New tools could catch disease outbreaks earlier - or predict them

A team at the Catalan Institution for Research and Advanced Studies is utilizing drones and weather stations to collect data on how mosquito breeding patterns are changing in response to climate shifts.
Every year, the villages which lie in the so-called ‘Nipah belt’— which stretches along the western border between Bangladesh and India, brace themselves for the latest outbreak. For since 1998, when Nipah virus—a form of hemorrhagic fever most common in Bangladesh—first spilled over into humans, it has been a grim annual visitor to the people of this region.
With a 70 percent fatality rate, no vaccine, and no known treatments, Nipah virus has been dubbed in the Western world as ‘the worst disease no one has ever heard of.’ Currently, outbreaks tend to be relatively contained because it is not very transmissible. The virus circulates throughout Asia in fruit eating bats, and only tends to be passed on to people who consume contaminated date palm sap, a sweet drink which is harvested across Bangladesh.
But as SARS-CoV-2 has shown the world, this can quickly change.
“Nipah virus is among what virologists call ‘the Big 10,’ along with things like Lassa fever and Crimean Congo hemorrhagic fever,” says Noam Ross, a disease ecologist at New York-based non-profit EcoHealth Alliance. “These are pretty dangerous viruses from a lethality perspective, which don’t currently have the capacity to spread into broader human populations. But that can evolve, and you could very well see a variant emerge that has human-human transmission capability.”
That’s not an overstatement. Surveys suggest that mammals harbour about 40,000 viruses, with roughly a quarter capable of infecting humans. The vast majority never get a chance to do so because we don’t encounter them, but climate change can alter that. Recent studies have found that as animals relocate to new habitats due to shifting environmental conditions, the coming decades will bring around 300,000 first encounters between species which normally don’t interact, especially in tropical Africa and southeast Asia. All these interactions will make it far more likely for hitherto unknown viruses to cross paths with humans.
That’s why for the last 16 years, EcoHealth Alliance has been conducting ongoing viral surveillance projects across Bangladesh. The goal is to understand why Nipah is so much more prevalent in the western part of the country, compared to the east, and keep a watchful eye out for new Nipah strains as well as other dangerous pathogens like Ebola.
"There are a lot of different infectious agents that are sensitive to climate change that don't have these sorts of software tools being developed for them," says Cat Lippi, medical geography researcher at the University of Florida.
Until very recently this kind of work has been hampered by the limitations of viral surveillance technology. The PREDICT project, a $200 million initiative funded by the United States Agency for International Development, which conducted surveillance across the Amazon Basin, Congo Basin and extensive parts of South and Southeast Asia, relied upon so-called nucleic acid assays which enabled scientists to search for the genetic material of viruses in animal samples.
However, the project came under criticism for being highly inefficient. “That approach requires a big sampling effort, because of the rarity of individual infections,” says Ross. “Any particular animal may be infected for a couple of weeks, maybe once or twice in its lifetime. So if you sample thousands and thousands of animals, you'll eventually get one that has an Ebola virus infection right now.”
Ross explains that there is now far more interest in serological sampling—the scientific term for the process of drawing blood for antibody testing. By searching for the presence of antibodies in the blood of humans and animals, scientists have a greater chance of detecting viruses which started circulating recently.
Despite the controversy surrounding EcoHealth Alliance’s involvement in so-called gain of function research—experiments that study whether viruses might mutate into deadlier strains—the organization’s separate efforts to stay one step ahead of pathogen evolution are key to stopping the next pandemic.
“Having really cheap and fast surveillance is really important,” says Ross. “Particularly in a place where there's persistent, low level, moderate infections that potentially have the ability to develop into more epidemic or pandemic situations. It means there’s a pathway that something more dangerous can come through."
