Q&A with Holden Thorp: Finding Better Ways to Communicate Science

Leaps.org spoke with Holden Thorp, Editor-in-Chief of the prestigious Science family of journals, about ways to improve science journalism, the challenges faced by experts, the lab leak theory and much more.
This month, Leaps.org had a chance to speak with Holden Thorp, Editor-in-Chief of the Science family of journals. We talked about the best ways to communicate science to the public, mistakes by public health officials during the pandemic, the lab leak theory, and bipartisanship for funding science research.
Before becoming editor of the Science journals, Thorp spent six years as provost of Washington University in St. Louis, where he is Rita Levi-Montalcini Distinguished University Professor and holds appointments in both chemistry and medicine. He joined Washington University after spending three decades at the University of North Carolina at Chapel Hill, where he served as the UNC's 10th chancellor from 2008 through 2013.
A North Carolina native, Thorp earned a doctorate in chemistry in 1989 at the California Institute of Technology and completed postdoctoral work at Yale University. He is a fellow of the National Academy of Inventors and the American Association for the Advancement of Science.
Read his full bio here.
This conversation was lightly edited by Leaps.org for style and format.
Matt Fuchs: You're a musician. It seems like many scientists are also musicians. Is there a link between the scientist brain and the musician brain?
Holden Thorp: I think [the overlap is] relatively common. I'm still a gigging bass player. I play in the pits for lots of college musicals. I think that it takes a certain discipline and requires you to learn a lot of rules about how music works, and then you try to be creative within that. That's similar to scientific research. So it makes sense. Music is something I've been able to sustain my whole life. I wouldn't be the same person if I let it go. When you're playing, especially for a musical, where the music is challenging, you can't let your mind wander. It’s like meditation.
MF: I bet it helps to do something totally different from your editing responsibilities. Maybe lets the subconscious take care of tough problems at work.
HT: Right.
MF: There's probably never been a greater need for clear and persuasive science communicators. Do we need more cross specialty training? For example, journalism schools prioritizing science training, and science programs that require more time learning how to communicate effectively?
HT: I think we need both. One of the challenges we've had with COVID has been, especially at the beginning, a lot of reporters who didn’t normally cover scientific topics got put on COVID—and ended up creating things that had to be cleaned up later. This isn't the last science-oriented crisis we're going to have. We've already got climate change, and we'll have another health crisis for sure. So it’d be good for journalism to be a little better prepared next time.
"Scientists are human beings who have ego and bravado and every other human weakness."
But on the other side, maybe it's even more important that scientists learn how to communicate and how likely it is that their findings will be politicized, twisted and miscommunicated. Because one thing that surprised me is how shocked a lot of scientists have been. Every scientific issue that reaches into public policy becomes politicized: climate change, evolution, stem cells.
Once one side decided to be cautious about the pandemic, you could be certain the other side was going to decide not to do that. That's not the fault of science. That’s just life in a political world. That, I think, caught people off guard. They weren't prepared to shape and process their messages in a way that accounted for that—and for the way that social media has intensified all of this.
MF: Early in the pandemic, there was a lack of clarity about public health recommendations, as you’d expect with a virus we hadn’t seen before. Should public officials and scientists have more humility in similar situations in the future? Public officials need to be authoritative for their guidance to be followed, so how do they lead a crisis response while displaying humility about what we don't know?
HS: I think scientists are people who like to have the answer. It's very tempting and common for scientists to kind of oversell what we know right now, while not doing as much as we should to remind people that science is a self-correcting process. And when we fail to do that – after we’ve collected more data and need to change how we're interpreting it – the people who want to undermine us have a perfect weapon to use against us. It's challenging. But I agree that scientists are human beings who have ego and bravado and every other human weakness.
For example, we wanted to tell everybody that we thought the vaccines would provide sterilizing immunity against infection. Well, we don't have too many other respiratory viruses where that's the case. And so it was more likely that we were going to have what we ended up with, which is that the vaccines were excellent in preventing severe disease and death. It would have been great if they provided sterilizing immunity and abruptly ended the pandemic a year ago. But it was overly optimistic to think that was going to be the case in retrospect.
