SCOOP: Largest Cryobank in the U.S. to Offer Ancestry Testing

Vanessa Colimorio (left) and Sharon Kochlany (right) at a farm with their four-year-old twin daughters and one-year-old son. The kids share the same sperm donor.
Sharon Kochlany and Vanessa Colimorio's four-year-old twin girls had a classic school assignment recently: make a family tree. They drew themselves and their one-year-old brother branching off from their moms, with aunts, uncles, and grandparents forking off to the sides.
The recently-gained sovereignty of queer families stands to be lost if a consumer DNA test brings a stranger's identity out of the woodwork.
What you don't see in the invisible space between Kochlany and Colimorio, however, is the sperm donor they used to conceive all three children.
To look at a family tree like this is to see in its purest form that kinship can supersede biology—the boundaries of where this family starts and stops are clear to everyone in it, in spite of a third party's genetic involvement. This kind of self-definition has always been synonymous with LGBTQ families, especially those that rely on donor gametes (sperm or eggs) to exist.
But the world around them has changed quite suddenly: The recent consumer DNA testing boom has made it more complicated than ever for families built through reproductive technology—openly, not secretively—to maintain the strong sense of autonomy and privacy that can be crucial for their emotional security. Prospective parents and cryobanks are now mulling how best to bring a new generation of donor-conceived people into this world in a way that leaves open the choice to know more about their ancestry without obliterating an equally important choice: the right not to know about biological relatives.
For queer parents who have long fought for social acceptance, having a biological relationship to their children has been revolutionary, and using an unknown donor as a means to this end especially so. Getting help from a friend often comes with the expectation that the friend will also have social involvement in the family, which some people are comfortable with, but being able to access sperm from an unknown donor—which queer parents have only been able to openly do since the early 1980s—grants them the reproductive autonomy to create families seemingly on their own. That recently-gained sovereignty stands to be lost if a consumer DNA test brings a stranger's identity out of the woodwork.
At the same time, it's natural for donor-conceived people to want to know more about where they come from ethnically, even if they don't want to know the identity of their donor. As a donor-conceived person myself, I know my donor's self-reported ethnicity, but have often wondered how accurate it is.
Opening the Pandora's box of a consumer DNA test as a way to find out has always felt profoundly unappealing to me, however. Many people have accidentally learned they're donor-conceived by unwittingly using these tools, but I already know that about myself going in, and subsequently know I'll be connected to a large web of people whose existence I'm not interested in learning about. In addition to possibly identifying my anonymous donor, his family could also show up, along with any donor-siblings—other people with whom I share a donor. My single lesbian mom is enough for me, and the trade off to learn more about my ethnic ancestry has never seemed worth it.
In 1992, when I was born, no one was planning for how consumer DNA tests might upend or illuminate one's sense of self. But the donor community has always had to stay nimble with balancing privacy concerns and psychological well-being, so it should come as no surprise that figuring out how to do so in 2020 includes finding a way to offer ancestry insight while circumventing consumer DNA tests.
A New Paradigm
This is the rationale behind unprecedented industry news that LeapsMag can exclusively break: Within the next few weeks, California Cryobank, the largest cryobank in the country, will begin offering genetically-verified ancestry information on the free public part of every donor's anonymous profile in its database, something no other cryobanks yet offer (an exact launch date was not available at the time of publication). Currently, California Cryobank's donor profiles include a short self-reported list that might merely say, "Ancestry: German, Lebanese, Scottish."
The new information will be a report in pie chart form that details exactly what percentages of a donor's DNA come from up to 26 ethnicities—it's analogous to, but on a smaller scale than, the format offered by consumer DNA testing companies, and uses the same base technology that looks for single nucleotide polymorphisms in DNA that are associated with specific ethnicities. But crucially, because the donor takes the DNA test through California Cryobank, not a consumer-facing service, the information is not connected in a network to anyone else's DNA test. It's also taken before any offspring exist so there's no chance of revealing a donor-conceived person's identity this way.
Later, when a donor-conceived person is born, grows up, and wants information about their ethnicity from the donor side, all they need is their donor's anonymous ID number to look it up. The donor-conceived person never takes a genetic test, and therefore also can't accidentally find donor siblings this way. People who want to be connected to donor siblings can use a sibling registry where other people who want to be found share donor ID numbers and look for matches (this is something that's been available for decades, and remains so).
