People With This Rare Disease Can Barely Eat Protein. Biotechnology May Change That.

The Brown family at the Grand Tetons (2019). Clockwise from left, Christine, Kevin, Keagan, Connor, and Kellen.
Imagine that the protein in bread, eggs, steak, even beans is not the foundation for a healthy diet, but a poison to your brain. That is the reality for people living with Phenylketonuria, or PKU. This cluster of rare genetic variations affects the ability to digest phenylalanine (Phe), one of the chemical building blocks of protein. The toxins can build up in the brain causing severe mental retardation.
Can a probiotic help digest the troublesome proteins before they can enter the bloodstream and travel to the brain? A Boston area biotech start up, Synlogic, believes it can. Their starting point is an E. coli bacterium that has been used as a probiotic for more than a century. The company then screened thousands of gene variants to identify ones that produced enzymes most efficient at slicing and dicing the target proteins and optimized them further through directed evolution. The results have been encouraging.
But Christine Brown knew none of this when the hospital called saying that standard newborn screening of her son Connor had come back positive for PKU. It was urgent that they visit a special metabolic clinic the next day, which was about a three-hour drive away.
“I was told not to go on the Internet,” Christine recalls, “So when somebody tells you not to go on the Internet, what do you do? Even back in 2005, right.” What she saw were the worst examples of retardation, which was a common outcome from PKU before newborn screening became routine. “We were just in complete shell shock, our whole world just kind of shattered and went into a tail spin.”
“I remember feeding him the night before our clinic visit and almost feeling like I was feeding him poison because I knew that breast milk must have protein in it,” she says.
“Some of my first memories are of asking, ‘Mommy, can I eat this? There were yes foods and no foods.'"
Over the next few days the dedicated staff of the metabolic clinic at the Waisman Center at the University of Wisconsin Madison began to walk she and husband Kevin back from that nightmare. They learned that a simple blood test to screen newborns had been developed in the early 1960s to detect PKU and that the condition could be managed with stringent food restrictions and vigilant monitoring of Phe levels.
Everything in Your Mouth Counts
PKU can be successfully managed with a severely restricted diet. That simple statement is factually true, but practically impossible to follow, as it requires slashing protein consumption by about 90 percent. To compensate for the missing protein, several times a day PKU patients take a medical formula – commonly referred to simply as formula – containing forms of proteins that are digestible to their bodies. Several manufacturers now add vitamins and minerals and offer a variety of formats and tastes to make it more consumer friendly, but that wasn't always the case.
“When I was a kid, it tasted horrible, was the consistency of house paint. I didn't think about it, I just drank it. I didn't like it but you get used to it after a while,” recalls Jeff Wolf, the twang of Appalachia still strong in his voice. Now age 50, he grew up in Ashland, Kentucky and was part of the first wave of persons with PKU who were identified at birth as newborn screening was rolled out across the US. He says the options of taste and consistency have improved tremendously over the years.
Some people with PKU are restricted to as little as 8 grams of protein a day from food. That's a handful of almonds or a single hard-boiled egg; a skimpy 4-ounce hamburger and slice of cheese adds up to half of their weekly protein ration. Anything above that daily allowance is more than the body can handle and toxic levels of Phe begin to accumulate in the brain.
“Some of my first memories are of asking, ‘Mommy, can I eat this? There were yes foods and no foods,’” recalls Les Clark. He has never eaten a hamburger, steak, or ribs, practically a sacrilege for someone raised in Stanton, a small town in northeastern Nebraska, a state where the number of cattle and hogs are several-fold those of people.
His grandmother learned how to make low protein bread, but it looked and tasted different. His mom struggled making birthday cakes. “I learned some bad words at a very young age” as mom struggled applying icing that would pull the cake apart or a slice would collapse into a heap of crumbs, Les recalls.

Les Clark with a birthday cake.
Courtesy Clark
Controlling the diet “is not so bad when you are a baby” because that's all you know, says Jerry Vockley, Director of the Center for Rare Disease Therapy at Children's Hospital of Pittsburgh. “But after a while, as you get older and you start tasting other things and you say, Well, gee, this stuff tastes way better than what you're giving me. What's the deal? It becomes harder to maintain the diet.”
First is the lure of forbidden foods as children venture into the community away from the watchful eyes of parents. The support system weakens further when they leave home for college or work. “Pizza was mighty tasty,” Wolf' says of his first slice.