MF: Both in terms of how science is communicated and received by the public, do we need to reform institutions or start new ones to instill the truth-seeking values that are so important to appreciating science?
HS: There are a whole bunch of different factors. I think the biggest one is that the social media algorithms reward their owners financially when they figure out how to keep people in their silos. Users are more likely to click on things that they agree with—and that promote conflict with people that they disagree with. That has caused an acceleration in hostilities that attend some of these disagreements.
But I think the other problem is that we haven’t found a way to explain things to people when it’s not a crisis. So, for example, a strong indicator of whether someone who might otherwise be vaccine hesitant decided to get their vaccine is if they understood how vaccines worked before the pandemic started. Because if you're trying to tell somebody that they're wrong if they don't get a vaccine, at the same time you're trying to explain how it works, that's a lot of explaining to do in a short period of time.
Lack of open-mindedness is a problem, but another issue is that we need more understanding of these issues baked into the culture already. That's partly due the fact that there hasn't been more reform in K through 12 and college teaching. And that scientists are very comfortable talking to each other, and not very comfortable talking to people who don't know all of our jargon and have to be persuaded to spend time listening to and thinking about what we're trying to tell them.
"We're almost to the point where clinging to the lab leak idea is close to being a fringe idea that almost doesn't need to be included in stories."
MF: You mentioned silos. There have been some interesting attempts in recent years to do “both sides journalism,” where websites like AllSides put different views on high profile issues side-by-side. Some people believe that's how the news should be reported. Should we let people see and decide for themselves which side is the most convincing?
HS: It depends if we're talking about science. On scientific issues, when they start, there's legitimate disagreement about among scientists. But eventually, things go back and forth, and people compete with each other and work their way to the answer. At some point, we reach more of a consensus.
For example, on climate change, I think it's gotten to the point now where it's irresponsible, if you're writing a story about climate change, to run a quote from somebody somewhere who's still—probably because of their political views—clinging to the idea that anthropogenic global warming is somehow not damaging the planet.
On things that aren't decided yet, that makes sense to run both. It's more a question of judgment of the journalists. I don't think the solution to it is put stark versions of each side, side-by-side and let people choose. The whole point of journalism is to inform people. If there's a consensus on something, that's part of what you're supposed to be informing them about.
MF: What about reporting on perspectives about the lab leak theory at various times during the pandemic?
HS: We’re the outlet that ran the letter that really restarted the whole debate. A bunch of well-known scientists said we should consider the lab leak theory more carefully. And in the aftermath of that, a bunch of those scientists who signed that letter concluded that the lab leak was very, very unlikely. Interestingly, publishing that letter actually drove us to more of a consensus. I would say now, we're almost to the point where clinging to the lab leak idea is close to being a fringe idea that almost doesn't need to be included in stories. But I would say there's been a lot of evolution on that over the last year since we ran that letter.
MF: Let's talk about bipartisanship in Congress. Research funding for the National Institutes of Health was championed for years by influential Republicans who supported science to advance health breakthroughs. Is that changing? Maybe especially with Sen. Roy Blunt retiring? Has bipartisanship on science funding been eroded by political battles during COVID?
HS: I'm optimistic that that won't be the case. Republican Congresses have usually been good for science funding. And that's because (former Sen.) Arlen Specter and Roy Blunt are two of the political figures who have pushed for science funding over the last couple decades. With Blunt retiring, we don't know who's going to step in for him. That's an interesting question. I hope there will be Republican champions for science funding.
MF: Is there too much conservatism baked into how we research new therapies and bring them to people who are sick, bench-to-bedside? I'm thinking of the criticisms that NIH or the FDA are overly bureaucratic. Are you hopeful about ARPA-H, President Biden’s proposed new agency for health innovation?
