The New Prospective Parenthood: When Does More Info Become Too Much?
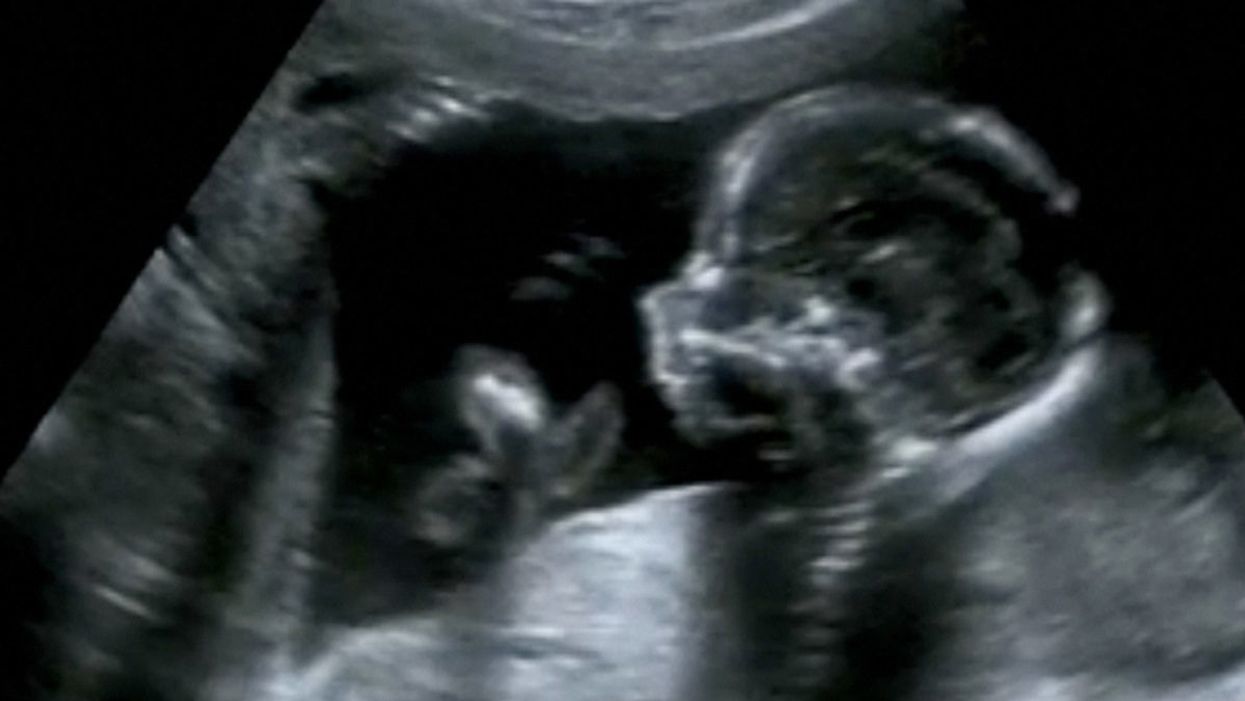
Obstetric ultrasound of a fourth-month fetus.
Peggy Clark was 12 weeks pregnant when she went in for a nuchal translucency (NT) scan to see whether her unborn son had Down syndrome. The sonographic scan measures how much fluid has accumulated at the back of the baby's neck: the more fluid, the higher the likelihood of an abnormality. The technician said the baby was in such an odd position, the test couldn't be done. Clark, whose name has been changed to protect her privacy, was told to come back in a week and a half to see if the baby had moved.
"With the growing sophistication of prenatal tests, it seems that the more questions are answered, the more new ones arise."
"It was like the baby was saying, 'I don't want you to know,'" she recently recalled.
When they went back, they found the baby had a thickened neck. It's just one factor in identifying Down's, but it's a strong indication. At that point, she was 13 weeks and four days pregnant. She went to the doctor the next day for a blood test. It took another two weeks for the results, which again came back positive, though there was still a .3% margin of error. Clark said she knew she wanted to terminate the pregnancy if the baby had Down's, but she didn't want the guilt of knowing there was a small chance the tests were wrong. At that point, she was too late to do a Chorionic villus sampling (CVS), when chorionic villi cells are removed from the placenta and sequenced. And she was too early to do an amniocentesis, which isn't done until between 14 and 20 weeks of the pregnancy. So she says she had to sit and wait, calling those few weeks "brutal."