Vockley estimates that about 90 percent of adults with PKU are off of treatment. Moving might mean finding a new metabolic clinic that treats PKU. A lapse in insurance coverage can be a factor. Finally there is plain fatigue from multiple daily dosing of barely tolerable formula, monitoring protein intake, and simply being different in terms of food restrictions. Most people want to fit in and not be defined by their medical condition.
Jeff Wolf was one of those who dropped out in his twenties and thirties. He stopped going to clinic, monitoring his Phe levels, and counting protein. But the earlier experience of living with PKU never completely left the back of his mind; he listened to his body whenever eating too much protein left him with the “fuzzy brain" of a protein hangover. About a decade ago he reconnected with a metabolic clinic, began taking formula and watching his protein intake. He still may go over his allotment for a single day but he tries to compensate on subsequent days so that his Phe levels come back into balance.

Jeff Wolf on a boat.
Courtesy Wolf
One of the trickiest parts of trying to manage phenylalanine intake is the artificial sweetener aspartame. The chemical is ubiquitous in diet and lite foods and drinks. Gum too, you don't even have to swallow to receive a toxic dose of Phe. Most PKUers say it is easier to simply avoid these products entirely rather than try to count their Phe content.
Treatments
Most rare diseases have no treatment. There are two drugs for PKU that provide some benefit to some portion of patients but those drugs often have their own burdens.
KUVAN® (sapropterin dihydrochloride) is a pill or powder that helps correct a protein folding error so that food proteins can be digested. It is approved for most types of PKU in adults and children one month and older, and often is used along with a protein-restricted diet.
“The problem is that it doesn't work for every [patient's genetic] mutation, and there are hundreds of mutations that have been identified with PKU. Two to three percent of patients will have a very dramatic response and if you're one of those small number of patients, it's great,” says Vockley. “If you have one of the other mutation, chances are pretty good you still are going to end up on a restricted diet.”
PALYNZIQ® (pegvaliase-pqpz) “has the potential to lower the Phe to normal levels, it's a real breakthrough in the field,” says Vockley. “But is a very hard drug to use. Most folks have to take either one or two 2ml injections a day of something that is basically a gel, and some individuals have to take three.”
Many PKUers have reactions at the site of the injection and some develop anaphylaxis, a severe potentially life-threatening allergic reaction that can happen within seconds and can occur at any time, even after long term use. Many patients using Palynziq carry an EpiPen, a self-injection devise containing a form of adrenaline that can reverse some of the symptoms of anaphylaxis.
Then there is the cost. With the Kuvan dosing for an adult, “you're talking between $100,000 and $200,000 a year. And Palynziq is three times that,” says Vockley. Insurance coverage through a private plan or a state program is essential. Some state programs provide generous coverage while others are skimpy. Most large insurance company plans cover the drugs, sometimes with significant copays, but companies that are self-insured are under no legal obligation to provide coverage.
Les Clark found that out the hard way when the company he worked for was sold. The new owner was self-insured and declined to continue covering his drugs. Almost immediately he was out of pocket an additional $1500 a month for formula, and that was with a substantial discount through the manufacturer's patient support program. He says, “If you don't have an insurance policy that will cover the formula, it's completely unaffordable.” He quickly began to look for a new job.
Hope
It's easy to see why PKUers are eager for advances that will make managing their condition more effect, easier, and perhaps more affordable. Synlogic's efforts have drawn their attention and raised hopes.
Just before Thanksgiving Jerry Vockley presented the latest data to a metabolism conference meeting in Australia. There were only 8 patients in this group of a phase 2 trial using the original version of the company's lead E. coli product, SYNB1618, but they were intensely studied. Each was given the probiotic and then a challenge meal. Vockley saw a 40% reduction in Phe absorption and later a 20% reduction in mean fasting Phe levels in the blood. The product was easy to use and tolerate.
The company also presented early results for SYNB1934, a follow on version that further genetically tweaked the E. coli to roughly double the capacity to chop up the target proteins. Synlogic is recruiting patients for studies to determine the best dosing, which they are planning for next year.
“It's an exciting approach,” says Lex Cowsert, Director of Research Development at the National PKU Alliance, a nonprofit that supports the patient, family, and research communities involved with PKU. “Every patient is different, every patient has a different tolerance for the type of therapy that they are willing to pursue,” and if it pans out, it will be a welcome addition, either alone or in combination with other approaches, to living with PKU.
