SCOOP: Largest Cryobank in the U.S. to Offer Ancestry Testing

Vanessa Colimorio (left) and Sharon Kochlany (right) at a farm with their four-year-old twin daughters and one-year-old son. The kids share the same sperm donor.
Sharon Kochlany and Vanessa Colimorio's four-year-old twin girls had a classic school assignment recently: make a family tree. They drew themselves and their one-year-old brother branching off from their moms, with aunts, uncles, and grandparents forking off to the sides.
The recently-gained sovereignty of queer families stands to be lost if a consumer DNA test brings a stranger's identity out of the woodwork.
What you don't see in the invisible space between Kochlany and Colimorio, however, is the sperm donor they used to conceive all three children.
To look at a family tree like this is to see in its purest form that kinship can supersede biology—the boundaries of where this family starts and stops are clear to everyone in it, in spite of a third party's genetic involvement. This kind of self-definition has always been synonymous with LGBTQ families, especially those that rely on donor gametes (sperm or eggs) to exist.
But the world around them has changed quite suddenly: The recent consumer DNA testing boom has made it more complicated than ever for families built through reproductive technology—openly, not secretively—to maintain the strong sense of autonomy and privacy that can be crucial for their emotional security. Prospective parents and cryobanks are now mulling how best to bring a new generation of donor-conceived people into this world in a way that leaves open the choice to know more about their ancestry without obliterating an equally important choice: the right not to know about biological relatives.
For queer parents who have long fought for social acceptance, having a biological relationship to their children has been revolutionary, and using an unknown donor as a means to this end especially so. Getting help from a friend often comes with the expectation that the friend will also have social involvement in the family, which some people are comfortable with, but being able to access sperm from an unknown donor—which queer parents have only been able to openly do since the early 1980s—grants them the reproductive autonomy to create families seemingly on their own. That recently-gained sovereignty stands to be lost if a consumer DNA test brings a stranger's identity out of the woodwork.
At the same time, it's natural for donor-conceived people to want to know more about where they come from ethnically, even if they don't want to know the identity of their donor. As a donor-conceived person myself, I know my donor's self-reported ethnicity, but have often wondered how accurate it is.
Opening the Pandora's box of a consumer DNA test as a way to find out has always felt profoundly unappealing to me, however. Many people have accidentally learned they're donor-conceived by unwittingly using these tools, but I already know that about myself going in, and subsequently know I'll be connected to a large web of people whose existence I'm not interested in learning about. In addition to possibly identifying my anonymous donor, his family could also show up, along with any donor-siblings—other people with whom I share a donor. My single lesbian mom is enough for me, and the trade off to learn more about my ethnic ancestry has never seemed worth it.
In 1992, when I was born, no one was planning for how consumer DNA tests might upend or illuminate one's sense of self. But the donor community has always had to stay nimble with balancing privacy concerns and psychological well-being, so it should come as no surprise that figuring out how to do so in 2020 includes finding a way to offer ancestry insight while circumventing consumer DNA tests.
A New Paradigm
This is the rationale behind unprecedented industry news that LeapsMag can exclusively break: Within the next few weeks, California Cryobank, the largest cryobank in the country, will begin offering genetically-verified ancestry information on the free public part of every donor's anonymous profile in its database, something no other cryobanks yet offer (an exact launch date was not available at the time of publication). Currently, California Cryobank's donor profiles include a short self-reported list that might merely say, "Ancestry: German, Lebanese, Scottish."
The new information will be a report in pie chart form that details exactly what percentages of a donor's DNA come from up to 26 ethnicities—it's analogous to, but on a smaller scale than, the format offered by consumer DNA testing companies, and uses the same base technology that looks for single nucleotide polymorphisms in DNA that are associated with specific ethnicities. But crucially, because the donor takes the DNA test through California Cryobank, not a consumer-facing service, the information is not connected in a network to anyone else's DNA test. It's also taken before any offspring exist so there's no chance of revealing a donor-conceived person's identity this way.
Later, when a donor-conceived person is born, grows up, and wants information about their ethnicity from the donor side, all they need is their donor's anonymous ID number to look it up. The donor-conceived person never takes a genetic test, and therefore also can't accidentally find donor siblings this way. People who want to be connected to donor siblings can use a sibling registry where other people who want to be found share donor ID numbers and look for matches (this is something that's been available for decades, and remains so).