By the time they did the amnio, she was already nearly 18 weeks pregnant and was getting really big. When that test also came back positive, she made the anguished decision to end the pregnancy.
Now, three years after Clark's painful experience, a newer form of prenatal testing routinely gives would-be parents more information much earlier on, especially for women who are over 35. As soon as nine weeks into their pregnancies, women can have a simple blood test to determine if there are abnormalities in the DNA of chromosomes 21, which indicates Down syndrome, as well as in chromosomes 13 and 18. Using next-generation sequencing technologies, the test separates out and examines circulating fetal cells in the mother's blood, which eliminates the risks of drawing fluid directly from the fetus or placenta.
"Finding out your baby has Down syndrome at 11 or 12 weeks is much easier for parents to make any decision they may want to make, as opposed to 16 or 17 weeks," said Dr. Leena Nathan, an obstetrician-gynecologist in UCLA's healthcare system. "People are much more willing or able to perhaps make a decision to terminate the pregnancy."
But with the growing sophistication of prenatal tests, it seems that the more questions are answered, the more new ones arise--questions that previous generations have never had to face. And as genomic sequencing improves in its predictive accuracy at the earliest stages of life, the challenges only stand to increase. Imagine, for example, learning your child's lifetime risk of breast cancer when you are ten weeks pregnant. Would you terminate if you knew she had a 70 percent risk? What about 40 percent? Lots of hard questions. Few easy answers. Once the cost of whole genome sequencing drops low enough, probably within the next five to ten years according to experts, such comprehensive testing may become the new standard of care. Welcome to the future of prospective parenthood.
"In one way, it's a blessing to have this information. On the other hand, it's very difficult to deal with."
How Did We Get Here?
Prenatal testing is not new. In 1979, amniocentesis was used to detect whether certain inherited diseases had been passed on to the fetus. Through the 1980s, parents could be tested to see if they carried disease like Tay-Sachs, Sickle cell anemia, Cystic fibrosis and Duchenne muscular dystrophy. By the early 1990s, doctors could test for even more genetic diseases and the CVS test was beginning to become available.
A few years later, a technique called preimplantation genetic diagnosis (PGD) emerged, in which embryos created in a lab with sperm and harvested eggs would be allowed to grow for several days and then cells would be removed and tested to see if any carried genetic diseases. Those that weren't affected could be transferred back to the mother. Once in vitro fertilization (IVF) took off, so did genetic testing. The labs test the embryonic cells and get them back to the IVF facilities within 24 hours so that embryo selection can occur. In the case of IVF, genetic tests are done so early, parents don't even have to decide whether to terminate a pregnancy. Embryos with issues often aren't even used.
"It was a very expensive endeavor but exciting to see our ability to avoid disorders, especially for families that don't want to terminate a pregnancy," said Sara Katsanis, an expert in genetic testing who teaches at Duke University. "In one way, it's a blessing to have this information (about genetic disorders). On the other hand, it's very difficult to deal with. To make that decision about whether to terminate a pregnancy is very hard."
Just Because We Can, Does It Mean We Should?
Parents in the future may not only find out whether their child has a genetic disease but will be able to potentially fix the problem through a highly controversial process called gene editing. But because we can, does it mean we should? So far, genes have been edited in other species, but to date, the procedure has not been used on an unborn child for reproductive purposes apart from research.
"There's a lot of bioethics debate and convening of groups to try to figure out where genetic manipulation is going to be useful and necessary, and where it is going to need some restrictions," said Katsanis. She notes that it's very useful in areas like cancer research, so one wouldn't want to over-regulate it.
There are already some criteria as to which genes can be manipulated and which should be left alone, said Evan Snyder, professor and director of the Center for Stem Cells and Regenerative Medicine at Sanford Children's Health Research Center in La Jolla, Calif. He noted that genes don't stand in isolation. That is, if you modify one that causes disease, will it disrupt others? There may be unintended consequences, he added.