Author's Note: Reporting this story was made possible by generous support from the National Press Foundation and the Fondation Ipsen. Thanks to the people who so generously shared their time and stories in speaking with me.
A vaccine for Lyme disease could be coming. But will patients accept it?
Hiking is one of Marci Flory's favorite activities, but her chronic Lyme disease sometimes renders her unable to walk. Biotech companies Pfizer and Valneva are testing a vaccine for Lyme disease in a Phase III clinical trial.
For more than two decades, Marci Flory, a 40-year-old emergency room nurse from Lawrence, Kan., has battled the recurring symptoms of chronic Lyme disease, an illness which she believes began after being bitten by a tick during her teenage years.
Over the years, Flory has been plagued by an array of mysterious ailments, ranging from fatigue to crippling pain in her eyes, joints and neck, and even postural tachycardia syndrome or PoTS, an abnormal increase in heart rate after sitting up or standing. Ten years ago, she began to experience the onset of neurological symptoms which ranged from brain fog to sudden headaches, and strange episodes of leg weakness which would leave her unable to walk.
“Initially doctors thought I had ALS, or less likely, multiple sclerosis,” she says. “But after repeated MRI scans for a year, they concluded I had a rare neurological condition called acute transverse myelitis.”
But Flory was not convinced. After ordering a variety of private blood tests, she discovered she was infected with a range of bacteria in the genus Borrelia that live in the guts of ticks, the infectious agents responsible for Lyme disease.
“It made sense,” she says. “Looking back, I was bitten in high school and misdiagnosed with mononucleosis. This was probably the start, and my immune system kept it under wraps for a while. The Lyme bacteria can burrow into every tissue in the body, go into cyst form and become dormant before reactivating.”
The reason why cases of Lyme disease are increasing is down to changing weather patterns, triggered by climate change, meaning that ticks are now found across a much wider geographic range than ever before.
When these species of bacteria are transmitted to humans, they can attack the nervous system, joints and even internal organs which can lead to serious health complications such as arthritis, meningitis and even heart failure. While Lyme disease can sometimes be successfully treated with antibiotics if spotted early on, not everyone responds to these drugs, and for patients who have developed chronic symptoms, there is no known cure. Flory says she knows of fellow Lyme disease patients who have spent hundreds of thousands of dollars seeking treatments.
Concerningly, statistics show that Lyme and other tick-borne diseases are on the rise. Recently released estimates based on health insurance records suggest that at least 476,000 Americans are diagnosed with Lyme disease every year, and many experts believe the true figure is far higher.
The reason why the numbers are growing is down to changing weather patterns, triggered by climate change, meaning that ticks are now found across a much wider geographic range than ever before. Health insurance data shows that cases of Lyme disease have increased fourfold in rural parts of the U.S. over the last 15 years, and 65 percent in urban regions.
As a result, many scientists who have studied Lyme disease feel that it is paramount to bring some form of protective vaccine to market which can be offered to people living in the most at-risk areas.
“Even the increased awareness for Lyme disease has not stopped the cases,” says Eva Sapi, professor of cellular and molecular biology at the University of New Haven. “Some of these patients are looking for answers for years, running from one doctor to another, so that is obviously a very big cost for our society at so many levels.”
Emerging vaccines – and backlash
But with the rising case numbers, interest has grown among the pharmaceutical industry and research communities. Vienna-based biotech Valneva have partnered with Pfizer to take their vaccine – a seasonal jab which offers protection against the six most common strains of Lyme disease in the northern hemisphere – into a Phase III clinical trial which began in August. Involving 6,000 participants in a number of U.S. states and northern Europe where Lyme disease is endemic, it could lead to a licensed vaccine by 2025, if it proves successful.
“For many years Lyme was considered a small market vaccine,” explains Monica E. Embers, assistant professor of parasitology at Tulane University in New Orleans. “Now we know that this is a much bigger problem, Pfizer has stepped up to invest in preventing this disease and other pharmaceutical companies may as well.”
Despite innovations, patient communities and their representatives remain ambivalent about the idea of a vaccine. Some of this skepticism dates back to the failed LYMErix vaccine which was developed in the late 1990s before being withdrawn from the market.