"With genetic testing, you have no control over who reaches out to you, and at what point in your life."
California Cryobank will require all new donors to consent to this extra level of genetic testing, setting a new standard for what information prospective parents and donor-conceived people can expect to have. In the immediate, this information will be most useful for prospective parents looking for donors with specific backgrounds, possibly ones similar to their own.
It's a solution that was actually hiding in plain sight. Two years ago, California Cryobank's partner Sema4, the company handling the genetic carrier testing that's used to screen for heritable diseases, started analyzing ethnic data in its samples. That extra information was being collected because it can help calculate a more accurate assessment of genetic risks that run in certain populations—like Ashkenazi Jews and Tay Sachs disease—than relying on oral family histories. Shortly after a plan to start collecting these extra data, Jamie Shamonki, chief medical officer of California Cryobank, realized the companies would be sitting on a goldmine for a different reason.
"I didn't want to use one of these genetic testing companies like Ancestry to accomplish this," says Shamonki. "The whole thing we're trying to accomplish is also privacy."
Consumer-facing DNA testing companies are not HIPAA compliant (whereas Sema4, which isn't direct-to-consumer, is HIPAA compliant), which means there are no legal privacy protections covering people who add their DNA to these databases. Although some companies, like 23andMe, allow users to opt-out of being connected with genetic relatives, the language can be confusing to navigate, requires a high level of knowledge and self-advocacy on the user's part, and, as an opt-out system, is not set up to protect the user from unwanted information by default; many unwittingly walk right into such information as a result.
Additionally, because consumer-facing DNA testing companies operate outside the legal purview that applies to other health care entities, like hospitals, even a person who does opt-out of being linked to genetic relatives is not protected in perpetuity from being re-identified in the future by a change in company policy. The safest option for people with privacy concerns is to stay out of these databases altogether.
For California Cryobank, the new information about donor heritage won't retroactively be added to older profiles in the system, so donor-conceived people who already exist won't benefit from the ancestry tool, but it'll be the new standard going forward. The company has about 500 available donors right now, many of which have been in their registry for a while; about 100 of those donors, all new, will have this ancestry data on their profiles.
Shamonki says it has taken about two years to get to the point of publicly including ancestry information on a donor's profile because it takes about nine months of medical and psychological screening for a donor to go from walking through the door to being added to their registry. The company wanted to wait to launch until it could offer this information for a significant number of donors. As more new donors come online under the new protocol, the number with ancestry information on their profiles will go up.
For Parents: An Unexpected Complication
While this change will no doubt be welcome progress for LGBTQ families contemplating parenthood, it'll never be possible to put this entire new order back in the box. What are such families who already have donor-conceived children losing in today's world of widespread consumer genetic testing?
Kochlany and Colimorio's twins aren't themselves much older than the moment at-home DNA testing really started to take off. They were born in 2015, and two years later the industry saw its most significant spike. By now, more than 26 million people's DNA is in databases like 23andMe and Ancestry; as a result, it's estimated that within a year, 90 percent of Americans of European descent will be identifiable through these consumer databases, by way of genetic third cousins, even if they didn't want to be found and never took the test themselves. This was the principle behind solving the Golden State Killer cold case.
The waning of privacy through consumer DNA testing fundamentally clashes with the priorities of the cyrobank industry, which has long sought to protect the privacy of donor-conceived people, even as open identification became standard. Since the 1980s, donors have been able to allow their identity to be released to any offspring who is at least 18 and wants the information. Lesbian moms pushed for this option early on so their children—who would obviously know they couldn't possibly be the biological product of both parents—would never feel cut off from the chance to know more about themselves. But importantly, the openness is not a two-way street: the donors can't ever ask for the identities of their offspring. It's the latter that consumer DNA testing really puts at stake.
"23andMe basically created the possibility that there will be donors who will have contact with their donor-conceived children, and that's not something that I think the donor community is comfortable with," says I. Glenn Cohen, director of Harvard Law School's Center for Health Law Policy, Biotechnology & Bioethics. "That's about the donor's autonomy, not the rearing parents' autonomy, or the donor-conceived child's autonomy."