HS: I think the challenge hasn't been cracked by the federal government. Maybe DARPA has done this outside of health science, but within health science, the federal government has had limited success at funding things that can be applied quickly, while having overwhelming success at funding basic research that eventually becomes important in applications. Can they do it the other way around? They’ll need people running ARPA-H who are application first. It’s ambitious. The way it was done in Operation Warp Speed is all the money was just given to the companies. If the hypothesis on ARPA-H is for the federal government to actually do what Moderna and BioNTech did for the vaccine, themselves, that's a radical idea. It's going to require thinking very differently than the way they think about dispersing grants for basic research.
MF: You’ve written a number of bold op-eds as editor of the Science journals. Are there any op-eds you're especially proud of as voicing a view that was important but not necessarily popular?
HS: I was one of the first people to come out hard against President Trump['s handling of] the pandemic. Lots of my brothers and sisters came along afterwards. To the extent that I was able to catalyze that, I'm proud of doing it. In the last few weeks, I published a paper objecting to the splitting of the OSTP director from the science advisor and, especially, not awarding the top part of the job to Alondra Nelson, who is a distinguished scientist at black female. And instead, giving part of it to Francis Collins. He’s certainly the most important science policy figure of my lifetime, but somebody who’s been doing this now for decades. I just think we have to push as hard as we can to get a cadre of young people leading us in Washington who represent the future of the country. I think the Biden administration leaned on a lot of figures from the past. I’m pushing them hard to try to stop it.
MF: I want to circle back to the erosion of the public’s trust in experts. Most experts are specialists, and specialists operate in silos that don’t capture the complexity of scientific knowledge. Are some pushbacks to experts and concerns about the perils of specialization valid?
HS: You're on the right track there. What we need is more respect for the generalist. We can't help the fact that you have to be very specialized to do a lot of stuff. But what we need is more partnership between specialists and people who can cross fields, especially into communication and social sciences. That handoff is just not really there right now. It's hard to get a hardcore scientist to respect people who are interested in science, education and science communication, and to treat them as equals. The last two years showed that they're at least as important, if not more so.
MF: I’m grateful that you’re leading the way in this area, Holden. Thank you for sharing your thoughts and your work.
New tech for prison reform spreads to 11 states
The U.S. has the highest incarceration rate in the world, costing $182 billion per year, partly because its antiquated data systems often fail to identify people who should be released. A tech nonprofit is trying to change that.
A new non-profit called Recidiviz is using data technology to reduce the size of the U.S. criminal justice system. The bi-coastal company (SF and NYC) is currently working with 11 states to improve their systems and, so far, has helped remove nearly 69,000 people — ones left floundering in jail or on parole when they should have been released.
“The root cause is fragmentation,” says Clementine Jacoby, 31, a software engineer who worked at Google before co-founding Recidiviz in 2019. In the 1970s and 80s, the U.S. built a series of disconnected data systems, and this patchwork is still being used by criminal justice authorities today. It requires parole officers to manually calculate release dates, leading to errors in many cases. “[They] have done everything they need to do to earn their release, but they're still stuck in the system,” Jacoby says.
Recidiviz has built a platform that connects the different databases, with the goal of identifying people who are already qualified for release but remain behind bars or on supervision. “Think of Recidiviz like Google Maps,” says Jacoby, who worked on Maps when she was at the tech giant. Google Maps takes in data from different sources – satellite images, street maps, local business data — and organizes it into one easy view. “Recidiviz does something similar with criminal justice data,” Jacoby explains, “making it easy to identify people eligible to come home or to move to less intensive levels of supervision.”
People like Jacoby’s uncle. His experience with incarceration is what inspired her passion for criminal justice reform in the first place.
The problems are vast
The U.S. has the highest incarceration rate in the world — 2 million people according to the watchdog group, Prison Policy Initiative — at a cost of $182 billion a year. The numbers could be a lot lower if not for an array of problems including inaccurate sentencing calculations, flawed algorithms and parole violations laws.
Sentencing miscalculations
To determine eligibility for release, the current system requires corrections officers to check 21 different requirements spread across five different databases for each of the 90 to 100 people under their supervision. These manual calculations are time prohibitive, says Jacoby, and fall victim to human error.