At the same time, scientists at Yale University are developing a messenger RNA vaccine which aims to train the immune system to respond to tick bites by exposing it to 19 proteins found in tick saliva. Whereas the Valneva vaccine targets the bacteria within ticks, the Yale vaccine attempts to provoke an instant and aggressive immune response at the site of the bite. This causes the tick to fall off and limits the potential for transmitting dangerous infections.
But despite these innovations, patient communities and their representatives remain ambivalent about the idea of a vaccine. Some of this skepticism dates back to the failed LYMErix vaccine which was developed in the late 1990s before being withdrawn from the market in 2002 after concerns were raised that it might induce autoimmune reactions in humans.
While this theory was ultimately disproved, the lingering stigma attached to LYMErix meant that most vaccine manufacturers chose to stay away from the disease for many years, something which Gregory Poland, head of the Mayo Clinic’s Vaccine Research Group in Minnesota, describes as a tragedy.
“Since 2002, we have not had a human Lyme vaccine in the U.S. despite the increasing number of cases,” says Poland. “Pretty much everyone in the field thinks they’re ten times higher than the official numbers, so you’re probably talking at least 400,000 each year. It’s an incredible burden but because of concerns about anti-vax protestors, until very recently, no manufacturer has wanted to touch this.”
Such was the backlash surrounding the failed LYMErix program that scientists have even explored the most creative of workarounds for protecting people in tick-populated regions, without needing to actually vaccinate them. One research program at the University of Tennessee came up with the idea of leaving food pellets containing a vaccine in woodland areas with the idea that rodents would eat the pellets, and the vaccine would then kill Borrelia bacteria within any ticks which subsequently fed on the animals.
Even the Pfizer-Valneva vaccine has been cautiously designed to try and allay any lingering concerns, two decades after LYMErix. “The concept is the same as the original LYMErix vaccine, but it has been made safer by removing regions that had the potential to induce autoimmunity,” says Embers. “There will always be individuals who oppose vaccines, Lyme or otherwise, but it will be a tremendous boost to public health to have the option.”
Vaccine alternatives
Researchers are also considering alternative immunization approaches in case sufficiently large numbers of people choose to reject any Lyme vaccine which gets approved. Researchers at UMass Chan Medical School have developed an artificially generated antibody, administered via an annual injection, which is capable of killing Borrelia bacteria in the guts of ticks before they can get into the human host.
So far animal studies have shown it to be 100 percent effective, while the scientists have completed a Phase I trial in which they tested it for safety on 48 volunteers in Nebraska. Because this approach provides the antibody directly, rather than triggering the human immune system to produce the antibody like a vaccine would, Embers predicts that it could be a viable alternative for the vaccine hesitant as well as providing an option for immunocompromised individuals who cannot produce enough of their own antibodies.
At the same time, many patient groups still raise concerns over the fact that numerous diagnostic tests for Lyme disease have been reported to have a poor accuracy. Without this, they argue that it is difficult to prove whether vaccines or any other form of immunization actually work. “If the disease is not understood enough to create a more accurate test and a universally accepted treatment protocol, particularly for those who weren’t treated promptly, how can we be sure about the efficacy of a vaccine?” says Natasha Metcalf, co-founder of the organization Lyme Disease UK.
Flory points out that there are so many different types of Borrelia bacteria which cause Lyme disease, that the immunizations being developed may only stop a proportion of cases. In addition, she says that chronic Lyme patients often report a whole myriad of co-infections which remain poorly understood and are likely to also be involved in the disease process.
