organs from Falun Gong practitioners. The organs were guaranteed to be healthy, as the practice forbids smoking tobacco and drinking alcohol.
Since 2006, evidence has continued to accumulate. China has a huge transplant industry and no plausible source of voluntary organ donations. Unlike the rest of the world, Chinese waiting times remain very short. Foreigners continue to come to China to avoid lengthy waiting lists. Prisoners of conscience, including Tibetans and Uyghurs as well as Falun Gong practitioners, are still being imprisoned and medically tested.
The Chinese government continues to deny these crimes, claiming that there is a volunteer donor system in place.
The China Tribunal heard from Uyghur witnesses who had recently been inside the notorious labour camps (also called "re-education" centers) in Xin Xiang. The witnesses reported terrible conditions, including overcrowding and torture, and were forced to have medical examinations. They saw other prisoners disappear without explanation following similar medical tests. As recently as 2018, doctors in Chinese hospitals were promising potential patients healthy Falun Gong organs in taped phone calls.
The Chinese government continues to deny these crimes, claiming that there is a volunteer donor system in place. In the Chinese system, prisoners are counted as volunteers.
China's forced organ harvesting from prisoners of conscience has international implications. A recent study found that most published Chinese transplant research is based on organs sourced from prisoners. International ethical guidance prohibits taking organs from prisoners and prohibits publication of research based on transplanted material from prisoners. The authors of that study called for retractions of the papers, some of which are in well-known scientific journals. So far Transplantation and PLOS One are among the journals that have already retracted over twenty articles in response. On questioning from the editors, the authors of the papers failed to respond or could not verify that the organs in the transplant research came from volunteers.
The international community has a moral obligation to act together to stop forced organ harvesting in China.
The China Tribunal concluded that forced organ harvesting remains China's main source of transplant organs. In their view, the commission of Crimes Against Humanity against the Uyghurs and Falun Gong has been proved beyond reasonable doubt. By their actions, the Chinese government has turned a life-saving altruistic practice into our worst nightmare. The international community has a moral obligation to act together to stop forced organ harvesting in China, and end these crimes against humanity.