"As the technical dilemmas get fixed, some of the ethical dilemmas get fixed. But others arise. It's kind of like ethical whack-a-mole."
But gene editing of embryos may take years to become an acceptable practice, if ever, so a more pressing issue concerns the rationale behind embryo selection during IVF. Prospective parents can end up with anywhere from zero to thirty embryos from the procedure and must choose only one (rarely two) to implant. Since embryos are routinely tested now for certain diseases, and selected or discarded based on that information, should it be ethical—and legal—to make selections based on particular traits, too? To date so far, parents can select for gender, but no other traits. Whether trait selection becomes routine is a matter of time and business opportunity, Katsanis said. So far, the old-fashioned way of making a baby combined with the luck of the draw seems to be the preferred method for the marketplace. But that could change.
"You can easily see a family deciding not to implant a lethal gene for Tay-Sachs or Duchene or Cystic fibrosis. It becomes more ethically challenging when you make a decision to implant girls and not any of the boys," said Snyder. "And then as we get better and better, we can start assigning genes to certain skills and this starts to become science fiction."
Once a pregnancy occurs, prospective parents of all stripes will face decisions about whether to keep the fetus based on the information that increasingly robust prenatal testing will provide. What influences their decision is the crux of another ethical knot, said Snyder. A clear-cut rationale would be if the baby is anencephalic, or it has no brain. A harder one might be, "It's a girl, and I wanted a boy," or "The child will only be 5' 2" tall in adulthood."
"Those are the extremes, but the ultimate question is: At what point is it a legitimate response to say, I don't want to keep this baby?'" he said. Of course, people's responses will vary, so the bigger conundrum for society is: Where should a line be drawn—if at all? Should a woman who is within the legal scope of termination (up to around 24 weeks, though it varies by state) be allowed to terminate her pregnancy for any reason whatsoever? Or must she have a so-called "legitimate" rationale?
"As the technical dilemmas get fixed, some of the ethical dilemmas get fixed. But others arise. It's kind of like ethical whack-a-mole," Snyder said.
One of the newer moles to emerge is, if one can fix a damaged gene, for how long should it be fixed? In one child? In the family's whole line, going forward? If the editing is done in the embryo right after the egg and sperm have united and before the cells begin dividing and becoming specialized, when, say, there are just two or four cells, it will likely affect that child's entire reproductive system and thus all of that child's progeny going forward.
"This notion of changing things forever is a major debate," Snyder said. "It literally gets into metaphysics. On the one hand, you could say, well, wouldn't it be great to get rid of Cystic fibrosis forever? What bad could come of getting rid of a mutant gene forever? But we're not smart enough to know what other things the gene might be doing, and how disrupting one thing could affect this network."
As with any tool, there are risks and benefits, said Michael Kalichman, Director of the Research Ethics Program at the University of California San Diego. While we can envision diverse benefits from a better understanding of human biology and medicine, it is clear that our species can also misuse those tools – from stigmatizing children with certain genetic traits as being "less than," aka dystopian sci-fi movies like Gattaca, to judging parents for making sure their child carries or doesn't carry a particular trait.
"The best chance to ensure that the benefits of this technology will outweigh the risks," Kalichman said, "is for all stakeholders to engage in thoughtful conversations, strive for understanding of diverse viewpoints, and then develop strategies and policies to protect against those uses that are considered to be problematic."
New implants let paraplegics surf the web and play computer games
Rodney Gorham, an Australian living with ALS, has reconnected with the world, thanks to a brain-machine interface called the Stentrode.
When I greeted Rodney Gorham, age 63, in an online chat session, he replied within seconds: “My pleasure.”
“Are you moving parts of your body as you type?” I asked.
This time, his response came about five minutes later: “I position the cursor with the eye tracking and select the same with moving my ankles.” Gorham, a former sales representative from Melbourne, Australia, living with amyotrophic lateral sclerosis, or ALS, a rare form of Lou Gehrig’s disease that impairs the brain’s nerve cells and the spinal cord, limiting the ability to move. ALS essentially “locks” a person inside their own body. Gorham is conversing with me by typing with his mind only–no fingers in between his brain and his computer.