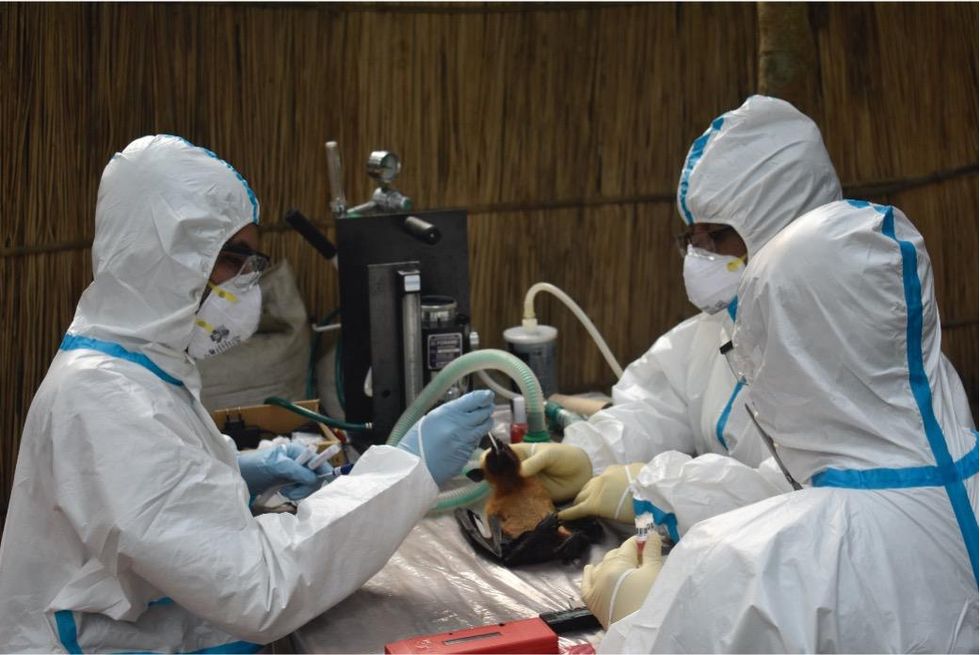
Scientists are searching for the presence of antibodies in the blood of humans and animals in hopes to detect viruses that recently started circulating.
EcoHealth Alliance
In Bangladesh, EcoHealth Alliance is attempting to do this using a newer serological technology known as a multiplex Luminex assay, which tests samples against a panel of known antibodies against many different viruses. It collects what Ross describes as a ‘footprint of information,’ which allows scientists to tell whether the sample contains the presence of a known pathogen or something completely different and needs to be investigated further.
By using this technology to sample human and animal populations across the country, they hope to gain an idea of whether there are any novel Nipah virus variants or strains from the same family, as well as other deadly viral families like Ebola.
This is just one of several novel tools being used for viral discovery in surveillance projects around the globe. Multiple research groups are taking PREDICT’s approach of looking for novel viruses in animals in various hotspots. They collect environmental DNA—mucus, faeces or shed skin left behind in soil, sediment or water—which can then be genetically sequenced.
Five years ago, this would have been a painstaking work requiring bringing collected samples back to labs. Today, thanks to the vast amounts of money spent on new technologies during COVID-19, researchers now have portable sequencing tools they can take out into the field.
Christopher Jerde, a researcher at the UC Santa Barbara Marine Science Institute, points to the Oxford Nanopore MinION sequencer as one example. “I tried one of the early versions of it four years ago, and it was miserable,” he says. “But they’ve really improved, and what we’re going to be able to do in the next five to ten years will be amazing. Instead of having to carefully transport samples back to the lab, we're going to have cigar box-shaped sequencers that we take into the field, plug into a laptop, and do the whole sequencing of an organism.”
In the past, viral surveillance has had to be very targeted and focused on known families of viruses, potentially missing new, previously unknown zoonotic pathogens. Jerde says that the rise of portable sequencers will lead to what he describes as “true surveillance.”
“Before, this was just too complex,” he says. “It had to be very focused, for example, looking for SARS-type viruses. Now we’re able to say, ‘Tell us all the viruses that are here?’ And this will give us true surveillance – we’ll be able to see the diversity of all the pathogens which are in these spots and have an understanding of which ones are coming into the population and causing damage.”
But being able to discover more viruses also comes with certain challenges. Some scientists fear that the speed of viral discovery will soon outpace the human capacity to analyze them all and assess the threat that they pose to us.