Kochlany and Colimorio have an open identification donor and fully support their children reaching out to California Cryobank to get more information about him if they want to when they're 18, but having a singular name revealed isn't the same thing as having contact, nor is it the same thing as revealing a web of dozens of extended genetic relations. Their concern now is that if their kids participate in genetic testing, a stranger—someone they're careful to refer to as only "the donor" and never "dad"—will reach out to the children to begin some kind of relationship. They know other people who are contemplating giving their children DNA tests, and feel staunchly that it wouldn't be right for their family.
"With genetic testing, you have no control over who reaches out to you, and at what point in your life," Kochlany says. "[People] reaching out and trying to say, 'Hey I know who your dad is' throws a curveball. It's like, 'Wait, I never thought I had a dad.' It might put insecurities in their minds."
"We want them to have the opportunity to choose whether or not they want to reach out," Colimorio adds.
Kochlany says that when their twins are old enough to start asking questions, she and Colimorio plan to frame it like this: "The donor was kind of like a technology that helped us make you a person, and make sure that you exist," she says, role playing a conversation with their kids. "But it's not necessarily that you're looking to this person [for] support or love, or because you're missing a piece."
It's a line in the sand that's present even for couples still far off from conceiving. When Mallory Schwartz, a film and TV producer in Los Angeles, and Lauren Pietra, a marriage and family therapy associate (and Shamonki's step-daughter), talk about getting married someday, it's a package deal with talking about how they'll approach having kids. They feel there are too many variables and choices to make around family planning as a same-sex couple these days to not have those conversations simultaneously. Consumer DNA databases are already on their minds.
"It frustrates me that the DNA databases are just totally unregulated," says Schwartz. "I hope they are by the time we do this. I think everyone deserves a right to privacy when making your family [using a sperm donor]."
"I wouldn't want to create a world where people who are donor-conceived feel like they can't participate in this technology because they're trying to shut out [other] information."
On the prospect of having a donor relation pop up non-consensually for a future child, Pietra says, "I don't like it. It would be really disappointing if the child didn't want [contact], and unfortunately they're on the receiving end."
You can see how important preserving the right to keep this door closed is when you look at what's going on at The Sperm Bank of California. This pioneering cryobank was the first in the world to openly serve LGBTQ people and single women, and also the first to offer the open identification option when it opened in 1982, but not as many people are asking for their donor's identity as expected.
"We're finding a third of young people are coming forward for their donor's identity," says Alice Ruby, executive director. "We thought it would be a higher number." Viewed the other way, two-thirds of the donor-conceived people who could ethically get their donor's identity through The Sperm Bank of California are not asking the cryobank for it.
Ruby says that part of what historically made an open identification program appealing, rather than invasive or nerve-wracking, is how rigidly it's always been formatted around mutual consent, and protects against surprises for all parties. Those [donor-conceived people] who wanted more information were never barred from it, while those who wanted to remain in the dark could. No one group's wish eclipsed the other's. The potential breakdown of a system built around consent, expectations, and respect for privacy is why unregulated consumer DNA testing is most concerning to her as a path for connecting with genetic relatives.
For the last few decades in cryobanks around the world, the largest cohort of people seeking out donor sperm has been lesbian couples, followed by single women. For infertile heterosexual couples, the smallest client demographic, Ruby says donor sperm offers a solution to a medical problem, but in contrast, it historically "provided the ability for [lesbian] couples and single moms to have some reproductive autonomy." Yes, it was still a solution to a biological problem, but it was also a solution to a social one.
The Sperm Bank of California updated its registration forms to include language urging parents, donor-conceived people, and donors not to use consumer DNA tests, and to go through the cryobank if they, understandably, want to learn more about who they're connected to. But truthfully, there's not much else cryobanks can do to protect clients on any side of the donor transaction from surprise contact right now—especially not from relatives of the donor who may not even know someone in their family has donated sperm.
