Are the gains from gain-of-function research worth the risks?
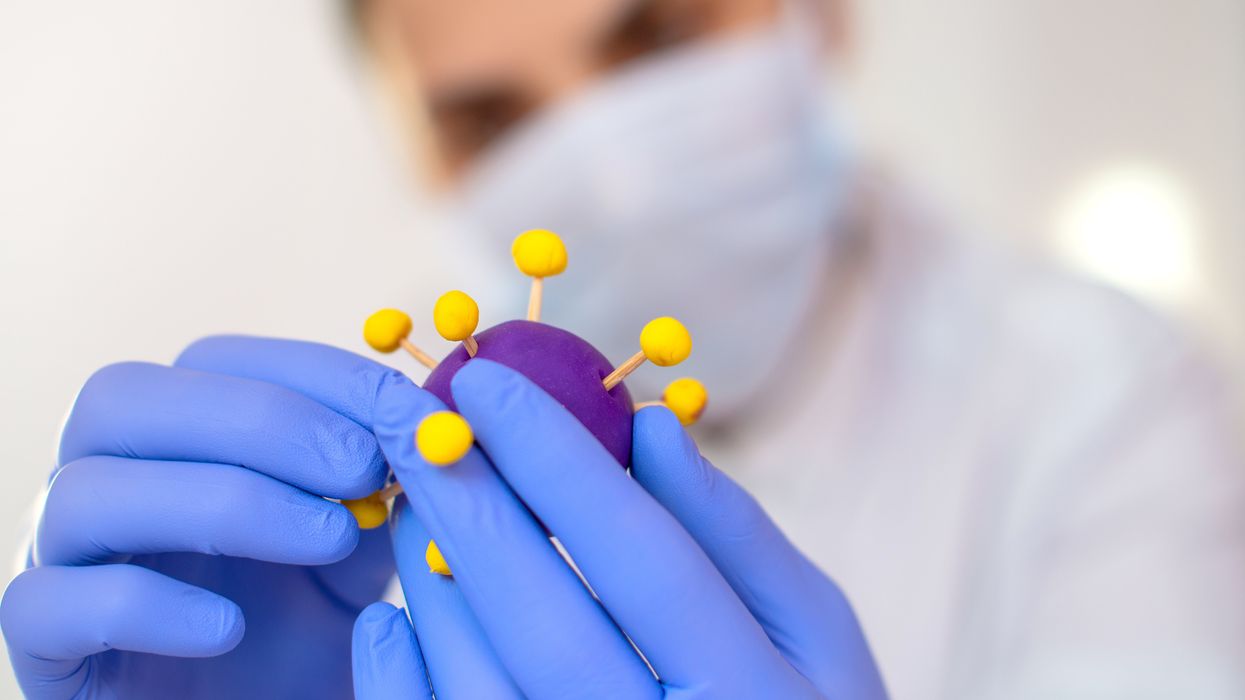
Gain-of-function research can make pathogens more infectious and deadly. It also enables scientists to prepare remedies in advance.
Scientists have long argued that gain-of-function research, which can make viruses and other infectious agents more contagious or more deadly, was necessary to develop therapies and vaccines to counter the pathogens in case they were used for biological warfare. As the SARS-CoV-2 origins are being investigated, one prominent theory suggests it had leaked from a biolab that conducted gain-of-function research, causing a global pandemic that claimed nearly 6.9 million lives. Now some question the wisdom of engaging in this type of research, stating that the risks may far outweigh the benefits.
“Gain-of-function research means genetically changing a genome in a way that might enhance the biological function of its genes, such as its transmissibility or the range of hosts it can infect,” says George Church, professor of genetics at Harvard Medical School. This can occur through direct genetic manipulation as well as by encouraging mutations while growing successive generations of micro-organism in culture. “Some of these changes may impact pathogenesis in a way that is hard to anticipate in advance,” Church says.
In the wake of the global pandemic, the pros and cons of gain-of-function research are being fiercely debated. Some scientists say this type of research is vital for preventing future pandemics or for preparing for bioweapon attacks. Others consider it another disaster waiting to happen. The Government Accounting Office issued a report charging that a framework developed by the U.S. Department of Health & Human Services (HHS) provided inadequate oversight of this potentially deadly research. There’s a movement to stop it altogether. In January, the Viral Gain-of-Function Research Moratorium Act (S. 81) was introduced into the Senate to cease awarding federal research funding to institutions doing gain-of-function studies.
