Will religious people reject organ transplants from pigs?
Scientists are getting better at transplanting pig organs into humans. But religious followers of prohibitions on consuming pork may reject these organs, even if their bodies could accept them.
The first successful recipient of a human heart transplant lived 18 days. The first artificial heart recipient lived just over 100.
Their brief post-transplant lives paved the way toward vastly greater successes. Former Vice President Dick Cheney relied on an artificial heart for nearly two years before receiving a human heart transplant. It still beats in his chest more than a decade later.
Organ transplantation recently reached its next phase with David Bennett. He survived for two months after becoming the first recipient of a pig’s heart genetically modified to function in a human body in February. Known as a xenotransplant, the procedure could pave the way for greatly expanding the use of transplanted vital organs to extend human lives.
Clinical trials would have to be held in the U.S. before xenotransplants become widespread; Bennett’s surgery was authorized under a special Food and Drug Administration program that addresses patients with life-threatening medical conditions.
German researchers plan to perform eight pig-to-human heart transplants as part of a clinical trial beginning in 2024. According to an email sent to Leaps.org by three scholars working on the German project, these procedures will focus on one of the reasons David Bennett did not survive longer: A porcine infection from his new heart.
The transplant team will conduct more sensitive testing of the donor organs, “which in all likelihood will be able to detect even low levels of virus in the xenograft,” note the scientists, Katharina Ebner, Jochen Ostheimer and Jochen Sautermeister. They are confident that the risk of infection with a porcine virus in the future will be significantly lower.
Moreover, hearts are not the only genetically modified organs that are being xenotransplanted. A team of surgeons at the University of Alabama at Birmingham successfully transplanted genetically modified pig kidneys into a brain-dead human recipient in September. The kidneys functioned normally for more than three days before the experiment ended. The UAB team is now moving forward with clinical trials focusing on transplanting pig kidneys into human patients.
Some experts believe the momentum for xenotransplantation is building, particularly given the recent successes. “I think there is a strong likelihood this will go mainstream,” says Brendan Parent of NYU Langone Health.
Douglas Anderson, a surgeon who is part of that kidney xenotransplant team, observes that, “organ shortages have been the major issue facing transplantation since its inception” and that xenotransplantation is a potential solution to that quandary. “It can’t be understated the number of people waiting for a kidney on dialysis, which has a significant mortality rate,” he says. According to the advocacy group Donate Life America, more than 100,000 people in the U.S. alone are waiting for a donated organ, and 85 percent of them need a kidney.
Other experts believe the momentum for xenotransplantation is building, particularly given the recent successes. “I think there is a strong likelihood this will go mainstream,” says Brendan Parent, director of transplant ethics and policy at NYU Langone Health, a New York City-based hospital system. Like the UAB team, surgeons at NYU Langone have had success coaxing modified pig kidneys to work in deceased humans.
“There is a genuinely good chance that within a generation, (xenotransplantation) might become very common in reasonably wealthy countries,” says Michael Reiss, professor of science education at University College in London. In addition to his academic position, Reiss sits on the Nuffield Council on Bioethics, a nonprofit that is one of Britain’s most prominent watchdogs regarding medical and scientific issues. Reiss is also an Anglican priest and has studied xenotransplantation from both a scientific and religious point of view.
Moreover, genetic modifications could one day lead to organs being specifically optimized for their recipients. That could ensure issues like donor rejection and the calculated risk of artificially suppressing recipient immune systems become concerns of the past.
Major bioethical, religious concerns
Despite the promise of xenotransplantation, numerous bioethical issues swirl around the procedure. They could be magnified if xenotransplantation evolves from one-off experiments to a routine medical procedure.
One of the biggest is the millennia-long prohibitions Islam and Judaism have had regarding the consumption of pork. Will followers of these religions assume such rules extend to those taboo materials being inserted into a human body?
“Initially, one’s instinctual reaction is that, oh, crumbs! – how are Jews and Muslims going to react to that?” Reiss says. But in a world where science and secularism are accepted on an everyday basis, he notes it is not a significant issue. Reiss points out that valves from pig hearts have been used in human patients for decades without any issues. He adds that both Islam and Judaism waive religious dietary restrictions if a human life is at risk.
“While nobody's saying an individual patient is to be forced to have these, the very high proportion of people who identify as Jews or Muslims when given this option are content with it,” he says.
Concurring with Reiss is Michael Gusamano, professor of health policy at Lehigh University and director of its Center for Ethics. He is currently performing research on the ethics of xenotransplantation for the National Institutes of Health.
“Leaders from all major religions have commented on this and have indicated that this is not inconsistent with religious doctrine,” Gusamano says in written remarks to Leaps.org. “Having said that, it is plausible to believe that some people will assume that this is inconsistent with the teaching of their religion and may object to…receiving a xenotransplant as part of routine medical care.”