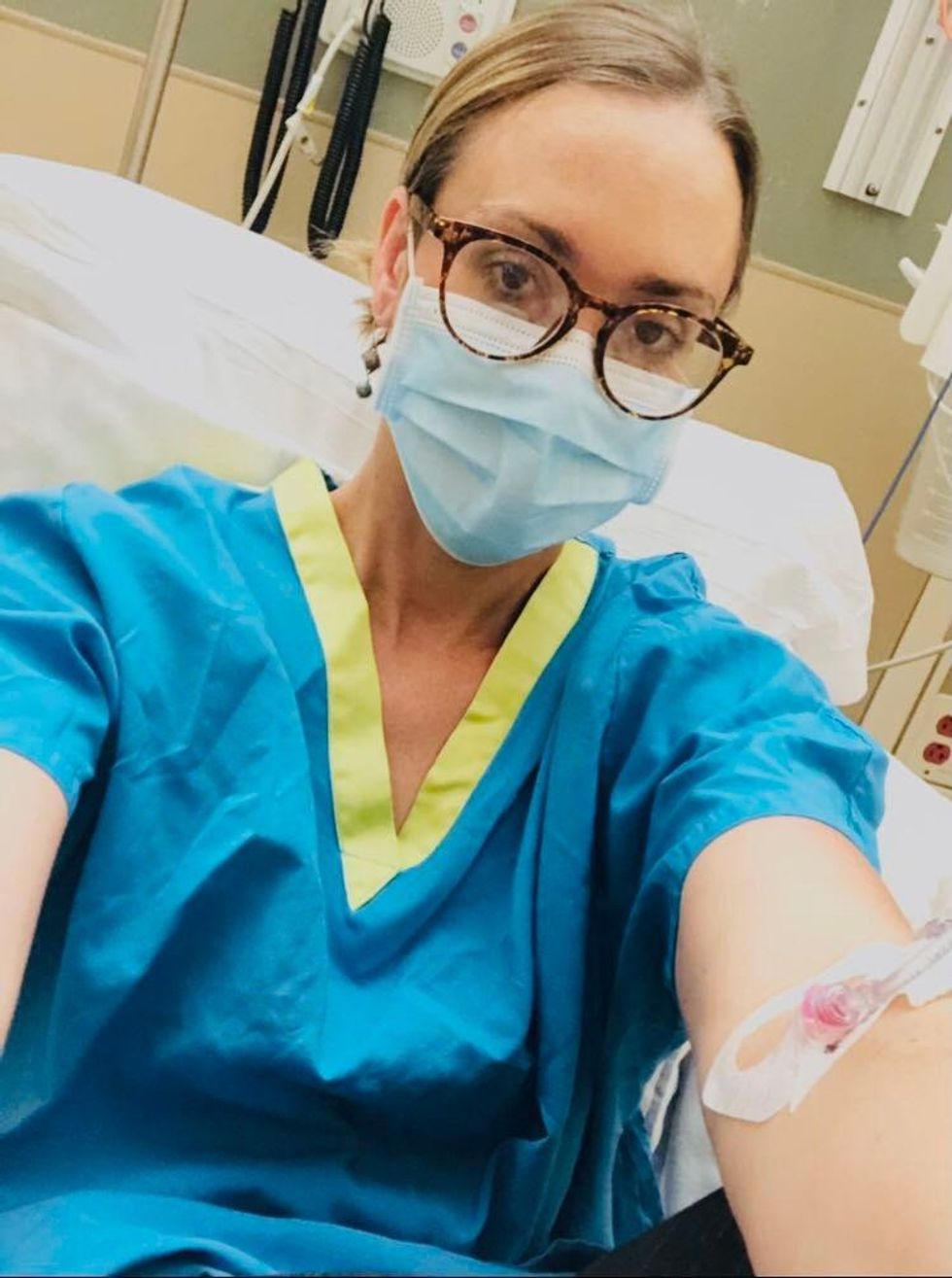
Marci Flory undergoes an infusion in an attempt to treat her Lyme disease symptoms.
Marci Flory
“I would love to see an effective Lyme vaccine but I have my reservations,” she says. “I am infected with four types of Borrelia bacteria, plus many co-infections – Babesia, Bartonella, Erlichiosis, Rickettsia, and Mycoplasma – all from a single Douglas County Kansas tick bite. Lyme never travels alone and the vaccine won’t protect against all the many strains of Borrelia and co-infections.”
Valneva CEO Thomas Lingelbach admits that the Pfizer-Valneva vaccine is not perfect, but predicts that it will still have significant impact if approved.
“We expect the vaccine to have 75 percent plus efficacy,” he says. “There is this legacy around the old Lyme vaccines, but the world is very, very different today. The number of clinical manifestations known to be caused by infection with Lyme Borreliosis has significantly increased, and the understanding around severity has certainly increased.”
Embers agrees that while it will still be important for doctors to monitor for other tick-borne infections which are not necessarily covered by the vaccine, having any clinically approved jab would still represent a major step forward in the fight against the disease.
“I think that any vaccine must be properly vetted, and these companies are performing extensive clinical trials to do just that,” she says. “Lyme is the most common tick-borne disease in the U.S. so the public health impact could be significant. However, clinicians and the general public must remain aware of all of the other tick-borne diseases such as Babesia and Anaplasma, and continue to screen for those when a tick bite is suspected.”
