New Options Are Emerging in the Search for Better Birth Control
A decade ago, Elizabeth Summers' options for birth control suddenly narrowed. Doctors diagnosed her with Factor V Leiden, a rare genetic disorder, after discovering blood clots in her lungs. The condition increases the risk of clotting, so physicians told Summers to stay away from the pill and other hormone-laden contraceptives. "Modern medicine has generally failed to provide me with an effective and convenient option," she says.
But new birth control options are emerging for women like Summers. These alternatives promise to provide more choices to women who can't ingest hormones or don't want to suffer their unpleasant side effects.
These new products have their own pros and cons. Still, doctors are welcoming new contraceptives following a long drought in innovation. "It's been a long time since we've had something new in the world of contraception," says Heather Irobunda, an obstetrician and gynecologist at NYC Health and Hospitals.
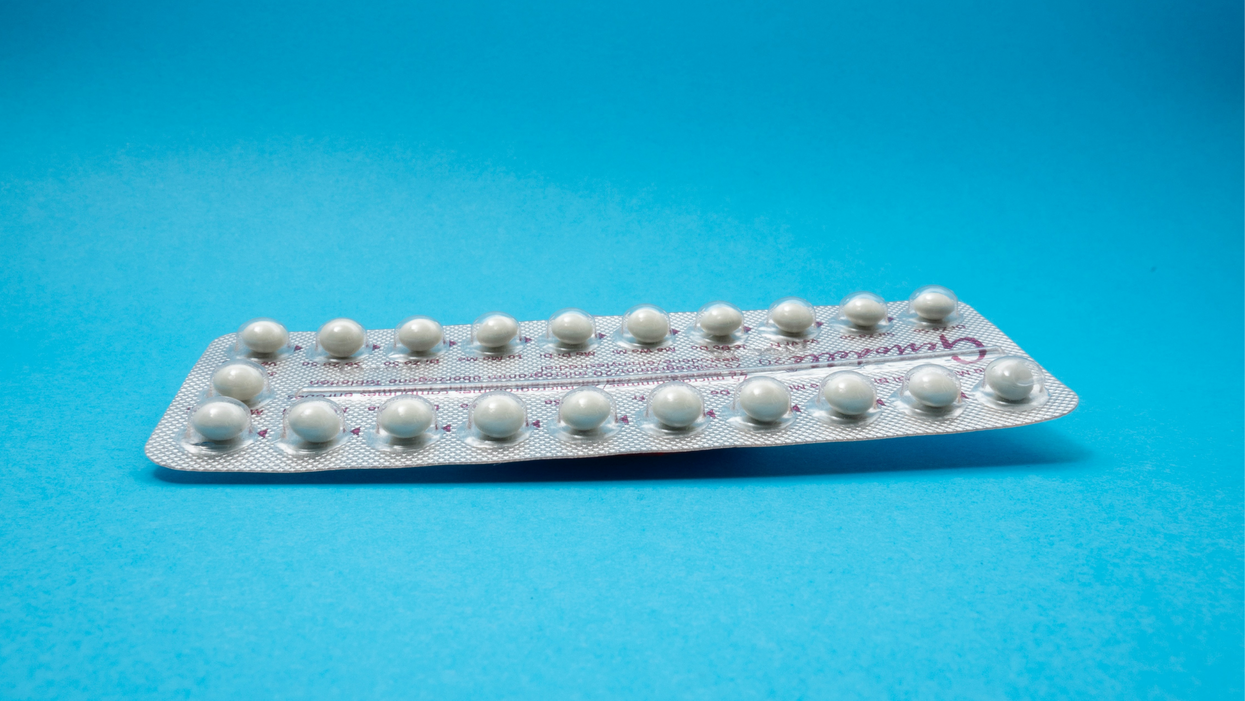
On social media, Irobunda often fields questions about one of these new options, a lubricating gel called Phexxi. San Diego-based Evofem, the company behind Phexxi, has been advertising the product on Hulu and Instagram after the gel was approved by the Food and Drug Administration in May 2020. The company's trendy ads target women who feel like condoms diminish the mood, but who also don't want to mess with an IUD or hormones.
Here's how it works: Phexxi is inserted via a tampon-like device up to an hour before sex. The gel regulates vaginal pH — essentially, the acidity levels — in a range that's inhospitable to sperm. It sounds a lot like spermicide, which is also placed in the vagina prior to sex to prevent pregnancy. But spermicide can damage the vagina's cell walls, which can increase the risk of contracting sexually transmitted diseases.
"Not only is innovation needed, but women want a non-hormonal option."