"With genetic testing, you have no control over who reaches out to you, and at what point in your life."
California Cryobank will require all new donors to consent to this extra level of genetic testing, setting a new standard for what information prospective parents and donor-conceived people can expect to have. In the immediate, this information will be most useful for prospective parents looking for donors with specific backgrounds, possibly ones similar to their own.
It's a solution that was actually hiding in plain sight. Two years ago, California Cryobank's partner Sema4, the company handling the genetic carrier testing that's used to screen for heritable diseases, started analyzing ethnic data in its samples. That extra information was being collected because it can help calculate a more accurate assessment of genetic risks that run in certain populations—like Ashkenazi Jews and Tay Sachs disease—than relying on oral family histories. Shortly after a plan to start collecting these extra data, Jamie Shamonki, chief medical officer of California Cryobank, realized the companies would be sitting on a goldmine for a different reason.
"I didn't want to use one of these genetic testing companies like Ancestry to accomplish this," says Shamonki. "The whole thing we're trying to accomplish is also privacy."
Consumer-facing DNA testing companies are not HIPAA compliant (whereas Sema4, which isn't direct-to-consumer, is HIPAA compliant), which means there are no legal privacy protections covering people who add their DNA to these databases. Although some companies, like 23andMe, allow users to opt-out of being connected with genetic relatives, the language can be confusing to navigate, requires a high level of knowledge and self-advocacy on the user's part, and, as an opt-out system, is not set up to protect the user from unwanted information by default; many unwittingly walk right into such information as a result.
Additionally, because consumer-facing DNA testing companies operate outside the legal purview that applies to other health care entities, like hospitals, even a person who does opt-out of being linked to genetic relatives is not protected in perpetuity from being re-identified in the future by a change in company policy. The safest option for people with privacy concerns is to stay out of these databases altogether.
For California Cryobank, the new information about donor heritage won't retroactively be added to older profiles in the system, so donor-conceived people who already exist won't benefit from the ancestry tool, but it'll be the new standard going forward. The company has about 500 available donors right now, many of which have been in their registry for a while; about 100 of those donors, all new, will have this ancestry data on their profiles.
Shamonki says it has taken about two years to get to the point of publicly including ancestry information on a donor's profile because it takes about nine months of medical and psychological screening for a donor to go from walking through the door to being added to their registry. The company wanted to wait to launch until it could offer this information for a significant number of donors. As more new donors come online under the new protocol, the number with ancestry information on their profiles will go up.
For Parents: An Unexpected Complication
While this change will no doubt be welcome progress for LGBTQ families contemplating parenthood, it'll never be possible to put this entire new order back in the box. What are such families who already have donor-conceived children losing in today's world of widespread consumer genetic testing?
Kochlany and Colimorio's twins aren't themselves much older than the moment at-home DNA testing really started to take off. They were born in 2015, and two years later the industry saw its most significant spike. By now, more than 26 million people's DNA is in databases like 23andMe and Ancestry; as a result, it's estimated that within a year, 90 percent of Americans of European descent will be identifiable through these consumer databases, by way of genetic third cousins, even if they didn't want to be found and never took the test themselves. This was the principle behind solving the Golden State Killer cold case.
The waning of privacy through consumer DNA testing fundamentally clashes with the priorities of the cyrobank industry, which has long sought to protect the privacy of donor-conceived people, even as open identification became standard. Since the 1980s, donors have been able to allow their identity to be released to any offspring who is at least 18 and wants the information. Lesbian moms pushed for this option early on so their children—who would obviously know they couldn't possibly be the biological product of both parents—would never feel cut off from the chance to know more about themselves. But importantly, the openness is not a two-way street: the donors can't ever ask for the identities of their offspring. It's the latter that consumer DNA testing really puts at stake.
"23andMe basically created the possibility that there will be donors who will have contact with their donor-conceived children, and that's not something that I think the donor community is comfortable with," says I. Glenn Cohen, director of Harvard Law School's Center for Health Law Policy, Biotechnology & Bioethics. "That's about the donor's autonomy, not the rearing parents' autonomy, or the donor-conceived child's autonomy."