While testifying before the House COVID Origins Select Committee on March 8th, Robert Redfield, former director of the U.S. Centers for Disease Control and Prevention, said that COVID-19 may have resulted from an accidental lab leak involving gain-of-function research. Redfield said his conclusion is based upon the “rapid and high infectivity for human-to-human transmission, which then predicts the rapid evolution of new variants.”
“It is a very, very, very small subset of life science research that could potentially generate a potential pandemic pathogen,” said Gerald Parker, associate dean for Global One Health at Texas A&M University.
“In my opinion,” Redfield continues, “the COVID-19 pandemic presents a case study on the potential dangers of such research. While many believe that gain-of-function research is critical to get ahead of viruses by developing vaccines, in this case, I believe that was the exact opposite.” Consequently, Redfield called for a moratorium on gain-of-function research until there is consensus about the value of such risky science.
What constitutes risky?
The Federal Select Agent Program lists 68 specific infectious agents as risky because they are either very contagious or very deadly. In order to work with these 68 agents, scientists must register with the federal government. Meanwhile, research on deadly pathogens that aren’t easily transmitted, or pathogens that are quite contagious but not deadly, can be conducted without such oversight. “If you’re not working with select agents, you’re not required to register the research with the federal government,” says Gerald Parker, associate dean for Global One Health at Texas A&M University. But the 68-item list may not have everything that could possibly become dangerous or be engineered to be dangerous, thus escaping the government’s scrutiny—an issue that new regulations aim to address.
In January 2017, the White House Office of Science and Technology Policy (OSTP) issued additional guidance. It required federal departments and agencies to follow a series of steps when reviewing proposed research that could create, transfer, or use potential pandemic pathogens resulting from the enhancement of a pathogen’s transmissibility or virulence in humans.
In defining risky pathogens, OSTP included viruses that were likely to be highly transmissible and highly virulent, and thus very deadly. The Proposed Biosecurity Oversight Framework for the Future of Science, outlined in 2023, broadened the scope to require federal review of research “that is reasonably anticipated to enhance the transmissibility and/or virulence of any pathogen” likely to pose a threat to public health, health systems or national security. Those types of experiments also include the pathogens’ ability to evade vaccines or therapeutics, or diagnostic detection.
However, Parker says that dangers of generating a pandemic-level germ are tiny. “It is a very, very, very small subset of life science research that could potentially generate a potential pandemic pathogen.” Since gain-of-function guidelines were first issued in 2017, only three such research projects have met those requirements for HHS review. They aimed to study influenza and bird flu. Only two of those projects were funded, according to the NIH Office of Science Policy. For context, NIH funded approximately 11,000 of the 54,000 grant applications it received in 2022.
Guidelines governing gain-of-function research are being strengthened, but Church points out they aren’t ideal yet. “They need to be much clearer about penalties and avoiding positive uses before they would be enforceable.”
What do we gain from gain-of-function research?
The most commonly cited reason to conduct gain-of-function research is for biodefense—the government’s ability to deal with organisms that may pose threats to public health.
In the era of mRNA vaccines, the advance preparedness argument may be even less relevant.
“The need to work with potentially dangerous viruses is central to our preparedness,” Parker says. “It’s essential that we know and understand the basic biology, microbiology, etc. of some of these dangerous pathogens.” That includes increasing our knowledge of the molecular mechanisms by which a virus could become a sustained threat to humans. “Knowing that could help us detect [risks] earlier,” Parker says—and could make it possible to have medical countermeasures, like vaccines and therapeutics, ready.
Most vaccines, however, aren’t affected by this type of research. Essentially, scientists hope they will never need to use it. Moreover, Paul Mango, HSS former deputy chief of staff for policy, and author of the 2022 book Warp Speed, says he believes that in the era of mRNA vaccines, the advance preparedness argument may be even less relevant. “That’s because these vaccines can be developed and produced in less than 12 months, unlike traditional vaccines that require years of development,” he says.
Can better oversight guarantee safety?
Another situation, which Parker calls unnecessarily dangerous, is when regulatory bodies cannot verify that the appropriate biosafety and biosecurity controls are in place.