In addition, Recidiviz found that policies aimed at helping to reduce the prison population don’t always work correctly. A key example is time off for good behavior laws that allow inmates to earn one day off for every 30 days of good behavior. Some states' data systems are built to calculate time off as one day per month of good behavior, rather than per day. Over the course of a decade-long sentence, Jacoby says these miscalculations can lead to a huge discrepancy in the calculated release data and the actual release date.
Algorithms
Commercial algorithm-based software systems for risk assessment continue to be widely used in the criminal justice system, even though a 2018 study published in Science Advances exposed their limitations. After the study went viral, it took three years for the Justice Department to issue a report on their own flawed algorithms used to reduce the federal prison population as part of the 2018 First Step Act. The program, it was determined, overestimated the risk of putting inmates of color into early-release programs.
Despite its name, Recidiviz does not build these types of algorithms for predicting recidivism, or whether someone will commit another crime after being released from prison. Rather, Jacoby says the company’s "descriptive analytics” approach is specifically intended to weed out incarceration inequalities and avoid algorithmic pitfalls.
Parole violation laws
Research shows that 350,000 people a year — about a quarter of the total prison population — are sent back not because they’ve committed another crime, but because they’ve broken a specific rule of their probation. “Things that wouldn't send you or I to prison, but would send someone on parole,” such as crossing county lines or being in the presence of alcohol when they shouldn’t be, are inflating the prison population, says Jacoby.
It’s personal for the co-founder and CEO
“I grew up with an uncle who went into the prison system,” Jacoby says. At 19, he was sentenced to ten years in prison for a non-violent crime. A few months after being released from jail, he was sent back for a non-violent parole violation.
“For my family, the fact that one in four prison admissions are driven not by a crime but by someone who's broken a rule on probation and parole was really profound because that happened to my uncle,” Jacoby says. The experience led her to begin studying criminal justice in high school, then college. She continued her dive into how the criminal justice system works as part of her Passion Project while at Google, a program that allows employees to spend 20 percent of their time on pro-bono work. Two colleagues whose family members had also been stuck in the system joined her.
As part of the project, Jacoby interviewed hundreds of people involved in the criminal justice system. “Those on the right, those on the left, agreed that bad data was slowing down reform,” she says. Their research brought them to North Dakota where they began to understand the root of the problem. The corrections department is making “huge, consequential decisions every day [without] … the data,” Jacoby says. In a new video by Recidiviz not yet released, Jacoby recounts her exchange with the state’s director of corrections who told her, “‘It’s not that we have the data and we just don’t know how to make it public; we don’t have the information you think we have.'"
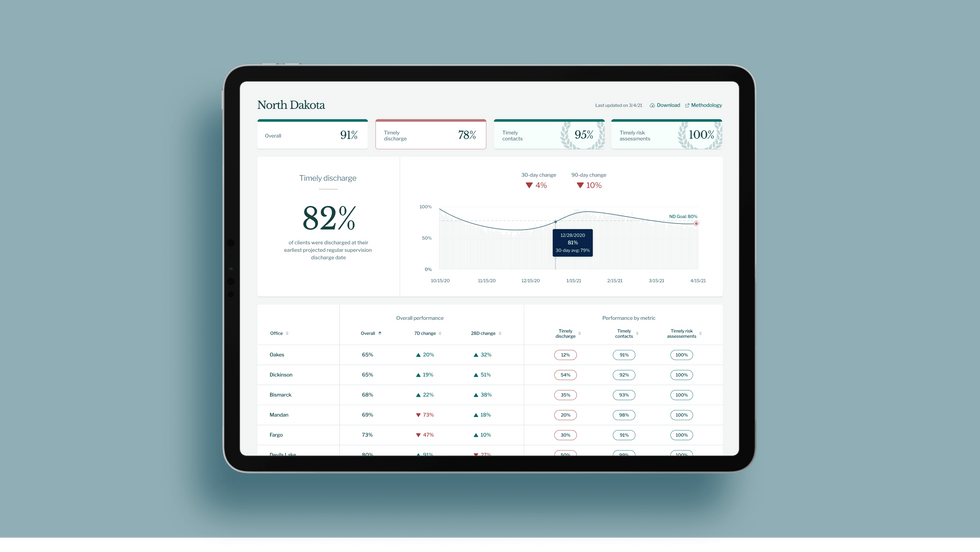
A mock-up (with fake data) of the types of dashboards and insights that Recidiviz provides to state governments.