A mosquito under the microscope.
Two years, six million deaths and still counting, scientists are searching for answers to prevent another COVID-19-like tragedy from ever occurring again. And it’s a gargantuan task.
Our disturbed ecosystems are creating more favorable conditions for the spread of infectious disease. Global warming, deforestation, rising sea levels and flooding have contributed to a rise in mosquito-borne infections and longer tick seasons. Disease-carrying animals are in closer range to other species and humans as they migrate to escape the heat. Bats are thought to have carried the SARS-CoV-2 virus to Wuhan, either directly or through another host animal, but thousands of novel viruses are lurking within other wild creatures.
Understanding how climate change contributes to the spread of disease is critical in predicting and thwarting future calamities. But the problem is that predictive models aren’t yet where they need to be for forecasting with certainty beyond the next year, as we could for weather, for instance.
The association between climate and infectious disease is poorly understood, says Irina Tezaur, a computational scientist at Sandia National Laboratories. “Correlations have been observed but it’s not known if these correlations translate to causal relationships.”
To make accurate longer-term predictions, scientists need more empirical data, multiple datasets specific to locations and diseases, and the ability to calculate risks that depend on unpredictable nature and human behavior. Another obstacle is that climate scientists and epidemiologists are not collaborating effectively, so some researchers are calling for a multidisciplinary approach, a new field called Outbreak Science.
Climate scientists are far ahead of epidemiologists in gathering essential data.
Earth System Models—combining the interactions of atmosphere, ocean, land, ice and biosphere—have been in place for two decades to monitor the effects of global climate change. These models must be combined with epidemiological and human model research, areas that are easily skewed by unpredictable elements, from extreme weather events to public environmental policy shifts.
“There is never just one driver in tracking the impact of climate on infectious disease,” says Joacim Rocklöv, a professor at the Heidelberg Institute of Global Health & Heidelberg Interdisciplinary Centre for Scientific Computing in Germany. Rocklöv has studied how climate affects vector-borne diseases—those transmitted to humans by mosquitoes, ticks or fleas. “You need to disentangle the variables to find out how much difference climate makes to the outcome and how much is other factors.” Determinants from deforestation to population density to lack of healthcare access influence the spread of disease.
Even though climate change is not the primary driver of infectious disease today, it poses a major threat to public health in the future, says Rocklöv.
The promise of predictive modeling
“Models are simplifications of a system we’re trying to understand,” says Jeremy Hess, who directs the Center for Health and the Global Environment at University of Washington in Seattle. “They’re tools for learning that improve over time with new observations.”
Accurate predictions depend on high-quality, long-term observational data but models must start with assumptions. “It’s not possible to apply an evidence-based approach for the next 40 years,” says Rocklöv. “Using models to experiment and learn is the only way to figure out what climate means for infectious disease. We collect data and analyze what already happened. What we do today will not make a difference for several decades.”
To improve accuracy, scientists develop and draw on thousands of models to cover as many scenarios as possible. One model may capture the dynamics of disease transmission while another focuses on immunity data or ocean influences or seasonal components of a virus. Further, each model needs to be disease-specific and often location-specific to be useful.
“All models have biases so it’s important to use a suite of models,” Tezaur stresses.
The modeling scientist chooses the drivers of change and parameters based on the question explored. The drivers could be increased precipitation, poverty or mosquito prevalence, for instance. Later, the scientist may need to isolate the effect of one driver so that will require another model.
There have been some related successes, such as the latest models for mosquito-borne diseases like Dengue, Zika and malaria as well as those for flu and tick-borne diseases, says Hess.
Rocklöv was part of a research team that used test data from 2018 and 2019 to identify regions at risk for West Nile virus outbreaks. Using AI, scientists were able to forecast outbreaks of the virus for the entire transmission season in Europe. “In the end, we want data-driven models; that’s what AI can accomplish,” says Rocklöv. Other researchers are making an important headway in creating a framework to predict novel host–parasite interactions.
Modeling studies can run months, years or decades. “The scientist is working with layers of data. The challenge is how to transform and couple different models together on a planetary scale,” says Jeanne Fair, a scientist at Los Alamos National Laboratory, Biosecurity and Public Health, in New Mexico.
Disease forecasting will require a significant investment into the infrastructure needed to collect data about the environment, vectors, and hosts a tall spatial and temporal resolutions.