Phexxi isn't without side effects either. The most common one is vaginal burning, according to a late-stage trial. It's also possible to develop a urinary tract infection while using the product. That same study found that during typical use, Phexxi is about 86 percent effective at preventing pregnancy. The efficacy rate is comparable to condoms but lower than birth control pills (91 percent) and significantly lower than an IUD (99 percent).
Phexxi – which comes in a pack of 12 – represents a tiny but growing part of the birth control market. Pharmacies dispensed more than 14,800 packs from April through June this year, a 65 percent increase over the previous quarter, according to data from Evofem.
"We've been able to demonstrate that not only is innovation needed, but women want a non-hormonal option," says Saundra Pelletier, Evofem's CEO.
Beyond contraception, the company is carrying out late-stage tests to gauge Phexxi's effectiveness at preventing the sexually transmitted infections chlamydia and gonorrhea.

Phexxi is inserted via a tampon-like device up to an hour before sex.
Phexxi
A New Pill
The first birth control pill arrived in 1960, combining the hormones estrogen and progestin to stop sperm from joining with an egg, giving women control over their fertility. Subsequent formulations sought to ease side effects, by way of lower amounts of estrogen. But some women still experience headaches and nausea – or more serious complications like blood clots. On social media, women recently noted that birth control pills are much more likely to cause blood clots than Johnson & Johnson's COVID-19 vaccine that was briefly paused to evaluate the risk of clots in women under age 50. What will it take, they wondered, for safer birth control?
Mithra Pharmaceuticals of Belgium sought to create a gentler pill. In April, the FDA approved Mithra's Nextstellis, which includes a naturally occurring estrogen, the first new estrogen in the U.S. in 50 years. Nextstellis selectively acts on tissues lining the uterus, while other birth control pills have a broader target.
A Phase 3 trial showed a 98 percent efficacy rate. Andrew London, an obstetrician and gynecologist, who practices at several Maryland hospitals, says the results are in line with some other birth control pills. But, he added, early studies indicate that Nextstellis has a lower risk of blood clotting, along with other potential benefits, which additional clinical testing must confirm.
"It's not going to be worse than any other pill. We're hoping it's going to be significantly better," says London.
The estrogen in Nexstellis, called estetrol, was skipped over by the pharmaceutical industry after its discovery in the 1960s. Estetrol circulates between the mother and fetus during pregnancy. Decades later, researchers took a new look, after figuring out how to synthesize estetrol in a lab, as well as produce estetrol from plants.
"That allowed us to really start to investigate the properties and do all this stuff you have to do for any new drug," says Michele Gordon, vice president of marketing in women's health at Mayne Pharma, which licensed Nextstellis.
Bonnie Douglas, who followed the development of Nextstellis as part of a search for better birth control, recently switched to the product. "So far, it's much more tolerable," says Douglas. Previously, the Midwesterner was so desperate to find a contraceptive with fewer side effects that she turned to an online pharmacy to obtain a different birth control pill that had been approved in Canada but not in the U.S.
Contraceptive Access
Even if a contraceptive lands FDA approval, access poses a barrier. Getting insurers to cover new contraceptives can be difficult. For the uninsured, state and federal programs can help, and companies should keep prices in a reasonable range, while offering assistance programs. So says Kelly Blanchard, president of the nonprofit Ibis Reproductive Health. "For innovation to have impact, you want to reach as many folks as possible," she says.
In addition, companies developing new contraceptives have struggled to attract venture capital. That's changing, though.
In 2015, Sabrina Johnson founded DARÉ Bioscience around the idea of women's health. She estimated the company would be fully funded in six months, based on her track record in biotech and the demand for novel products.
But it's been difficult to get male investors interested in backing new contraceptives. It took Johnson two and a half years to raise the needed funds, via a reverse merger that took the company public. "There was so much education that was necessary," Johnson says, adding: "The landscape has changed considerably."
Johnson says she would like to think DARÉ had something to do with the shift, along with companies like Organon, a spinout of pharma company Merck that's focused on reproductive health. In surveying the fertility landscape, DARÉ saw limited non-hormonal options. On-demand options – like condoms – can detract from the moment. Copper IUDs must be inserted by a doctor and removed if a woman wants to return to fertility, and this method can have onerous side effects.
So, DARÉ created Ovaprene, a hormone-free device that's designed to be inserted into the vagina monthly by the user. The mesh product acts as a barrier, while releasing a chemical that immobilizes sperm. In an early study, the company reported that Ovaprene prevented almost all sperm from entering the cervical canal. The results, DARÉ believes, indicate high efficacy.
A late-stage study, slated to kick off next year, will be the true judge. Should Ovaprene eventually win regulatory approval, drug giant Bayer will handle commercializing the device.