Recidiviz
As a software engineer, Jacoby says the comment made no sense to her — until she witnessed it first-hand. “We spent a lot of time driving around in cars with corrections directors and parole officers watching them use these incredibly taxing, frankly terrible, old data systems,” Jacoby says.
As they weeded through thousands of files — some computerized, some on paper — they unearthed the consequences of bad data: Hundreds of people in prison well past their release date and thousands more whose release from parole was delayed because of minor paperwork issues. They found individuals stuck in parole because they hadn’t checked one last item off their eligibility list — like simply failing to provide their parole officer with a paystub. And, even when parolees advocated for themselves, the archaic system made it difficult for their parole officers to confirm their eligibility, so they remained in the system. Jacoby and her team also unpacked specific policies that drive racial disparities — such as fines and fees.
The Solution
It’s more than a trivial technical challenge to bring the incomplete, fragmented data onto a 21st century data platform. It takes months for Recidiviz to sift through a state’s information systems to connect databases “with the goal of tracking a person all the way through their journey and find out what’s working for 18- to 25-year-old men, what’s working for new mothers,” explains Jacoby in the video.
TED Talk: How bad data traps people in the U.S. justice system

TED Fellow Clementine Jacoby's TED Talk went live on Jan. 13. It describes how we can fix bad data in the criminal justice system, "bringing thousands of people home, reducing costs and improving public safety along the way."
Clementine Jacoby • TED2022
Ojmarrh Mitchell, an associate professor in the School of Criminology and Criminal Justice at Arizona State University, who is not involved with the company, says what Recidiviz is doing is “remarkable.” His perspective goes beyond academic analysis. In his pre-academic years, Mitchell was a probation officer, working within the framework of the “well known, but invisible” information sharing issues that plague criminal justice departments. The flexibility of Recidiviz’s approach is what makes it especially innovative, he says. “They identify the specific gaps in each jurisdiction and tailor a solution for that jurisdiction.”
On the downside, the process used by Recidiviz is “a bit opaque,” Mitchell says, with few details available on how Recidiviz designs its tools and tracks outcomes. By sharing more information about how its actions lead to progress in a given jurisdiction, Recidiviz could help reformers in other places figure out which programs have the best potential to work well.
The eleven states in which Recidiviz is working include California, Colorado, Maine, Michigan, Missouri, Pennsylvania and Tennessee. And a pilot program launched last year in Idaho, if scaled nationally, with could reduce the number of people in the criminal justice system by a quarter of a million people, Jacoby says. As part of the pilot, rather than relying on manual calculations, Recidiviz is equipping leaders and the probation officers with actionable information with a few clicks of an app that Recidiviz built.
Mitchell is disappointed that there’s even the need for Recidiviz. “This is a problem that government agencies have a responsibility to address,” he says. “But they haven’t.” For one company to come along and fill such a large gap is “remarkable.”
How Leqembi became the biggest news in Alzheimer’s disease in 40 years, and what comes next
Betsy Groves, 73, with her granddaughter. Groves learned in 2021 that she has Alzheimer's disease. She hopes to take Leqembi, a drug approved by the FDA last week.
A few months ago, Betsy Groves traveled less than a mile from her home in Cambridge, Mass. to give a talk to a bunch of scientists. The scientists, who worked for the pharmaceutical companies Biogen and Eisai, wanted to know how she lived her life, how she thought about her future, and what it was like when a doctor’s appointment in 2021 gave her the worst possible news. Groves, 73, has Alzheimer’s disease. She caught it early, through a lumbar puncture that showed evidence of amyloid, an Alzheimer’s hallmark, in her cerebrospinal fluid. As a way of dealing with her diagnosis, she joined the Alzheimer’s Association’s National Early-Stage Advisory Board, which helped her shift into seeing her diagnosis as something she could use to help others.