A Tricky Position
Personally, I've known I was donor-conceived from day one. It has never been a source of confusion, angst, or curiosity, and in fact has never loomed particularly large for me in any way. I see it merely as a type of reproductive technology—on par with in vitro fertilization—that enabled me to exist, and, now that I do exist, is irrelevant. Being confronted with my donor's identity or any donor siblings would make this fact of my conception bigger than I need it to be, as an adult with a full-blown identity derived from all of my other life experiences. But I still wonder about the minutiae of my ethnicity in much the same way as anyone else who wonders, and feel there's no safe way for me to find out without relinquishing some of my existential independence.
"People obviously want to participate in 23andMe and Ancestry because they're interested in knowing more about themselves," says Shamonki. "I wouldn't want to create a world where people who are donor-conceived feel like they can't participate in this technology because they're trying to shut out [other] information."
After all, it was the allure of that exact conceit—knowing more about oneself—that seemed to magnetically draw in millions of people to these tools in the first place. It's an experience that clearly taps into a population-wide psychic need, even—perhaps especially—if one's origins are a mystery.
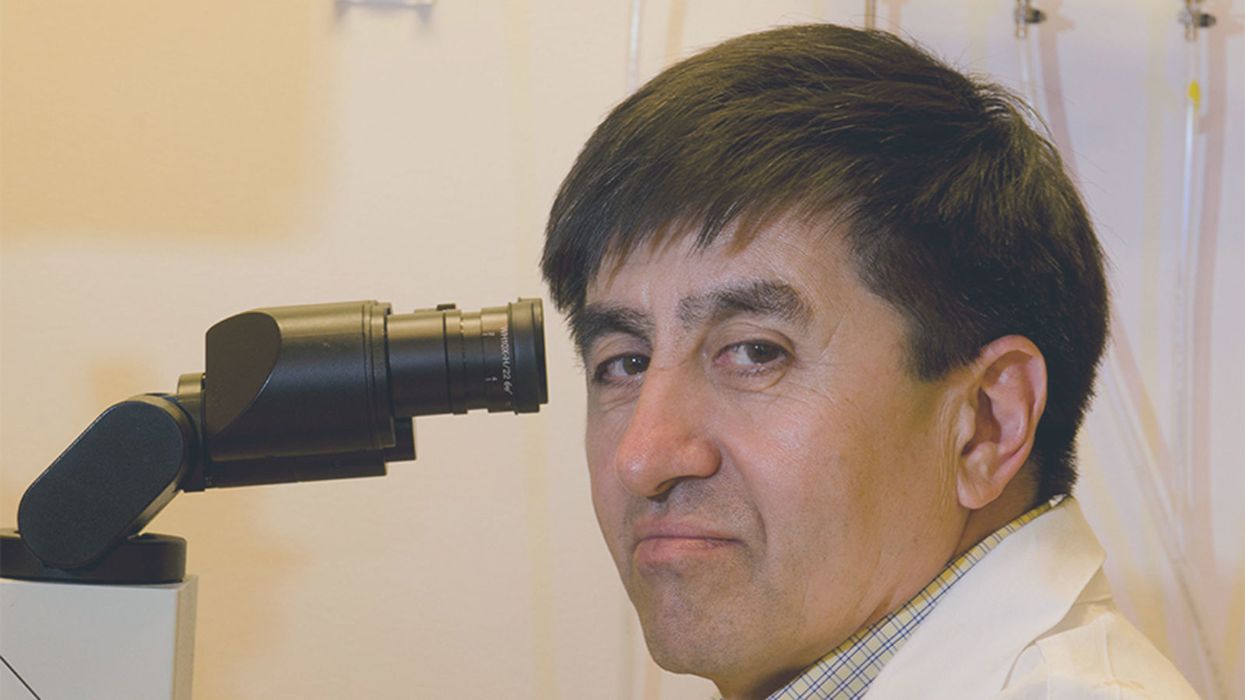
Dr. Shoukhrat Mitalipov, May 13, 2013.
Biologist Shoukhrat Mitalipov is famous—and controversial--in the world of cutting-edge fertility treatments. A decade ago, he pioneered mitochondrial replacement therapy, paving the way for the world's first "three-parent" babies to be born free of a devastating inherited disease.
He sees his work toward embryo gene therapy as not only moral, but necessary.
In 2017, he shocked the world again when his group at Oregon Health and Science University became the first to repair a genetic mutation causing heart disease in dozens of human embryos. The embryos were later destroyed a part of the experiment; current policy in the U.S. prohibits such research from moving into clinical trials.