Other new forms of birth control in development are further out, and that's assuming they perform well in clinical trials. Among them: a once-a-month birth control pill, along with a male version of the birth control pill. The latter is often brought up among women who say it's high time that men take a more proactive role in birth control.
For Summers, her search for a safe and convenient birth control continues. She tried Phexxi, which caused irritation. Still, she's excited that a non-hormonal option now exists. "I'm sure it will work for others," she says.
An image from one of TRIPP's virtual reality experiences for mental wellness. Nanea Reeves is TRIPP's CEO.
The "Making Sense of Science" podcast features interviews with leading experts about health innovations and the ethical questions they raise. The podcast is hosted by Matt Fuchs, editor of Leaps.org, the award-winning science outlet.
My guest today is Nanea Reeves, the CEO of TRIPP, a wellness platform with some big differences from meditation apps you may have tried like Calm and Headspace. TRIPP's experiences happen in virtual reality, and its realms are designed based on scientific findings about states of mindfulness. Users report feelings of awe and wonder and even mystical experiences. Nanea brings over 15 years of leadership in digital distribution, apps and video game technologies. Before co-founding TRIPP, she had several other leadership roles in tech with successful companies like textPlus and Machinima. Read her full bio below in the links section.

Nanea Reeves, CEO of TRIPP.
TRIPP
Listen to the Episode
Listen on Apple | Listen on Spotify | Listen on Stitcher | Listen on Amazon | Listen on Google
This conversation coincided with National Brain Awareness Week. The topic is a little different from the Making Sense of Science podcast’s usual focus on breakthroughs in treating and preventing disease, but there’s a big overlap when it comes to breakthroughs in optimal health. Nanea’s work is at the leading edge of health, technology and the science of wellness.
With TRIPP, you might find yourself deep underwater, looking up at the sunlight shimmering on the ocean surface, or in the cosmos staring down at a planet glowing with an arresting diversity of colors. Using TRIPP for the past six months has been a window for me into the future of science-informed wellness and an overall fascinating experience, as was my conversation with Nanea.
Show notes:
Nanea and I discuss her close family members' substance addictions and her own struggle with mental illness as a teen, which led to her first meditation experiences, and much more:
- The common perception that technology is an obstacle for mental well-being, a narrative that overlooks how tech can also increase wellness when it’s designed right.
- Emerging ways of measuring meditation experiences by recording brain waves - and the shortcomings of the ‘measured self’ movement.
- Why TRIPP’s users multiplied during the stress and anxiety of the pandemic, and how TRIPP can can be used to enhance emotional states.
- Ways in which TRIPP’s visuals and targeted sound frequencies have been informed by innovative research from psychologists like Johns Hopkins’ Matthew Johnson.
- Ways to design apps and other technologies to better fulfill the true purpose of mindfulness meditation. (Hint: not simply relaxation.)
- And of course, because the topic is mental wellness and tech, I had to get Nanea's thoughts on Elon Musk, Neuralink and brain machine interfaces.
Here are links for learning more about TRIPP:
- TRIPP website: https://www.tripp.com/about/
- Nanea Reeves bio: https://www.tripp.com/team/nanea-reeves/
- Study of data collected by UK's Office for National Statistics on behavior during the pandemic, which suggests that TRIPP enhanced users' psychological and emotional mindsets: https://link.springer.com/chapter/10.1007/978-3-03...
- Research that's informed TRIPP: https://www.tripp.com/research/
- Washington Post Top Pick at CES: https://www.washingtonpost.com/technology/2019/01/...
- TRIPP's new offering, PsyAssist, to provide support for ketamine-assisted therapy: https://www.mobihealthnews.com/news/tripp-acquires...
- Randomized pilot trial involving TRIPP: https://bmjopen.bmj.com/content/bmjopen/11/4/e0441...
Leaps.org spoke with Holden Thorp, Editor-in-Chief of the prestigious Science family of journals, about ways to improve science journalism, the challenges faced by experts, the lab leak theory and much more.
This month, Leaps.org had a chance to speak with Holden Thorp, Editor-in-Chief of the Science family of journals. We talked about the best ways to communicate science to the public, mistakes by public health officials during the pandemic, the lab leak theory, and bipartisanship for funding science research.
Before becoming editor of the Science journals, Thorp spent six years as provost of Washington University in St. Louis, where he is Rita Levi-Montalcini Distinguished University Professor and holds appointments in both chemistry and medicine. He joined Washington University after spending three decades at the University of North Carolina at Chapel Hill, where he served as the UNC's 10th chancellor from 2008 through 2013.