Gain-of-function studies, Parker points out, are conducted at the basic research level, and they’re performed in high-containment labs. “As long as all the processes, procedures and protocols are followed and there’s appropriate oversight at the institutional and scientific level, it can be conducted safely.”
Globally, there are 69 Biosafety Level 4 (BSL4) labs operating, under construction or being planned, according to recent research from King’s College London and George Mason University for Global BioLabs. Eleven of these 18 high-containment facilities that are planned or under construction are in Asia. Overall, three-quarters of the BSL4 labs are in cities, increasing public health risks if leaks occur.
Researchers say they are confident in the oversight system for BSL4 labs within the U.S. They are less confident in international labs. Global BioLabs’ report concurs. It gives the highest scores for biosafety to industrialized nations, led by France, Australia, Canada, the U.S. and Japan, and the lowest scores to Saudi Arabia, India and some developing African nations. Scores for biosecurity followed similar patterns.
“There are no harmonized international biosafety and biosecurity standards,” Parker notes. That issue has been discussed for at least a decade. Now, in the wake of SARS and the COVID-19 pandemic, scientists and regulators are likely to push for unified oversight standards. “It’s time we got serious about international harmonization of biosafety and biosecurity standards and guidelines,” Parker says. New guidelines are being worked on. The National Science Advisory Board for Biosecurity (NSABB) outlined its proposed recommendations in the document titled Proposed Biosecurity Oversight Framework for the Future of Science.
The debates about whether gain-of-function research is useful or poses unnecessary risks to humanity are likely to rage on for a while. The public too has a voice in this debate and should weigh in by communicating with their representatives in government, or by partaking in educational forums or initiatives offered by universities and other institutions. In the meantime, scientists should focus on improving the research regulations, Parker notes. “We need to continue to look for lessons learned and for gaps in our oversight system,” he says. “That’s what we need to do right now.”
A new type of cancer therapy is shrinking deadly brain tumors with just one treatment
MRI scans after a new kind of immunotherapy for brain cancer show remarkable progress in one patient just days after the first treatment.
Few cancers are deadlier than glioblastomas—aggressive and lethal tumors that originate in the brain or spinal cord. Five years after diagnosis, less than five percent of glioblastoma patients are still alive—and more often, glioblastoma patients live just 14 months on average after receiving a diagnosis.
But an ongoing clinical trial at Mass General Cancer Center is giving new hope to glioblastoma patients and their families. The trial, called INCIPIENT, is meant to evaluate the effects of a special type of immune cell, called CAR-T cells, on patients with recurrent glioblastoma.
How CAR-T cell therapy works
CAR-T cell therapy is a type of cancer treatment called immunotherapy, where doctors modify a patient’s own immune system specifically to find and destroy cancer cells. In CAR-T cell therapy, doctors extract the patient’s T-cells, which are immune system cells that help fight off disease—particularly cancer. These T-cells are harvested from the patient and then genetically modified in a lab to produce proteins on their surface called chimeric antigen receptors (thus becoming CAR-T cells), which makes them able to bind to a specific protein on the patient’s cancer cells. Once modified, these CAR-T cells are grown in the lab for several weeks so that they can multiply into an army of millions. When enough cells have been grown, these super-charged T-cells are infused back into the patient where they can then seek out cancer cells, bind to them, and destroy them. CAR-T cell therapies have been approved by the US Food and Drug Administration (FDA) to treat certain types of lymphomas and leukemias, as well as multiple myeloma, but haven’t been approved to treat glioblastomas—yet.
CAR-T cell therapies don’t always work against solid tumors, such as glioblastomas. Because solid tumors contain different kinds of cancer cells, some cells can evade the immune system’s detection even after CAR-T cell therapy, according to a press release from Massachusetts General Hospital. For the INCIPIENT trial, researchers modified the CAR-T cells even further in hopes of making them more effective against solid tumors. These second-generation CAR-T cells (called CARv3-TEAM-E T cells) contain special antibodies that attack EFGR, a protein expressed in the majority of glioblastoma tumors. Unlike other CAR-T cell therapies, these particular CAR-T cells were designed to be directly injected into the patient’s brain.