And that policy doesn't look like it's going to change anytime soon, despite recent political wavering. Last month, a House subcommittee dropped the ban that has blocked the Food and Drug Administration since 2015 from considering any clinical trials of genetically altered embryos intended to create a baby. The move raised the hopes of supporters who want to see such research move forward and angered critics who feel that the science is getting ahead of the ethics. But yesterday, a House committee decided to restore the ban on gene-edited babies after all.
As for Mitalipov, he told leapsmag that he sees his work toward embryo gene therapy as not only moral, but necessary. This interview has been edited and condensed for clarity.
What motivates you to pursue this line of research, even though it is highly controversial?
It's my expertise, I'm an embryologist. We study early development in humans -- sperm, egg, and the first five days of development -- and try to use our knowledge to treat human diseases, particularly in that early stage. This is how IVF started, as a treatment for infertility. It's a very successful cell therapy treatment, with millions of children born. [Now the idea is] to actually to use this IVF platform not as much to treat infertility, but also to treat heritable genetic diseases, because this is a very important stage when gametes from either dad or mom will transmit mutations. This is the bottleneck where we could actually interfere and repair that mutation.
Many people are hesitant to support embryo editing because of "designer babies," yet polls do show that Americans are more open to embryo editing for the purpose of disease prevention. Where should society draw a line?
Yeah, I agree with most Americans that we don't have to edit -- meaning you could make all kind of changes. Instead we do gene repair, which is a therapeutic application.
Gene repair is quite different than gene editing. It involves [focusing on] already known disease-causing mutations and how we can turn them back to normal.
Thousands of gene mutations cause human diseases, like Crohn's, for example, or mutations causing cancer, heart disease. These are well-described, well-studied cause-and-effect diseases and we need to do something about it because otherwise it's impossible to treat once the mutation is already passed to a child.
Early intervention is the best in any disease, but in genetics, "early" means you have to do it at the time of fertilization. That's when we are dealing with one copy of the mutation or maybe two, versus when you have a whole body with billions of cells in solid tissues that we cannot really access and target. So this is the most efficient way of preventing thousands and thousands of genetic diseases. I understand that we have to make sure that it's very safe, of course, and efficient as well. But at the same time, I think this is the future. We have to work toward developing these technologies.
"If we continue banning the research everywhere and not funding it, maybe 100 years will not be enough."
What's your opinion of Dr. He Jiankui and the Chinese CRISPR'ed babies?
This is a case where he was doing gene editing, not gene repair. He hasn't corrected anything, he induced a mutation to normal human genes, hoping that this would somehow confer resistance to HIV, which is still unclear.
I think such straightforward editing is unacceptable specifically for human embryos. He's approach has also never been tested in an animal model. That's why the reaction from the public and scientists was very negative, because this is the case where the doctor does this without any expertise in this area, without knowing probably much about what he is doing, and he acquired it without any oversights, which is troubling. And of course, it negatively affects the legitimate research that is going on in some labs.
What might the future of embryo gene therapy look like?
Hopefully in 10 years from now, thousands and thousands of families that know they carry germline mutations…could go through IVF and we would correct it, and they could have healthy children.
Right now, we have some tools. We cannot correct, but we can select. So what happens is the parents become pregnant and then at about three months along, we can biopsy the amniotic fluid and say, "Hey unfortunately you passed on this mutation." And that means this child, if it's born, will be affected, so we give parents a choice of terminating the pregnancy.
Or we could do it much earlier, so parents go to the IVF clinic where we retrieve about ten eggs, after stimulating a woman's ovaries. Each of them will be fertilized so we have ten embryos that develop. We have a five-day window where we can keep them in the lab. And we basically reap a few cells, we do a biopsy from each of these ten, and we say, "Hey embryo number 1 and number 4 are not mutant, but the others are."
Then we can take these two and the other eight usually will be thrown away. That's the technology that we have now. Some ethicists argue on religious grounds that we have this selection technology available, so why do we need germline gene therapy [i.e. repairing the disease-causing mutations in an embryo]?
I don't understand the moral argument there, because all the available technology is based on selective destruction of the embryo.