A North Carolina native, Thorp earned a doctorate in chemistry in 1989 at the California Institute of Technology and completed postdoctoral work at Yale University. He is a fellow of the National Academy of Inventors and the American Association for the Advancement of Science.
Read his full bio here.
This conversation was lightly edited by Leaps.org for style and format.
Matt Fuchs: You're a musician. It seems like many scientists are also musicians. Is there a link between the scientist brain and the musician brain?
Holden Thorp: I think [the overlap is] relatively common. I'm still a gigging bass player. I play in the pits for lots of college musicals. I think that it takes a certain discipline and requires you to learn a lot of rules about how music works, and then you try to be creative within that. That's similar to scientific research. So it makes sense. Music is something I've been able to sustain my whole life. I wouldn't be the same person if I let it go. When you're playing, especially for a musical, where the music is challenging, you can't let your mind wander. It’s like meditation.
MF: I bet it helps to do something totally different from your editing responsibilities. Maybe lets the subconscious take care of tough problems at work.
HT: Right.
MF: There's probably never been a greater need for clear and persuasive science communicators. Do we need more cross specialty training? For example, journalism schools prioritizing science training, and science programs that require more time learning how to communicate effectively?
HT: I think we need both. One of the challenges we've had with COVID has been, especially at the beginning, a lot of reporters who didn’t normally cover scientific topics got put on COVID—and ended up creating things that had to be cleaned up later. This isn't the last science-oriented crisis we're going to have. We've already got climate change, and we'll have another health crisis for sure. So it’d be good for journalism to be a little better prepared next time.
"Scientists are human beings who have ego and bravado and every other human weakness."
But on the other side, maybe it's even more important that scientists learn how to communicate and how likely it is that their findings will be politicized, twisted and miscommunicated. Because one thing that surprised me is how shocked a lot of scientists have been. Every scientific issue that reaches into public policy becomes politicized: climate change, evolution, stem cells.
Once one side decided to be cautious about the pandemic, you could be certain the other side was going to decide not to do that. That's not the fault of science. That’s just life in a political world. That, I think, caught people off guard. They weren't prepared to shape and process their messages in a way that accounted for that—and for the way that social media has intensified all of this.
MF: Early in the pandemic, there was a lack of clarity about public health recommendations, as you’d expect with a virus we hadn’t seen before. Should public officials and scientists have more humility in similar situations in the future? Public officials need to be authoritative for their guidance to be followed, so how do they lead a crisis response while displaying humility about what we don't know?
HS: I think scientists are people who like to have the answer. It's very tempting and common for scientists to kind of oversell what we know right now, while not doing as much as we should to remind people that science is a self-correcting process. And when we fail to do that – after we’ve collected more data and need to change how we're interpreting it – the people who want to undermine us have a perfect weapon to use against us. It's challenging. But I agree that scientists are human beings who have ego and bravado and every other human weakness.
For example, we wanted to tell everybody that we thought the vaccines would provide sterilizing immunity against infection. Well, we don't have too many other respiratory viruses where that's the case. And so it was more likely that we were going to have what we ended up with, which is that the vaccines were excellent in preventing severe disease and death. It would have been great if they provided sterilizing immunity and abruptly ended the pandemic a year ago. But it was overly optimistic to think that was going to be the case in retrospect.
MF: Both in terms of how science is communicated and received by the public, do we need to reform institutions or start new ones to instill the truth-seeking values that are so important to appreciating science?
HS: There are a whole bunch of different factors. I think the biggest one is that the social media algorithms reward their owners financially when they figure out how to keep people in their silos. Users are more likely to click on things that they agree with—and that promote conflict with people that they disagree with. That has caused an acceleration in hostilities that attend some of these disagreements.
But I think the other problem is that we haven’t found a way to explain things to people when it’s not a crisis. So, for example, a strong indicator of whether someone who might otherwise be vaccine hesitant decided to get their vaccine is if they understood how vaccines worked before the pandemic started. Because if you're trying to tell somebody that they're wrong if they don't get a vaccine, at the same time you're trying to explain how it works, that's a lot of explaining to do in a short period of time.
Lack of open-mindedness is a problem, but another issue is that we need more understanding of these issues baked into the culture already. That's partly due the fact that there hasn't been more reform in K through 12 and college teaching. And that scientists are very comfortable talking to each other, and not very comfortable talking to people who don't know all of our jargon and have to be persuaded to spend time listening to and thinking about what we're trying to tell them.
"We're almost to the point where clinging to the lab leak idea is close to being a fringe idea that almost doesn't need to be included in stories."