After her talk, Groves stayed for lunch with the scientists, who were eager to put a face to their work. Biogen and Eisai were about to release the first drug to successfully combat Alzheimer’s in 40 years of experimental disaster. Their drug, which is known by the scientific name lecanemab and the marketing name Leqembi, was granted accelerated approval by the U.S. Food and Drug Administration last Friday, Jan. 6, after a study in 1,800 people showed that it reduced cognitive decline by 27 percent over 18 months.
It is no exaggeration to say that this result is a huge deal. The field of Alzheimer’s drug development has been absolutely littered with failures. Almost everything researchers have tried has tanked in clinical trials. “Most of the things that we've done have proven not to be effective, and it's not because we haven’t been taking a ton of shots at goal,” says Anton Porsteinsson, director of the University of Rochester Alzheimer's Disease Care, Research, and Education Program, who worked on the lecanemab trial. “I think it's fair to say you don't survive in this field unless you're an eternal optimist.”
As far back as 1984, a cure looked like it was within reach: Scientists discovered that the sticky plaques that develop in the brains of those who have Alzheimer’s are made up of a protein fragment called beta-amyloid. Buildup of beta-amyloid seemed to be sufficient to disrupt communication between, and eventually kill, memory cells. If that was true, then the cure should be straightforward: Stop the buildup of beta-amyloid; stop the Alzheimer’s disease.
It wasn’t so simple. Over the next 38 years, hundreds of drugs designed either to interfere with the production of abnormal amyloid or to clear it from the brain flamed out in trials. It got so bad that neuroscience drug divisions at major pharmaceutical companies (AstraZeneca, Pfizer, Bristol-Myers, GSK, Amgen) closed one by one, leaving the field to smaller, scrappier companies, like Cambridge-based Biogen and Tokyo-based Eisai. Some scientists began to dismiss the amyloid hypothesis altogether: If this protein fragment was so important to the disease, why didn’t ridding the brain of it do anything for patients? There was another abnormal protein that showed up in the brains of Alzheimer’s patients, called tau. Some researchers defected to the tau camp, or came to believe the proteins caused damage in combination.
The situation came to a head in 2021, when the FDA granted provisional approval to a drug called aducanumab, marketed as Aduhelm, against the advice of its own advisory council. The approval was based on proof that Aduhelm reduced beta-amyloid in the brain, even though one research trial showed it had no effect on people’s symptoms or daily life. Aduhelm could also cause serious side effects, like brain swelling and amyloid related imaging abnormalities (known as ARIA, these are basically micro-bleeds that appear on MRI scans). Without a clear benefit to memory loss that would make these risks worth it, Medicare refused to pay for Aduhelm among the general population. Two congressional committees launched an investigation into the drug’s approval, citing corporate greed, lapses in protocol, and an unjustifiably high price. (Aduhelm was also produced by the pharmaceutical company Biogen.)
To be clear, Leqembi is not the cure Alzheimer’s researchers hope for. While the drug is the first to show clear signs of a clinical benefit, the scientific establishment is split on how much of a difference Leqembi will make in the real world.
So far, Leqembi is like Aduhelm in that it has been given accelerated approval only for its ability to remove amyloid from the brain. Both are monoclonal antibodies that direct the immune system to attack and clear dysfunctional beta-amyloid. The difference is that, while that’s all Aduhelm was ever shown to do, Leqembi’s makers have already asked the FDA to give it full approval – a decision that would increase the likelihood that Medicare will cover it – based on data that show it also improves Alzheimer’s sufferer’s lives. Leqembi targets a different type of amyloid, a soluble version called “protofibrils,” and that appears to change the effect. “It can give individuals and their families three, six months longer to be participating in daily life and living independently,” says Claire Sexton, PhD, senior director of scientific programs & outreach for the Alzheimer's Association. “These types of changes matter for individuals and for their families.”