With [IVF gene therapy], we will take ten embryos and every embryo we'll make healthy because we can get rid of the mutations. How could embryo destruction be morally superior?
How long do you think it will take for this technology to be available to prospective parents?
It depends how many legitimate labs with expertise can get into this field and resolve all the scientific questions. If we continue banning the research everywhere and not funding it, maybe 100 years will not be enough.
So far, I think that my lab is the only one legitimately working on it. But we would like five, 10, maybe 100 labs in this country and Europe really working. Because we have scientific challenges that we need to resolve before we could say, "Hey now we know how to correct [a given mutation] and now this could be efficient, and there are no side effects or very little." And then we could say, "Okay, I think we've done everything we could in petri dishes and in animals, and now we are ready to transplant this embryo in a patient and see what happens."
"There's just no way you could sink your head into the sand and say, 'Oh, we just ban it and then hopefully everything will go away.'"
Does banning emerging technology actually work?
Banning it usually means it will leak out to a gray area where there's no regulation and many private IVF clinics will just use it while it is still premature. So I think we have to regulate the clinical testing. There's just no way you could sink your head into the sand and say, "Oh, we just ban it and then hopefully everything will go away." That's not going to happen.
If this technology does become feasible and legal in the future, do you think that more and more couples will choose IVF and gene therapy versus the natural method of rolling the dice?
As sequencing technology is becoming available, like 23andMe, more and more parents will realize what kind of mutations they carry. And if your spouse carries the same mutation on the same locus, now you have very high chance of transmitting it. Most of the time today, we find out these families carry it once they have one or two children with that condition.
Of course, parents can just do it naturally in the bedroom and have a chance of transmitting or not transmitting mutations, but hopefully eventually we can say, "Hey, because of your condition, you don't want to play this Russian Roulette. Let's just do IVF." And hopefully the government will cover that kind of treatment because right now IVF is not covered in most states. And we do this therapy and then they have a healthy child.
We have 10,000 different mutations in the human population. That means probably billions of people carry mutations. And unless they go through this gene therapy through IVF, they will keep transmitting them. And we're going to keep having millions and millions of children with diseases. We have to do something about it.
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.
Can Soil Solve the Climate Crisis?
A child sits on the cracked earth, staring at the sun.
When Rattan Lal was awarded the Japan Prize for Biological Production, Ecology in April—the Asian equivalent of a Nobel—the audience at Tokyo's National Theatre included the emperor and empress. Lal's acceptance speech, however, was down-to-earth in the most literal sense.
Carbon, in its proper place, holds landscapes and ecosystems together.
"I'd like to begin, rather unconventionally, with the conclusion of my presentation," he told the assembled dignitaries. "And the conclusion is four words: In soil we trust."
That statement could serve as the motto for a climate crisis-fighting strategy that has gained remarkable momentum over the past five years or so—and whose rise to international prominence was reflected in that glittering award ceremony. Lal, a septuagenarian professor of soil science at Ohio State University, is one of the foremost exponents of carbon farming, an approach that centers on correcting a man-made, planetary chemical imbalance.
A Solution to Several Problems at Once?
The chemical in question is carbon. Too much of it in the atmosphere (in the form of carbon dioxide, a potent greenhouse gas) is the main driver of global heating. Too little of it in the soil is the bane of farmers in many parts of the world, and a threat to our ability to feed a ballooning global population. Advocates say agriculture can mitigate both problems—by adopting techniques that keep more soil carbon from escaping skyward, and draw more atmospheric carbon down into fields and pastures.
The potential impacts go beyond slowing climate change and boosting food production. "There are so many benefits," says Lal. "Water quality, drought, flooding, biodiversity—this is a natural solution for all these problems." That's because carbon, in its proper place, holds landscapes and ecosystems together. Plants extract it from the air and convert it into sugars for energy; they also transfer it to the soil through their roots and in the process of decomposition. In the ground, carbon feeds microbes and fungi that form the basis of complex food webs. It helps soil absorb and retain water, resist erosion, and hold onto nitrogen and phosphorous—keeping those nutrients from running off into waterways and creating toxic algal blooms.