Kochlany and Colimorio have an open identification donor and fully support their children reaching out to California Cryobank to get more information about him if they want to when they're 18, but having a singular name revealed isn't the same thing as having contact, nor is it the same thing as revealing a web of dozens of extended genetic relations. Their concern now is that if their kids participate in genetic testing, a stranger—someone they're careful to refer to as only "the donor" and never "dad"—will reach out to the children to begin some kind of relationship. They know other people who are contemplating giving their children DNA tests, and feel staunchly that it wouldn't be right for their family.
"With genetic testing, you have no control over who reaches out to you, and at what point in your life," Kochlany says. "[People] reaching out and trying to say, 'Hey I know who your dad is' throws a curveball. It's like, 'Wait, I never thought I had a dad.' It might put insecurities in their minds."
"We want them to have the opportunity to choose whether or not they want to reach out," Colimorio adds.
Kochlany says that when their twins are old enough to start asking questions, she and Colimorio plan to frame it like this: "The donor was kind of like a technology that helped us make you a person, and make sure that you exist," she says, role playing a conversation with their kids. "But it's not necessarily that you're looking to this person [for] support or love, or because you're missing a piece."
It's a line in the sand that's present even for couples still far off from conceiving. When Mallory Schwartz, a film and TV producer in Los Angeles, and Lauren Pietra, a marriage and family therapy associate (and Shamonki's step-daughter), talk about getting married someday, it's a package deal with talking about how they'll approach having kids. They feel there are too many variables and choices to make around family planning as a same-sex couple these days to not have those conversations simultaneously. Consumer DNA databases are already on their minds.
"It frustrates me that the DNA databases are just totally unregulated," says Schwartz. "I hope they are by the time we do this. I think everyone deserves a right to privacy when making your family [using a sperm donor]."
"I wouldn't want to create a world where people who are donor-conceived feel like they can't participate in this technology because they're trying to shut out [other] information."
On the prospect of having a donor relation pop up non-consensually for a future child, Pietra says, "I don't like it. It would be really disappointing if the child didn't want [contact], and unfortunately they're on the receiving end."
You can see how important preserving the right to keep this door closed is when you look at what's going on at The Sperm Bank of California. This pioneering cryobank was the first in the world to openly serve LGBTQ people and single women, and also the first to offer the open identification option when it opened in 1982, but not as many people are asking for their donor's identity as expected.
"We're finding a third of young people are coming forward for their donor's identity," says Alice Ruby, executive director. "We thought it would be a higher number." Viewed the other way, two-thirds of the donor-conceived people who could ethically get their donor's identity through The Sperm Bank of California are not asking the cryobank for it.
Ruby says that part of what historically made an open identification program appealing, rather than invasive or nerve-wracking, is how rigidly it's always been formatted around mutual consent, and protects against surprises for all parties. Those [donor-conceived people] who wanted more information were never barred from it, while those who wanted to remain in the dark could. No one group's wish eclipsed the other's. The potential breakdown of a system built around consent, expectations, and respect for privacy is why unregulated consumer DNA testing is most concerning to her as a path for connecting with genetic relatives.
For the last few decades in cryobanks around the world, the largest cohort of people seeking out donor sperm has been lesbian couples, followed by single women. For infertile heterosexual couples, the smallest client demographic, Ruby says donor sperm offers a solution to a medical problem, but in contrast, it historically "provided the ability for [lesbian] couples and single moms to have some reproductive autonomy." Yes, it was still a solution to a biological problem, but it was also a solution to a social one.
The Sperm Bank of California updated its registration forms to include language urging parents, donor-conceived people, and donors not to use consumer DNA tests, and to go through the cryobank if they, understandably, want to learn more about who they're connected to. But truthfully, there's not much else cryobanks can do to protect clients on any side of the donor transaction from surprise contact right now—especially not from relatives of the donor who may not even know someone in their family has donated sperm.