The brain-computer interface enabling this feat is called the Stentrode. It's the brainchild of Synchron, a company backed by Amazon’s Jeff Bezos and Microsoft cofounder Bill Gates. After Gorham’s neurologist recommended that he try it, he became one of the first volunteers to have an 8mm stent, laced with small electrodes, implanted into his jugular vein and guided by a surgeon into a blood vessel near the part of his brain that controls movement.
After arriving at their destination, these tiny sensors can detect neural activity. They relay these messages through a small receiver implanted under the skin to a computer, which then translates the information into words. This minimally invasive surgery takes a day and is painless, according to Gorham. Recovery time is typically short, about two days.
When a paralyzed patient thinks about trying to move their arms or legs, the motor cortex will fire patterns that are specific to the patient’s thoughts.
When a paralyzed patient such as Gorham thinks about trying to move their arms or legs, the motor cortex will fire patterns that are specific to the patient’s thoughts. This pattern is detected by the Stentrode and relayed to a computer that learns to associate this pattern with the patient’s physical movements. The computer recognizes thoughts about kicking, making a fist and other movements as signals for clicking a mouse or pushing certain letters on a keyboard. An additional eye-tracking device controls the movement of the computer cursor.
The process works on a letter by letter basis. That’s why longer and more nuanced responses often involve some trial and error. “I have been using this for about two years, and I enjoy the sessions,” Gorham typed during our chat session. Zafar Faraz, field clinical engineer at Synchron, sat next to Gorham, providing help when required. Gorham had suffered without internet access, but now he looks forward to surfing the web and playing video games.

Gorham, age 63, has been enjoying Stentrode sessions for about two years.
Rodeny Dekker
The BCI revolution
In the summer of 2021, Synchron became the first company to receive the FDA’s Investigational Device Exemption, which allows research trials on the Stentrode in human patients. This past summer, the company, together with scientists from Icahn School of Medicine at Mount Sinai and the Neurology and Neurosurgery Department at Utrecht University, published a paper offering a framework for how to develop BCIs for patients with severe paralysis – those who can't use their upper limbs to type or use digital devices.
Three months ago, Synchron announced the enrollment of six patients in a study called COMMAND based in the U.S. The company will seek approval next year from the FDA to make the Stentrode available for sale commercially. Meanwhile, other companies are making progress in the field of BCIs. In August, Neuralink announced a $280 million financing round, the biggest fundraiser yet in the field. Last December, Synchron announced a $75 million financing round. “One thing I can promise you, in five years from now, we’re not going to be where we are today. We're going to be in a very different place,” says Elad I. Levy, professor of neurosurgery and radiology at State University of New York in Buffalo.
The risk of hacking exists, always. Cybercriminals, for example, might steal sensitive personal data for financial reasons, blackmailing, or to spread malware to other connected devices while extremist groups could potentially hack BCIs to manipulate individuals into supporting their causes or carrying out actions on their behalf.
“The prospect of bestowing individuals with paralysis a renewed avenue for communication and motor functionality is a step forward in neurotech,” says Hayley Nelson, a neuroscientist and founder of The Academy of Cognitive and Behavioral Neuroscience. “It is an exciting breakthrough in a world of devastating, scary diseases,” says Neil McArthur, a professor of philosophy and director of the Centre for Professional and Applied Ethics at the University of Manitoba. “To connect with the world when you are trapped inside your body is incredible.”
While the benefits for the paraplegic community are promising, the Stentrode’s long-term effectiveness and overall impact needs more research on safety. “Potential risks like inflammation, damage to neural tissue, or unexpected shifts in synaptic transmission due to the implant warrant thorough exploration,” Nelson says.
There are also concens about data privacy concerns and the policies of companies to safeguard information processed through BCIs. “Often, Big Tech is ahead of the regulators because the latter didn’t envisage such a turn of events...and companies take advantage of the lack of legal framework to push forward,” McArthur says. Hacking is another risk. Cybercriminals could steal sensitive personal data for financial reasons, blackmailing, or to spread malware to other connected devices. Extremist groups could potentially hack BCIs to manipulate individuals into supporting their causes or carrying out actions on their behalf.