A history of clashes
Despite those assurances, science has long clashed with theology. Although Galileo proved the planets revolved around the sun, the Catholic Church found him guilty of heresy and rewarded his discovery with house arrest for the last decade of his life. A revolt occurred in mid-19th century India after native-born soldiers believed the ammunition supplied by their British occupiers had been lubricated with pork and beef tallow. Given they had to use their mouths to tear open ammunition pouches, this violated both the tenets of Islam and Hinduism. And one of the conspiracy theories hatched as a result of COVID-19 was that the vaccines developed to fight the disease were the “mark of the beast” – a sign of impending Armageddon under evangelical Christian theology.
The German xenotransplant research team has encountered such potential concerns when the procedure is regarded through a religious lens. “The pastors in our research suspected that many recipients might feel disgust and revulsion,” they write. “Even beyond these special religious reservations, cultural scripts about pigs as inferior living beings are also generally widespread and effective in the western world, so that here too possible disgust reactions cannot be ruled out.”
The German researchers add that “Jewish and Muslim hospital pastoral workers believe possible considerable problems in this respect, which must be dealt with psychosocially, religiously, and pastorally prior to a possible transplantation in order to strengthen the acceptance of the received organ by the patients and their relatives.”
Parent, the director at NYU Langone, shares a concern that xenotransplantation could move “too fast,” although much of his worry is focused on zoonotic disease transmission – pig viruses jumping into humans as a result of such procedures.
Another ethical issue
Moreover, the way pigs and other animals are raised for transplants could pose future ethical dilemmas.
Reiss notes that pigs raised for medical procedures have to be grown and kept in what are known as a designated pathogen-free facility, or DPF. Such facilities are kept painstakingly antiseptic so as to minimize the risk of zoonotic transmissions. But given pigs are fond of outdoor activities such as wallowing in mud and sleeping on hay, they lead “stunningly boring lives” that they probably do not enjoy, Reiss observes.
Ethical concerns with using pigs may push transplantation medicine into its next logical phase: Growing functional organs for transplant in a laboratory setting.
“There’s no doubt that these research pigs have gotten much better veterinary care, et cetera, (compared to farmed pigs). But it’s not a great life,” Reiss says. “And although it hasn’t so far dominated the discussion, I think as the years go by, rather as we’ve seen with the use of apes and now monkeys in medical research, more and more theologians will get uncomfortable about us just assuming we can do this with…pigs.”
The German research team raises the same concerns, but has taken a fairly sanguine view on the topic. “The impairments of the species-typical behavior will certainly provoke criticism and perhaps also public protest. But the number of animals affected is very small in relation to slaughter cattle,” the German researchers note. “Moreover, the conditions there and also in several animal experiments are far worse.”
Observers say that may push transplantation medicine into its next logical phase: Growing functional organs for transplant in a laboratory setting. Anderson, the UAB transplant surgeon, believes such an accomplishment remains decades away.
But other experts believe there is a moral imperative that xenotransplantation remain a temporary solution. “I think we have a duty to go in that direction,” Parent says. “We have to go that way, with the xenotransplantation process (as) a steppingstone and research path that will be useful for bioengineered organs.”
Thanks to safety cautions from the COVID-19 pandemic, a strain of influenza has been completely eliminated.
If you were one of the millions who masked up, washed your hands thoroughly and socially distanced, pat yourself on the back—you may have helped change the course of human history.
Scientists say that thanks to these safety precautions, which were introduced in early 2020 as a way to stop transmission of the novel COVID-19 virus, a strain of influenza has been completely eliminated. This marks the first time in human history that a virus has been wiped out through non-pharmaceutical interventions, such as vaccines.
The flu shot, explained
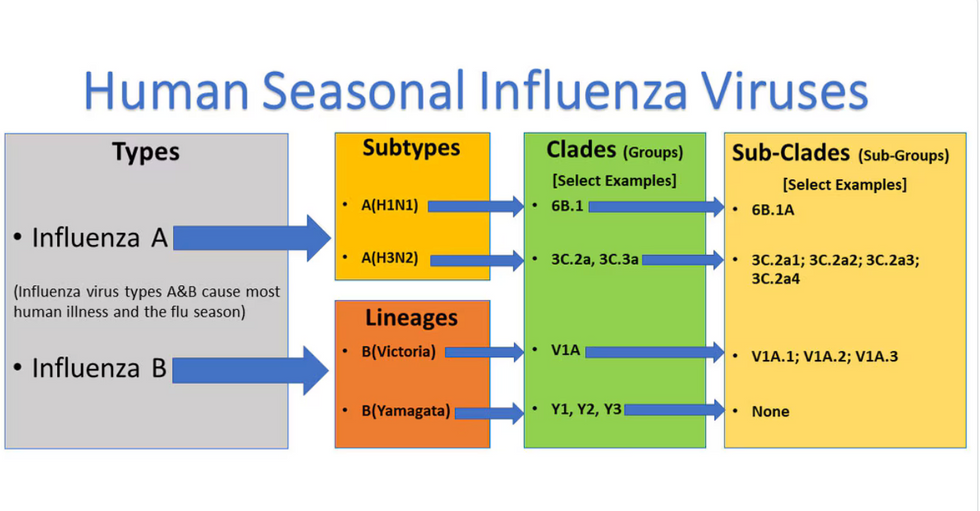
Influenza viruses type A and B are responsible for the majority of human illnesses and the flu season.