A Tricky Position
Personally, I've known I was donor-conceived from day one. It has never been a source of confusion, angst, or curiosity, and in fact has never loomed particularly large for me in any way. I see it merely as a type of reproductive technology—on par with in vitro fertilization—that enabled me to exist, and, now that I do exist, is irrelevant. Being confronted with my donor's identity or any donor siblings would make this fact of my conception bigger than I need it to be, as an adult with a full-blown identity derived from all of my other life experiences. But I still wonder about the minutiae of my ethnicity in much the same way as anyone else who wonders, and feel there's no safe way for me to find out without relinquishing some of my existential independence.
"People obviously want to participate in 23andMe and Ancestry because they're interested in knowing more about themselves," says Shamonki. "I wouldn't want to create a world where people who are donor-conceived feel like they can't participate in this technology because they're trying to shut out [other] information."
After all, it was the allure of that exact conceit—knowing more about oneself—that seemed to magnetically draw in millions of people to these tools in the first place. It's an experience that clearly taps into a population-wide psychic need, even—perhaps especially—if one's origins are a mystery.
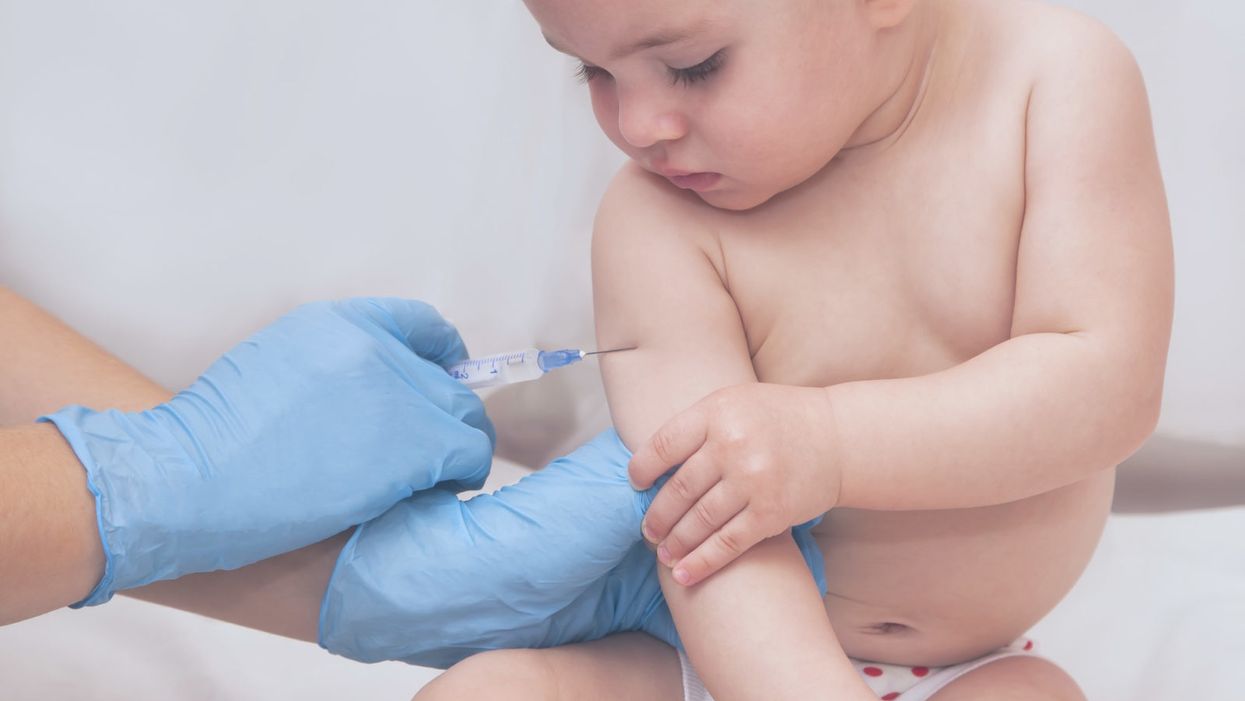
Not Vaccinating Your Kids Endangers Public Health
A pediatrician gives a one-year-old child a vaccine.
[Editor's Note: This opinion essay is in response to our current Big Question, which we posed to experts with different viewpoints: "Where should society draw the line between requiring vaccinations for children and allowing parental freedom of choice?"]