Government and private support for research into carbon-conscious agriculture is on the rise, and growing numbers of farmers are exploring such methods. How much difference these methods can make, however, remains a matter of debate. Lal sees carbon farming as a way to buy time until CO2 emissions can be brought under control. Skeptics insist that such projections are overly optimistic. Some allies, meanwhile, think Lal's vision is too timid. "Farming can actually fix the climate," says Tim LaSalle, co-founder of the Center for Regenerative Agriculture at California State University, Chico. "That should be our only focus."
Yet Can soil solve the climate crisis? may be not be the key question in assessing the promise of carbon farming, since it implies that action is worthwhile only if a solution is ensured. A more urgent line of inquiry might be: Can the climate crisis be solved without addressing soil?
A Chance Meeting Leads to the Mission of a Lifetime
Lal was among the earliest scientists to grapple with that question. Born in Pakistan, he grew up on a tiny subsistence farm in India, where his family had fled as refugees. The only one of his siblings who learned to read and write, he attended a local agricultural university, then headed to Ohio State on scholarship for his PhD. In 1982, he was working at the International Institute of Tropical Agriculture in Nigeria, trying to develop sustainable alternatives to Africa's traditional slash-and-burn farming, when a distinguished visitor dropped by: oceanographer Roger Revelle, who 25 years earlier had published the first paper warning that fossil fuel combustion could throw the climate dangerously off-kilter.

Rattan Lal, Distinguished University Professor of Soil Science at Ohio State, received the Japan Prize at a ceremony in April.
(Photo: Ken Chamberlain. CFAES.)
Lal showed Revelle the soil in his test plots—hard and reddish, like much of Africa's agricultural land. Then (as described in Kristin Ohlson's book The Soil Will Save Us), he led the visitor to the nearby forest, where the soil was dark, soft, and wriggling with earthworms. In the forest, the soil's carbon content was 2 to 3 percent; in Lal's plots, it had dwindled to 0.5 percent. When Revelle asked him where all that carbon had gone, Lal confessed he didn't know. Revelle suggested that much of it might have floated into the atmosphere, adding to the burden of greenhouse gases. "Since then," Lal told me, "I've been looking for ways to put it back."
Back at Ohio State, Lal found that the United States Department of Agriculture (USDA) and Environmental Protection Agency (EPA) were also interested in the connection between soil carbon and climate change. With a small group of other scientists, he began investigating the dimensions of the problem, and how it might be solved.
Comparing carbon in forested and cultivated soils around the globe, the researchers calculated that about 100 billion tons had vanished into the air since the dawn of agriculture 10,000 years ago. The culprits were common practices—including plowing, overgrazing, and keeping fallow fields bare—that exposed soil carbon to oxygen, transforming it into carbon dioxide. Yet the process could also be reversed, Lal and his colleagues argued. Although there was a limit to the amount of carbon that soil could hold, they theorized that it would be possible to sequester several billion tons of global CO2 emissions each year for decades before reaching maximum capacity.
Lal set up projects on five continents to explore practices that could help accomplish that goal, such as minimizing tillage, planting cover crops, and leaving residue on fields after harvest. He organized conferences, pumped out papers and books. As other researchers launched similar efforts, policymakers worldwide took notice.
But before long, recalls Colorado State University soil scientist Keith Paustian (a fellow carbon-farming pioneer, who served with Lal on the UN's International Panel on Climate Change), official attention "kind of faded away. The bigger imperative was to cut emissions." And because agriculture accounted for only about 13 percent of greenhouse gas pollution, Paustian says, the sectors that emitted the most—energy and transportation—got the bulk of funding.
A Movement on the Rise
In recent years, however, carbon farming has begun to look like an idea whose time has come. One factor is that efforts to reduce emissions haven't worked; in 2018 alone, global CO2 output rose by an estimated 2.7 percent, according to the Global Carbon Project. Last month, researchers from the Scripps Institute of Oceanography reported that atmospheric CO2—under 350 ppm when Lal began his quest—had reached 415 ppm, the highest in 3 million years. And with the world's population expected to approach 10 billion by 2050, the need for sustainable technologies to augment food production has grown increasingly pressing.