Centers for Disease Control
For more than a decade, flu shots have protected against two types of the influenza virus–type A and type B. While there are four different strains of influenza in existence (A, B, C, and D), only strains A, B, and C are capable of infecting humans, and only A and B cause pandemics. In other words, if you catch the flu during flu season, you’re most likely sick with flu type A or B.
Flu vaccines contain inactivated—or dead—influenza virus. These inactivated viruses can’t cause sickness in humans, but when administered as part of a vaccine, they teach a person’s immune system to recognize and kill those viruses when they’re encountered in the wild.
Each spring, a panel of experts gives a recommendation to the US Food and Drug Administration on which strains of each flu type to include in that year’s flu vaccine, depending on what surveillance data says is circulating and what they believe is likely to cause the most illness during the upcoming flu season. For the past decade, Americans have had access to vaccines that provide protection against two strains of influenza A and two lineages of influenza B, known as the Victoria lineage and the Yamagata lineage. But this year, the seasonal flu shot won’t include the Yamagata strain, because the Yamagata strain is no longer circulating among humans.
How Yamagata Disappeared

Flu surveillance data from the Global Initiative on Sharing All Influenza Data (GISAID) shows that the Yamagata lineage of flu type B has not been sequenced since April 2020.
Nature
Experts believe that the Yamagata lineage had already been in decline before the pandemic hit, likely because the strain was naturally less capable of infecting large numbers of people compared to the other strains. When the COVID-19 pandemic hit, the resulting safety precautions such as social distancing, isolating, hand-washing, and masking were enough to drive the virus into extinction completely.
Because the strain hasn’t been circulating since 2020, the FDA elected to remove the Yamagata strain from the seasonal flu vaccine. This will mark the first time since 2012 that the annual flu shot will be trivalent (three-component) rather than quadrivalent (four-component).
Should I still get the flu shot?
The flu shot will protect against fewer strains this year—but that doesn’t mean we should skip it. Influenza places a substantial health burden on the United States every year, responsible for hundreds of thousands of hospitalizations and tens of thousands of deaths. The flu shot has been shown to prevent millions of illnesses each year (more than six million during the 2022-2023 season). And while it’s still possible to catch the flu after getting the flu shot, studies show that people are far less likely to be hospitalized or die when they’re vaccinated.
Another unexpected benefit of dropping the Yamagata strain from the seasonal vaccine? This will possibly make production of the flu vaccine faster, and enable manufacturers to make more vaccines, helping countries who have a flu vaccine shortage and potentially saving millions more lives.
After his grandmother’s dementia diagnosis, one man invented a snack to keep her healthy and hydrated.
Founder Lewis Hornby and his grandmother Pat, sampling Jelly Drops—an edible gummy containing water and life-saving electrolytes.
On a visit to his grandmother’s nursing home in 2016, college student Lewis Hornby made a shocking discovery: Dehydration is a common (and dangerous) problem among seniors—especially those that are diagnosed with dementia.
Hornby’s grandmother, Pat, had always had difficulty keeping up her water intake as she got older, a common issue with seniors. As we age, our body composition changes, and we naturally hold less water than younger adults or children, so it’s easier to become dehydrated quickly if those fluids aren’t replenished. What’s more, our thirst signals diminish naturally as we age as well—meaning our body is not as good as it once was in letting us know that we need to rehydrate. This often creates a perfect storm that commonly leads to dehydration. In Pat’s case, her dehydration was so severe she nearly died.
When Lewis Hornby visited his grandmother at her nursing home afterward, he learned that dehydration especially affects people with dementia, as they often don’t feel thirst cues at all, or may not recognize how to use cups correctly. But while dementia patients often don’t remember to drink water, it seemed to Hornby that they had less problem remembering to eat, particularly candy.

Hornby wanted to create a solution for elderly people who struggled keeping their fluid intake up. He spent the next eighteen months researching and designing a solution and securing funding for his project. In 2019, Hornby won a sizable grant from the Alzheimer’s Society, a UK-based care and research charity for people with dementia and their caregivers. Together, through the charity’s Accelerator Program, they created a bite-sized, sugar-free, edible jelly drop that looked and tasted like candy. The candy, called Jelly Drops, contained 95% water and electrolytes—important minerals that are often lost during dehydration. The final product launched in 2020—and was an immediate success. The drops were able to provide extra hydration to the elderly, as well as help keep dementia patients safe, since dehydration commonly leads to confusion, hospitalization, and sometimes even death.
Not only did Jelly Drops quickly become a favorite snack among dementia patients in the UK, but they were able to provide an additional boost of hydration to hospital workers during the pandemic. In NHS coronavirus hospital wards, patients infected with the virus were regularly given Jelly Drops to keep their fluid levels normal—and staff members snacked on them as well, since long shifts and personal protective equipment (PPE) they were required to wear often left them feeling parched.
In April 2022, Jelly Drops launched in the United States. The company continues to donate 1% of its profits to help fund Alzheimer’s research.