The INCIPIENT trial results
The INCIPIENT trial involved three patients who were enrolled in the study between March and July 2023. All three patients—a 72-year-old man, a 74-year-old man, and a 57-year-old woman—were treated with chemo and radiation and enrolled in the trial with CAR-T cells after their glioblastoma tumors came back.
The results, which were published earlier this year in the New England Journal of Medicine (NEJM), were called “rapid” and “dramatic” by doctors involved in the trial. After just a single infusion of the CAR-T cells, each patient experienced a significant reduction in their tumor sizes. Just two days after receiving the infusion, the glioblastoma tumor of the 72-year-old man decreased by nearly twenty percent. Just two months later the tumor had shrunk by an astonishing 60 percent, and the change was maintained for more than six months. The most dramatic result was in the 57-year-old female patient, whose tumor shrank nearly completely after just one infusion of the CAR-T cells.
The results of the INCIPIENT trial were unexpected and astonishing—but unfortunately, they were also temporary. For all three patients, the tumors eventually began to grow back regardless of the CAR-T cell infusions. According to the press release from MGH, the medical team is now considering treating each patient with multiple infusions or prefacing each treatment with chemotherapy to prolong the response.
While there is still “more to do,” says co-author of the study neuro-oncologist Dr. Elizabeth Gerstner, the results are still promising. If nothing else, these second-generation CAR-T cell infusions may someday be able to give patients more time than traditional treatments would allow.
“These results are exciting but they are also just the beginning,” says Dr. Marcela Maus, a doctor and professor of medicine at Mass General who was involved in the clinical trial. “They tell us that we are on the right track in pursuing a therapy that has the potential to change the outlook for this intractable disease.”
A recent study in The Lancet Oncology showed that AI found 20 percent more cancers on mammogram screens than radiologists alone.
Since the early 2000s, AI systems have eliminated more than 1.7 million jobs, and that number will only increase as AI improves. Some research estimates that by 2025, AI will eliminate more than 85 million jobs.
But for all the talk about job security, AI is also proving to be a powerful tool in healthcare—specifically, cancer detection. One recently published study has shown that, remarkably, artificial intelligence was able to detect 20 percent more cancers in imaging scans than radiologists alone.
Published in The Lancet Oncology, the study analyzed the scans of 80,000 Swedish women with a moderate hereditary risk of breast cancer who had undergone a mammogram between April 2021 and July 2022. Half of these scans were read by AI and then a radiologist to double-check the findings. The second group of scans was read by two researchers without the help of AI. (Currently, the standard of care across Europe is to have two radiologists analyze a scan before diagnosing a patient with breast cancer.)
The study showed that the AI group detected cancer in 6 out of every 1,000 scans, while the radiologists detected cancer in 5 per 1,000 scans. In other words, AI found 20 percent more cancers than the highly-trained radiologists.

But even though the AI was better able to pinpoint cancer on an image, it doesn’t mean radiologists will soon be out of a job. Dr. Laura Heacock, a breast radiologist at NYU, said in an interview with CNN that radiologists do much more than simply screening mammograms, and that even well-trained technology can make errors. “These tools work best when paired with highly-trained radiologists who make the final call on your mammogram. Think of it as a tool like a stethoscope for a cardiologist.”
AI is still an emerging technology, but more and more doctors are using them to detect different cancers. For example, researchers at MIT have developed a program called MIRAI, which looks at patterns in patient mammograms across a series of scans and uses an algorithm to model a patient's risk of developing breast cancer over time. The program was "trained" with more than 200,000 breast imaging scans from Massachusetts General Hospital and has been tested on over 100,000 women in different hospitals across the world. According to MIT, MIRAI "has been shown to be more accurate in predicting the risk for developing breast cancer in the short term (over a 3-year period) compared to traditional tools." It has also been able to detect breast cancer up to five years before a patient receives a diagnosis.
The challenges for cancer-detecting AI tools now is not just accuracy. AI tools are also being challenged to perform consistently well across different ages, races, and breast density profiles, particularly given the increased risks that different women face. For example, Black women are 42 percent more likely than white women to die from breast cancer, despite having nearly the same rates of breast cancer as white women. Recently, an FDA-approved AI device for screening breast cancer has come under fire for wrongly detecting cancer in Black patients significantly more often than white patients.
As AI technology improves, radiologists will be able to accurately scan a more diverse set of patients at a larger volume than ever before, potentially saving more lives than ever.