And it’s a constantly changing picture. A modeling study in an April 2022 issue of Nature predicted that thousands of animals will migrate to cooler locales as temperatures rise. This means that various species will come into closer contact with people and other mammals for the first time. This is likely to increase the risk of emerging infectious disease transmitted from animals to humans, especially in Africa and Asia.
Other things can happen too. Global warming could precipitate viral mutations or new infectious diseases that don’t respond to antimicrobial treatments. Insecticide-resistant mosquitoes could evolve. Weather-related food insecurity could increase malnutrition and weaken people’s immune systems. And the impact of an epidemic will be worse if it co-occurs during a heatwave, flood, or drought, says Hess.
The devil is in the climate variables
Solid predictions about the future of climate and disease are not possible with so many uncertainties. Difficult-to-measure drivers must be added to the empirical model mix, such as land and water use, ecosystem changes or the public’s willingness to accept a vaccine or practice social distancing. Nor is there any precedent for calculating the effect of climate changes that are accelerating at a faster speed than ever before.
The most critical climate variables thought to influence disease spread are temperature, precipitation, humidity, sunshine and wind, according to Tezaur’s research. And then there are variables within variables. Influenza scientists, for example, found that warm winters were predictors of the most severe flu seasons in the following year.
The human factor may be the most challenging determinant. To what degree will people curtail greenhouse gas emissions, if at all? The swift development of effective COVID-19 vaccines was a game-changer, but will scientists be able to repeat it during the next pandemic? Plus, no model could predict the amount of internet-fueled COVID-19 misinformation, Fair noted. To tackle this issue, infectious disease teams are looking to include more sociologists and political scientists in their modeling.
Addressing the gaps
Currently, researchers are focusing on the near future, predicting for next year, says Fair. “When it comes to long-term, that’s where we have the most work to do.” While scientists cannot foresee how political influences and misinformation spread will affect models, they are positioned to make headway in collecting and assessing new data streams that have never been merged.
Disease forecasting will require a significant investment into the infrastructure needed to collect data about the environment, vectors, and hosts at all spatial and temporal resolutions, Fair and her co-authors stated in their recent study. For example real-time data on mosquito prevalence and diversity in various settings and times is limited or non-existent. Fair also would like to see standards set in mosquito data collection in every country. “Standardizing across the US would be a huge accomplishment,” she says.

Understanding how climate change contributes to the spread of disease is critical for thwarting future calamities.
Jeanne Fair
Hess points to a dearth of data in local and regional datasets about how extreme weather events play out in different geographic locations. His research indicates that Africa and the Middle East experienced substantial climate shifts, for example, but are unrepresented in the evidentiary database, which limits conclusions. “A model for dengue may be good in Singapore but not necessarily in Port-au-Prince,” Hess explains. And, he adds, scientists need a way of evaluating models for how effective they are.
The hope, Rocklöv says, is that in the future we will have data-driven models rather than theoretical ones. In turn, sharper statistical analyses can inform resource allocation and intervention strategies to prevent outbreaks.
Most of all, experts emphasize that epidemiologists and climate scientists must stop working in silos. If scientists can successfully merge epidemiological data with climatic, biological, environmental, ecological and demographic data, they will make better predictions about complex disease patterns. Modeling “cross talk” and among disciplines and, in some cases, refusal to release data between countries is hindering discovery and advances.
It’s time for bold transdisciplinary action, says Hess. He points to initiatives that need funding in disease surveillance and control; developing and testing interventions; community education and social mobilization; decision-support analytics to predict when and where infections will emerge; advanced methodologies to improve modeling; training scientists in data management and integrated surveillance.
Establishing a new field of Outbreak Science to coordinate collaboration would accelerate progress. Investment in decision-support modeling tools for public health teams, policy makers, and other long-term planning stakeholders is imperative, too. We need to invest in programs that encourage people from climate modeling and epidemiology to work together in a cohesive fashion, says Tezaur. Joining forces is the only way to solve the formidable challenges ahead.
This article originally appeared in One Health/One Planet, a single-issue magazine that explores how climate change and other environmental shifts are increasing vulnerabilities to infectious diseases by land and by sea. The magazine probes how scientists are making progress with leaders in other fields toward solutions that embrace diverse perspectives and the interconnectedness of all lifeforms and the planet.