Society has a right and at times an obligation to require children to be vaccinated. Vaccines are one of the most effective medical and public health interventions. They save lives and prevent suffering. The vast majority of parents in the United States fully vaccinate their children according to the recommended immunization schedule. These parents are making decisions so that the interests of their children and the interest of society are the same. There are no ethical tensions.
"Measles is only a plane ride away from American children."
A strong scientific basis supports the recommended immunization schedule. The benefits of recommended vaccines are much bigger than the risks. However, a very small proportion of parents are ideologically opposed to vaccines. A slightly larger minority of parents do not believe that all of the recommended vaccines are in their child's best interests.
Forgoing vaccinations creates risk to the child of contracting diseases. It also creates risk to communities and vulnerable groups of people who cannot be vaccinated because of their age or health status.
For example, many vaccines are not able to be given to newborns, such as the measles vaccine which is recommended at 12-15 months of age, leaving young children vulnerable. Many diseases are particularly dangerous for young children. There are also some children who can't be vaccinated, such as pediatric cancer patients who are undergoing chemotherapy or radiation treatment. These children are at increased risk of serous complication or death.
Then there are people who are vaccinated but remain susceptible to disease because no vaccine is 100% effective. In the case of measles, two doses of vaccines protect 97% of people, leaving 3% still susceptible even after being fully vaccinated. All of these groups of people – too young, not eligible, and vaccinated but still susceptible – are dependent on almost everyone else to get vaccinated in order for them to be protected.
Sadly, even though measles has been largely controlled because most people get the very safe and very effective vaccine, we are now seeing dangerous new outbreaks because some parents are refusing vaccines for their children, especially in Europe. Children have died. Measles is only a plane ride away from American children.
There have been repeated measles outbreaks in the United States – such as the Disneyland outbreak and six outbreaks already this year - because of communities where too many parents refuse the vaccine and measles is brought over, often from Europe.
The public health benefits cannot be emphasized enough: Vaccines are not just about protecting your child. Vaccines protect other children and the entire community. Vaccine-preventable diseases (with the exception of tetanus) are spread from person to person. The decision of a parent to not vaccinate their child can endanger other children and vulnerable people.
As a vaccine safety researcher for 20 years, I believe that the community benefit of vaccination can provide justification to limit parental autonomy.
Given these tensions between parental autonomy and the protective value of vaccines, the fundamental question remains: Should society require all children to submit to vaccinations? As a vaccine safety researcher for 20 years, I believe that the community benefit of vaccination can provide justification to limit parental autonomy.
In the United States, we see this balancing act though state requirements for vaccinations to enter school and the varying availability of non-medical exemptions to these laws. Mandatory vaccination in the United States are all state laws. All states require children entering school to receive vaccines and permit medical exemptions. There are a lot of differences between states regarding which vaccines are required, target populations (daycare, school entry, middle school, college), and existence and types of non-medical (religious or philosophical) exemptions that are permitted.
Amid recent measles outbreaks, for instance, California eliminated all non-medical exemptions, making it one of three states that only permit medical exemptions. The existence and enforcement of these school laws reflect broad public support for vaccines to protect the community from disease outbreaks.
I worry about how many kids must suffer, and even die, from diseases like measles until enough is enough. Such tragedies have no place in the modern era. All parents want to do right by their children. All parents deserve autonomy when it comes to decision-making. But when their choices confer serious risks to others, the buck should stop. Our nation would be better off—both medically and ethically—if we did not turn our backs on our most vulnerable individuals.
[Editor's Note: Read the opposite viewpoint here.]
Your Body Has This Astonishing Magical Power
A fierce champion fighter in action, representing the incredible power of the human immune system.
It's vacation time. You and your family visit a country where you've never been and, in fact, your parents or grandparents had never been. You find yourself hiking beside a beautiful lake. It's a gorgeous day. You dive in. You are not alone.
How can your T cells and B cells react to a pathogen they've never seen?
In the water swim parasites, perhaps a parasite called giardia. The invader slips in through your mouth or your urinary tract. This bug is entirely new to you, and there's more. It might be new to everyone you've ever met or come into contact with. The parasite may have evolved in this setting for hundreds of thousands of years so that it's different from any giardia bug you've ever come into contact with before or that thrives in the region where you live.
How can your T cells and B cells react to a pathogen they've never seen, never knew existed, and were never inoculated against, and that you, or your doctors, in all their wisdom, could never have foreseen?