To be clear, Leqembi is not the cure Alzheimer’s researchers hope for. It does not halt or reverse the disease, and people do not get better. While the drug is the first to show clear signs of a clinical benefit, the scientific establishment is split on how much of a difference Leqembi will make in the real world. It has “a rather small effect,” wrote NIH Alzheimer’s researcher Madhav Thambisetty, MD, PhD, in an email to Leaps.org. “It is unclear how meaningful this difference will be to patients, and it is unlikely that this level of difference will be obvious to a patient (or their caregivers).” Another issue is cost: Leqembi will become available to patients later this month, but Eisai is setting the price at $26,500 per year, meaning that very few patients will be able to afford it unless Medicare chooses to reimburse them for it.
The same side effects that plagued Aduhelm are common in Leqembi treatment as well. In many patients, amyloid doesn’t just accumulate around neurons, it also forms deposits in the walls of blood vessels. Blood vessels that are shot through with amyloid are more brittle. If you infuse a drug that targets amyloid, brittle blood vessels in the brain can develop leakage that results in swelling or bleeds. Most of these come with no symptoms, and are only seen during testing, which is why they are called “imaging abnormalities.” But in situations where patients have multiple diseases or are prescribed incompatible drugs, they can be serious enough to cause death. The three deaths reported from Leqembi treatment (so far) are enough to make Thambisetty wonder “how well the drug may be tolerated in real world clinical practice where patients are likely to be sicker and have multiple other medical conditions in contrast to carefully selected patients in clinical trials.”
Porsteinsson believes that earlier detection of Alzheimer’s disease will be the next great advance in treatment, a more important step forward than Leqembi’s approval.
Still, there are reasons to be excited. A successful Alzheimer’s drug can pave the way for combination studies, in which patients try a known effective drug alongside newer, more experimental ones; or preventative studies, which take place years before symptoms occur. It also represents enormous strides in researchers’ understanding of the disease. For example, drug dosages have increased massively—in some cases quadrupling—from the early days of Alzheimer’s research. And patient selection for studies has changed drastically as well. Doctors now know that you’ve got to catch the disease early, through PET-scans or CSF tests for amyloid, if you want any chance of changing its course.
Porsteinsson believes that earlier detection of Alzheimer’s disease will be the next great advance in treatment, a more important step forward than Leqembi’s approval. His lab already uses blood tests for different types of amyloid, for different types of tau, and for measures of neuroinflammation, neural damage, and synaptic health, but commercially available versions from companies like C2N, Quest, and Fuji Rebio are likely to hit the market in the next couple of years. “[They are] going to transform the diagnosis of Alzheimer's disease,” Porsteinsson says. “If someone is experiencing memory problems, their physicians will be able to order a blood test that will tell us if this is the result of changes in your brain due to Alzheimer's disease. It will ultimately make it much easier to identify people at a very early stage of the disease, where they are most likely to benefit from treatment.”
Learn more about new blood tests to detect Alzheimer's
Early detection can help patients for more philosophical reasons as well. Betsy Groves credits finding her Alzheimer’s early with giving her the space to understand and process the changes that were happening to her before they got so bad that she couldn’t. She has been able to update her legal documents and, through her role on the Advisory Group, help the Alzheimer’s Association with developing its programs and support services for people in the early stages of the disease. She still drives, and because she and her husband love to travel, they are hoping to get out of grey, rainy Cambridge and off to Texas or Arizona this spring.
Because her Alzheimer’s disease involves amyloid deposits (a “substantial portion” do not, says Claire Sexton, which is an additional complication for research), and has not yet reached an advanced stage, Groves may be a good candidate to try Leqembi. She says she’d welcome the opportunity to take it. If she can get access, Groves hopes the drug will give her more days to be fully functioning with her husband, daughters, and three grandchildren. Mostly, she avoids thinking about what the latter stages of Alzheimer’s might be like, but she knows the time will come when it will be her reality. “So whatever lecanemab can do to extend my more productive ways of engaging with relationships in the world,” she says. “I'll take that in a minute.”