“We have to protect patient identity, patient safety and patient integrity,” Levy says. “In the same way that we protect our phones or computers from hackers, we have to stay ahead with anti-hacking software.” Even so, Levy thinks the anticipated benefits for the quadriplegic community outweigh the potential risks. “We are on the precipice of an amazing technology. In the future, we would be able to connect patients to peripheral devices that enhance their quality of life.”
In the near future, the Stentrode could enable patients to use the Stentrode to activate their wheelchairs, iPods or voice modulators. Synchron's focus is on using its BCI to help patients with significant mobility restrictions—not to enhance the lives of healthy people without any illnesses. Levy says we are not prepared for the implications of endowing people with superpowers.
I wondered what Gorham thought about that. “Pardon my question, but do you feel like you have sort of transcended human nature, being the first in a big line of cybernetic people doing marvelous things with their mind only?” was my last question to Gorham.
A slight smile formed on his lips. In less than a minute, he typed: “I do a little.”
Leading XPRIZE Healthspan and Beating Negativity with Dr. Peter Diamandis
XPRIZE founder and chairman Peter Diamandis launches XPRIZE Healthspan at an event on November 29.
A new competition by the XPRIZE Foundation is offering $101 million to researchers who discover therapies that give a boost to people aged 65-80 so their bodies perform more like when they were middle-aged.
For today’s podcast episode, I talked with Dr. Peter Diamandis, XPRIZE’s founder and executive chairman. Under Peter’s leadership, XPRIZE has launched 27 previous competitions with over $300 million in prize purses. The latest contest aims to enhance healthspan, or the period of life when older people can play with their grandkids without any restriction, disability or disease. Such breakthroughs could help prevent chronic diseases that are closely linked to aging. These illnesses are costly to manage and threaten to overwhelm the healthcare system, as the number of Americans over age 65 is rising fast.
In this competition, called XPRIZE Healthspan, multiple awards are available, depending on what’s achieved, with support from the nonprofit Hevolution Foundation and Chip Wilson, the founder of Lululemon and nonprofit SOLVE FSHD. The biggest prize, $81 million, is for improvements in cognition, muscle and immunity by 20 years. An improvement of 15 years will net $71 million, and 10 years will net $61 million.
In our conversation for this episode, Peter talks about his plans for XPRIZE Healthspan and why exponential technologies make the current era - even with all of its challenges - the most exciting time in human history. We discuss the best mental outlook that supports a person in becoming truly innovative, as well as the downsides of too much risk aversion. We talk about how to overcome the negativity bias in ourselves and in mainstream media, how Peter has shifted his own mindset to become more positive over the years, how to inspire a culture of innovation, Peter’s personal recommendations for lifestyle strategies to live longer and healthier, the innovations we can expect in various fields by 2030, the future of education and the importance of democratizing tech and innovation.
In addition to Peter’s pioneering leadership of XPRIZE, he is also the Executive Founder of Singularity University. In 2014, he was named by Fortune as one of the “World’s 50 Greatest Leaders.” As an entrepreneur, he’s started over 25 companies in the areas of health-tech, space, venture capital and education. He’s Co-founder and Vice-Chairman of two public companies, Celularity and Vaxxinity, plus being Co-founder & Chairman of Fountain Life, a fully-integrated platform delivering predictive, preventative, personalized and data-driven health. He also serves as Co-founder of BOLD Capital Partners, a venture fund with a half-billion dollars under management being invested in exponential technologies and longevity companies. Peter is a New York Times Bestselling author of four books, noted during our conversation and in the show notes of this episode. He has degrees in molecular genetics and aerospace engineering from MIT and holds an M.D. from Harvard Medical School.
Show links
- Peter Diamandis bio
- New XPRIZE Healthspan
- Peter Diamandis books
- 27 XPRIZE competitions and counting
- Life Force by Peter Diamandis and Tony Robbins
- Peter Diamandis Twitter
- Longevity Insider newsletter – AI identifies the news
- Peter Diamandis Longevity Handbook
- Hevolution funding for longevity

XPRIZE Founder Peter Diamandis speaks with Mehmoud Khan, CEO of Hevolution Foundation, at the launch of XPRIZE Healthspan.
Hevolution Foundation