This is the infinity problem.
For years, this was the greatest mystery in immunology.
As I reported An Elegant Defense -- my book about the science of the immune system told through the lives of scientists and medical patients -- I was repeatedly struck by the profundity of this question. It is hard to overstate: how can we survive in a world with such myriad possible threats?

Matt Richtel's new book about the science of the immune system, An Elegant Defense, was published this month.
To further underscore the quandary, the immune system has to neutralize threats without killing the rest of the body. If the immune system could just kill the rest of the body too, the solution to the problem would be easy. Nuke the whole party. That obviously won't work if we are to survive. So the immune system has to be specific to the threat while also leaving most of our organism largely alone.
"God had two options," Dr. Mark Brunvand told me. "He could turn us into ten-foot-tall pimples, or he could give us the power to fight 10 to the 12th power different pathogens." That's a trillion potential bad actors. Why pimples? Pimples are filled with white blood cells, which are rich with immune system cells. In short, you could be a gigantic immune system and nothing else, or you could have some kind of secret power that allowed you to have all the other attributes of a human being—brain, heart, organs, limbs—and still somehow magically be able to fight infinite pathogens.
Dr. Brunvand is a retired Denver oncologist, one of the many medical authorities in the book – from wizened T-cell innovator Dr. Jacques Miller, to the finder of fever, Dr. Charles Dinarello, to his eminence Dr. Anthony Fauci at the National Institutes of Health to newly minted Nobel-Prize winner Jim Allison.
In the case of Dr. Brunvand, the oncologist also is integral to one of the book's narratives, a remarkable story of a friend of mine named Jason. Four years ago, he suffered late, late stage cancer, with 15 pounds of lymphoma growing in his back, and his oncologist put him into hospice. Then Jason became one of the first people ever to take an immunotherapy drug for lymphoma and his tumors disappeared. Through Jason's story, and a handful of other fascinating tales, I showcase how the immune system works.
There are two options for creating such a powerful immune system: we could be pimples or have some other magical power.
Dr. Brunvand had posited to me that there were two options for creating such a powerful and multifaceted immune system: we could be pimples or have some other magical power. You're not a pimple. So what was the ultimate solution?
Over the years, there were a handful of well-intentioned, thoughtful theories, but they strained to account for the inexplicable ability of the body to respond to virtually anything. The theories were complex and suffered from that peculiar side effect of having terrible names—like "side-chain theory" and "template-instructive hypothesis."
This was the background when along came Susumu Tonegawa.
***
Tonegawa was born in 1939, in the Japanese port city of Nagoya, and was reared during the war. Lucky for him, his father was moved around in his job, and so Tonegawa grew up in smaller towns. Otherwise, he might've been in Nagoya on May 14,1944, when the United States sent nearly 550 B-29 bombers to take out key industrial sites there and destroyed huge swaths of the city.
Fifteen years later, in 1959, Tonegawa was a promising student when a professor in Kyoto told him that he should go to the United States because Japan lacked adequate graduate training in molecular biology. A clear, noteworthy phenomenon was taking shape: Immunology and its greatest discoveries were an international affair, discoveries made through cooperation among the world's best brains, national boundaries be damned.
Tonegawa wound up at the University of California at San Diego, at a lab in La Jolla, "the beautiful Southern California town near the Mexican border." There, in multicultural paradise, he received his PhD, studying in the lab of Masaki Hayashi and then moved to the lab of Renato Dulbecco. Dr. Dulbecco was born in Italy, got a medical degree, was recruited to serve in World War II, where he fought the French and then, when Italian fascism collapsed, joined the resistance and fought the Germans. (Eventually, he came to the United States and in 1975 won a Nobel Prize for using molecular biology to show how viruses can lead, in some cases, to tumor creation.)
In 1970, Tonegawa—now armed with a PhD—faced his own immigration conundrum. His visa was set to expire by the end of 1970, and he was forced to leave the country for two years before he could return. He found a job in Switzerland at the Basel Institute for Immunology.
***
Around this time, new technology had emerged that allowed scientists to isolate different segments of an organism's genetic material. The technology allowed segments to be "cut" and then compared to one another. A truism emerged: If a researcher took one organism's genome and cut precisely the same segment over and over again, the resulting fragment of genetic material would match each time.