Today, carbon-conscious methods are central to the burgeoning movement known as "regenerative agriculture," which also embraces other practices aimed at improving soil health and farming in an ecologically sound (though not always strictly organic) manner. In the United States, the latest Farm Bill includes $25 million to incentivize soil-based carbon sequestration. State and local governments across the country are supporting such efforts, as are at least a dozen nonprofits. The Department of Energy's Advanced Projects Research Agency (ARPA-e) is working to develop crops and technologies aimed at increasing soil carbon accumulation by 50 percent. General Mills recently announced plans to advance regenerative farming on 1 million acres by 2030, and many smaller companies have made their own commitments.
The toughest challenge, Lal suggests, may be persuading farmers to change their ways.
Internationally, the biggest initiative is the French-led "4 per 1,000" initiative, which aims to increase the amount of carbon in the soil of farms and rangelands worldwide by 0.4 percent per year—a rate that the project's website contends would "halt the increase of CO2 (carbon dioxide) concentration in the atmosphere related to human activities."
Given the current pace of research, Lal believes that goal—which equates to sequestering 3.6 billion tons of CO2 annually, or 10 percent of global emissions—is doable. The toughest challenge, he suggests, may be persuading farmers to change their ways. Although carbon farming can reduce costs for chemical inputs such as herbicides and fertilizers, while building rich topsoil, agriculturalists tend to be a conservative lot.
And getting low-income farmers to leave crop residue on fields, instead of using it for fuel or animal feed, will require more than speeches about melting glaciers. Lal proposes a $16 per acre subsidy, totaling $64 billion for the world's 4 billion acres of cropland. "That's not a very large amount," he says, "if you're investing in the health of the planet."
Experimental Methods Attract Supporters and Skeptics
Some experts question whether enough CO2 can be stashed in the soil to prevent the rise in average global temperature from surpassing the 2º C mark—set by the 2016 Paris Agreement as the limit beyond which climate change would become catastrophic. But others insist that carbon farming's goal should be to reverse climate change, not just to put it on pause.
"That's the only way out of this predicament," says Tim LaSalle, whose Center for Regenerative Agriculture supports the use of experimental methods ranging from multi-species cover cropping to fungal-dominant compost solutions. Using such techniques, a few researchers and farmers claim to be able to transfer carbon to the soil at rates many times higher than with established practices. Although several of these methods have yet to be documented in peer-reviewed studies, LaSalle believes they point the way forward. "We can't fix the climate, or even come close to it, using Rattan's numbers," he says, referring to Lal. "If we can replicate these experiments, we can fix it."
Even scientists sympathetic to regenerative ag warn that relying on unproven techniques is risky. "Some of these claims are beyond anything we've seen in agricultural science," says Andrew McGuire, an agronomist at Washington State University. "They could be right, but extraordinary claims require extraordinary evidence."
Still, the assorted methods currently being tested—which also include amending soil with biochar (made by heating agricultural wastes with minimal oxygen), planting long-rooted perennial crops instead of short-rooted annuals, and deploying grazing animals in ways that enrich soil rather than depleting it—offer a catalogue of hope at a time when environmental despair is all too tempting.
Last October, the National Academy of Sciences, Engineering, and Medicine issued a report acknowledging that it was too late to stave off apocalyptic overheating just by reducing CO2 emissions; removing carbon from the atmosphere would be necessary as well. The document laid out several options for doing so—most of which, it cautioned, had serious limitations.
"Soil is a bridge to the future. We can't do without it."
One possibility was planting more forests. To absorb enough carbon dioxide, however, trees might have to replace areas of farmland, reducing the food supply. Another option was creating biomass plantations to fuel power plants, whose emissions would be stored underground. But land use would be a problem: "You'd need to cover an area the size of India," explains Paustian, who was a co-author of the report. Yet another alternative was direct-air capture, in which chemical processes would be used to extract CO2 from the air. The technology was still in its infancy, though—and the costs and power requirements would likely be astronomical.
The report took up agriculture-based methods on page 95. Those needed further research as well, the authors wrote, to determine which approaches would be most effective. But of all the alternatives, this one seemed the least problematic. "Soil carbon is probably what you can do first, cheapest, and with the most additional co-benefits," says Paustian. "If we can make progress in that area, it's a huge advantage."
In any case, he and other researchers agree, we have little choice but to try. "Soil is a bridge to the future," Lal says. "We can't do without it."