“I think we're already there,” says Jason Ladner, assistant professor at Northern Arizona University’s Pathogen and Microbiome Institute. “If you look at all the papers on the expanding RNA virus sphere, there are all of these deposited partial or complete viral sequences in groups that we just don't know anything really about yet.” Bats, for example, carry a myriad of viruses, whose ability to infect human cells we understand very poorly.
Cultivating these viruses under laboratory conditions and testing them on organoids— miniature, simplified versions of organs created from stem cells—can help with these assessments, but it is a slow and painstaking work. One hope is that in the future, machine learning could help automate this process. The new SpillOver Viral Risk Ranking platform aims to assess the risk level of a given virus based on 31 different metrics, while other computer models have tried to do the same based on the similarity of a virus’s genomic sequence to known zoonotic threats.
However, Ladner says that these types of comparisons are still overly simplistic. For one thing, scientists are still only aware of a few hundred zoonotic viruses, which is a very limited data sample for accurately assessing a novel pathogen. Instead, he says that there is a need for virologists to develop models which can determine viral compatibility with human cells, based on genomic data.
“One thing which is really useful, but can be challenging to do, is understand the cell surface receptors that a given virus might use,” he says. “Understanding whether a virus is likely to be able to use proteins on the surface of human cells to gain entry can be very informative.”
As the Earth’s climate heats up, scientists also need to better model the so-called vector borne diseases such as dengue, Zika, chikungunya and yellow fever. Transmitted by the Aedes mosquito residing in humid climates, these blights currently disproportionally affect people in low-income nations. But predictions suggest that as the planet warms and the pests find new homes, an estimated one billion people who currently don’t encounter them might be threatened by their bites by 2080. “When it comes to mosquito-borne diseases we have to worry about shifts in suitable habitat,” says Cat Lippi, a medical geography researcher at the University of Florida. “As climate patterns change on these big scales, we expect to see shifts in where people will be at risk for contracting these diseases.”
Public health practitioners and government decision-makers need tools to make climate-informed decisions about the evolving threat of different infectious diseases. Some projects are already underway. An ongoing collaboration between the Catalan Institution for Research and Advanced Studies and researchers in Brazil and Peru is utilizing drones and weather stations to collect data on how mosquitoes change their breeding patterns in response to climate shifts. This information will then be fed into computer algorithms to predict the impact of mosquito-borne illnesses on different regions.

The team at the Catalan Institution for Research and Advanced Studies is using drones and weather stations to collect data on how mosquito breeding patterns change due to climate shifts.
Gabriel Carrasco
Lippi says that similar models are urgently needed to predict how changing climate patterns affect respiratory, foodborne, waterborne and soilborne illnesses. The UK-based Wellcome Trust has allocated significant assets to fund such projects, which should allow scientists to monitor the impact of climate on a much broader range of infections. “There are a lot of different infectious agents that are sensitive to climate change that don't have these sorts of software tools being developed for them,” she says.
COVID-19’s havoc boosted funding for infectious disease research, but as its threats begin to fade from policymakers’ focus, the money may dry up. Meanwhile, scientists warn that another major infectious disease outbreak is inevitable, potentially within the next decade, so combing the planet for pathogens is vital. “Surveillance is ultimately a really boring thing that a lot of people don't want to put money into, until we have a wide scale pandemic,” Jerde says, but that vigilance is key to thwarting the next deadly horror. “It takes a lot of patience and perseverance to keep looking.”
This article originally appeared in One Health/One Planet, a single-issue magazine that explores how climate change and other environmental shifts are increasing vulnerabilities to infectious diseases by land and by sea. The magazine probes how scientists are making progress with leaders in other fields toward solutions that embrace diverse perspectives and the interconnectedness of all lifeforms and the planet.
Biologists are Growing Mini-Brains. What If They Become Conscious?
Brain coral metaphorically represents the concept of growing organoids in a dish that may eventually attain consciousness--if scientists can first determine what that means.
Few images are more uncanny than that of a brain without a body, fully sentient but afloat in sterile isolation. Such specters have spooked the speculatively-minded since the seventeenth century, when René Descartes declared, "I think, therefore I am."
Since August 29, 2019, the prospect of a bodiless but functional brain has begun to seem far less fantastical.