When you jump in that lake in a foreign land, filled with alien bugs, your body, astonishingly, well might have a defender that recognizes the creature.
This might sound obvious, but it was key to defining the consistency of an organism's genetic structure.
Then Tonegawa found the anomaly.
He was cutting segments of genetic material from within B cells. He began by comparing the segments from immature B cells, meaning, immune system cells that were still developing. When he compared identical segments in these cells, they yielded, predictably, identical fragments of genetic material. That was consistent with all previous knowledge.
But when he compared the segments to identical regions in mature B cells, the result was entirely different. This was new, distinct from any other cell or organism that had been studied. The underlying genetic material had changed.
"It was a big revelation," said Ruslan Medzhitov, a Yale scholar. "What he found, and is currently known, is that the antibody-encoding genes are unlike all other normal genes."
The antibody-encoding genes are unlike all other normal genes.
Yes, I used italics. Your immune system's incredible capabilities begin from a remarkable twist of genetics. When your immune system takes shape, it scrambles itself into millions of different combinations, random mixtures and blends. It is a kind of genetic Big Bang that creates inside your body all kinds of defenders aimed at recognizing all kinds of alien life forms.
So when you jump in that lake in a foreign land, filled with alien bugs, your body, astonishingly, well might have a defender that recognizes the creature.
Light the fireworks and send down the streamers!
As Tonegawa explored further, he discovered a pattern that described the differences between immature B cells and mature ones. Each of them shared key genetic material with one major variance: In the immature B cell, that crucial genetic material was mixed in with, and separated by, a whole array of other genetic material.
As the B cell matured into a fully functioning immune system cell, much of the genetic material dropped out. And not just that: In each maturing B cell, different material dropped out. What had begun as a vast array of genetic coding sharpened into this particular, even unique, strand of genetic material.
***
This is complex stuff. But a pep talk: This section is as deep and important as any in describing the wonder of the human body. Dear reader, please soldier on!
***
Researchers, who, eventually, sought a handy way to define the nature of the genetic change to the material of genes, labeled the key genetic material in an antibody with three initials: V, D, and J.
The letter V stands for variable. The variable part of the genetic material is drawn from hundreds of genes.
D stands for diversity, which is drawn from a pool of dozens of different genes.
And J is drawn from another half dozen genes.
In an immature B cell, the strands of V, D, and J material are in separate groupings, and they are separated by a relatively massive distance. But as the cell matures, a single, random copy of V remains, along with a single each of D and J, and all the other intervening material drops out. As I began to grasp this, it helped me to picture a line of genetic material stretching many miles. Suddenly, three random pieces step forward, and the rest drops away.
The combination of these genetic slices, grouped and condensed into a single cell, creates, by the power of math, trillions of different and virtually unique genetic codes.
In anticipation of threats from the unfathomable, our defenses evolved as infinity machines.
Or if you prefer a different metaphor, the body has randomly made hundreds of millions of different keys, or antibodies. Each fits a lock that is located on a pathogen. Many of these antibodies are combined such that they are alien genetic material—at least to us—and their locks will never surface in the human body. Some may not exist in the entire universe. Our bodies have come stocked with keys to the rarest and even unimaginable locks, forms of evil the world has not yet seen, but someday might. In anticipation of threats from the unfathomable, our defenses evolved as infinity machines.
"The discoveries of Tonegawa explain the genetic background allowing the enormous richness of variation among antibodies," the Nobel Prize committee wrote in its award to him years later, in 1987. "Beyond deeper knowledge of the basic structure of the immune system these discoveries will have importance in improving immunological therapy of different kinds, such as, for instance, the enforcement of vaccinations and inhibition of reactions during transplantation. Another area of importance is those diseases where the immune defense of the individual now attacks the body's own tissues, the so-called autoimmune diseases."
Indeed, these revelations are part of a period of time it would be fair to call the era of immunology, stretching from the middle of the 20th century to the present. During that period, we've come from sheer ignorance of the most basic aspects of the immune system to now being able to tinker under the hood with monoclonal antibodies and other therapies. And we are, in many ways, just at the beginning.