MF: You mentioned silos. There have been some interesting attempts in recent years to do “both sides journalism,” where websites like AllSides put different views on high profile issues side-by-side. Some people believe that's how the news should be reported. Should we let people see and decide for themselves which side is the most convincing?
HS: It depends if we're talking about science. On scientific issues, when they start, there's legitimate disagreement about among scientists. But eventually, things go back and forth, and people compete with each other and work their way to the answer. At some point, we reach more of a consensus.
For example, on climate change, I think it's gotten to the point now where it's irresponsible, if you're writing a story about climate change, to run a quote from somebody somewhere who's still—probably because of their political views—clinging to the idea that anthropogenic global warming is somehow not damaging the planet.
On things that aren't decided yet, that makes sense to run both. It's more a question of judgment of the journalists. I don't think the solution to it is put stark versions of each side, side-by-side and let people choose. The whole point of journalism is to inform people. If there's a consensus on something, that's part of what you're supposed to be informing them about.
MF: What about reporting on perspectives about the lab leak theory at various times during the pandemic?
HS: We’re the outlet that ran the letter that really restarted the whole debate. A bunch of well-known scientists said we should consider the lab leak theory more carefully. And in the aftermath of that, a bunch of those scientists who signed that letter concluded that the lab leak was very, very unlikely. Interestingly, publishing that letter actually drove us to more of a consensus. I would say now, we're almost to the point where clinging to the lab leak idea is close to being a fringe idea that almost doesn't need to be included in stories. But I would say there's been a lot of evolution on that over the last year since we ran that letter.
MF: Let's talk about bipartisanship in Congress. Research funding for the National Institutes of Health was championed for years by influential Republicans who supported science to advance health breakthroughs. Is that changing? Maybe especially with Sen. Roy Blunt retiring? Has bipartisanship on science funding been eroded by political battles during COVID?
HS: I'm optimistic that that won't be the case. Republican Congresses have usually been good for science funding. And that's because (former Sen.) Arlen Specter and Roy Blunt are two of the political figures who have pushed for science funding over the last couple decades. With Blunt retiring, we don't know who's going to step in for him. That's an interesting question. I hope there will be Republican champions for science funding.
MF: Is there too much conservatism baked into how we research new therapies and bring them to people who are sick, bench-to-bedside? I'm thinking of the criticisms that NIH or the FDA are overly bureaucratic. Are you hopeful about ARPA-H, President Biden’s proposed new agency for health innovation?
HS: I think the challenge hasn't been cracked by the federal government. Maybe DARPA has done this outside of health science, but within health science, the federal government has had limited success at funding things that can be applied quickly, while having overwhelming success at funding basic research that eventually becomes important in applications. Can they do it the other way around? They’ll need people running ARPA-H who are application first. It’s ambitious. The way it was done in Operation Warp Speed is all the money was just given to the companies. If the hypothesis on ARPA-H is for the federal government to actually do what Moderna and BioNTech did for the vaccine, themselves, that's a radical idea. It's going to require thinking very differently than the way they think about dispersing grants for basic research.
MF: You’ve written a number of bold op-eds as editor of the Science journals. Are there any op-eds you're especially proud of as voicing a view that was important but not necessarily popular?
HS: I was one of the first people to come out hard against President Trump['s handling of] the pandemic. Lots of my brothers and sisters came along afterwards. To the extent that I was able to catalyze that, I'm proud of doing it. In the last few weeks, I published a paper objecting to the splitting of the OSTP director from the science advisor and, especially, not awarding the top part of the job to Alondra Nelson, who is a distinguished scientist at black female. And instead, giving part of it to Francis Collins. He’s certainly the most important science policy figure of my lifetime, but somebody who’s been doing this now for decades. I just think we have to push as hard as we can to get a cadre of young people leading us in Washington who represent the future of the country. I think the Biden administration leaned on a lot of figures from the past. I’m pushing them hard to try to stop it.
MF: I want to circle back to the erosion of the public’s trust in experts. Most experts are specialists, and specialists operate in silos that don’t capture the complexity of scientific knowledge. Are some pushbacks to experts and concerns about the perils of specialization valid?
HS: You're on the right track there. What we need is more respect for the generalist. We can't help the fact that you have to be very specialized to do a lot of stuff. But what we need is more partnership between specialists and people who can cross fields, especially into communication and social sciences. That handoff is just not really there right now. It's hard to get a hardcore scientist to respect people who are interested in science, education and science communication, and to treat them as equals. The last two years showed that they're at least as important, if not more so.
MF: I’m grateful that you’re leading the way in this area, Holden. Thank you for sharing your thoughts and your work.