In Meditations on First Philosophy (1641), the French penseur spins a chilling thought experiment: he imagines "having no hands or eyes, or flesh, or blood or senses," but being tricked by a demon into believing he has all these things, and a world to go with them. A disembodied brain itself becomes a demon in the classic young-adult novel A Wrinkle in Time (1962), using mind control to subjugate a planet called Camazotz. In the sci-fi blockbuster The Matrix (1999), most of humanity endures something like Descartes' nightmare—kept in womblike pods by their computer overlords, who fill the captives' brains with a synthetized reality while tapping their metabolic energy as a power source.
Since August 29, 2019, however, the prospect of a bodiless but functional brain has begun to seem far less fantastical. On that date, researchers at the University of California, San Diego published a study in the journal Cell Stem Cell, reporting the detection of brainwaves in cerebral organoids—pea-size "mini-brains" grown in the lab. Such organoids had emitted random electrical impulses in the past, but not these complex, synchronized oscillations. "There are some of my colleagues who say, 'No, these things will never be conscious,'" lead researcher Alysson Muotri, a Brazilian-born biologist, told The New York Times. "Now I'm not so sure."

Alysson Muotri has no qualms about his creations attaining consciousness as a side effect of advancing medical breakthroughs.
(Credit: ZELMAN STUDIOS)
Muotri's findings—and his avowed ambition to push them further—brought new urgency to simmering concerns over the implications of brain organoid research. "The closer we come to his goal," said Christof Koch, chief scientist and president of the Allen Brain Institute in Seattle, "the more likely we will get a brain that is capable of sentience and feeling pain, agony, and distress." At the annual meeting of the Society for Neuroscience, researchers from the Green Neuroscience Laboratory in San Diego called for a partial moratorium, warning that the field was "perilously close to crossing this ethical Rubicon and may have already done so."
Yet experts are far from a consensus on whether brain organoids can become conscious, whether that development would necessarily be dreadful—or even how to tell if it has occurred.
So how worried do we need to be?
***
An organoid is a miniaturized, simplified version of an organ, cultured from various types of stem cells. Scientists first learned to make them in the 1980s, and have since turned out mini-hearts, lungs, kidneys, intestines, thyroids, and retinas, among other wonders. These creations can be used for everything from observation of basic biological processes to testing the effects of gene variants, pathogens, or medications. They enable researchers to run experiments that might be less accurate using animal models and unethical or impractical using actual humans. And because organoids are three-dimensional, they can yield insights into structural, developmental, and other matters that an ordinary cell culture could never provide.
In 2006, Japanese biologist Shinya Yamanaka developed a mix of proteins that turned skin cells into "pluripotent" stem cells, which could subsequently be transformed into neurons, muscle cells, or blood cells. (He later won a Nobel Prize for his efforts.) Developmental biologist Madeline Lancaster, then a post-doctoral student at the Institute of Molecular Biotechnology in Vienna, adapted that technique to grow the first brain organoids in 2013. Other researchers soon followed suit, cultivating specialized mini-brains to study disorders ranging from microcephaly to schizophrenia.
Muotri, now a youthful 45-year-old, was among the boldest of these pioneers. His team revealed the process by which Zika virus causes brain damage, and showed that sofosbuvir, a drug previously approved for hepatitis C, protected organoids from infection. He persuaded NASA to fly his organoids to the International Space Station, where they're being used to trace the impact of microgravity on neurodevelopment. He grew brain organoids using cells implanted with Neanderthal genes, and found that their wiring differed from organoids with modern DNA.
Like the latter experiment, Muotri's brainwave breakthrough emerged from a longtime obsession with neuroarchaeology. "I wanted to figure out how the human brain became unique," he told me in a phone interview. "Compared to other species, we are very social. So I looked for conditions where the social brain doesn't function well, and that led me to autism." He began investigating how gene variants associated with severe forms of the disorder affected neural networks in brain organoids.
Tinkering with chemical cocktails, Muotri and his colleagues were able to keep their organoids alive far longer than earlier versions, and to culture more diverse types of brain cells. One team member, Priscilla Negraes, devised a way to measure the mini-brains' electrical activity, by planting them in a tray lined with electrodes. By four months, the researchers found to their astonishment, normal organoids (but not those with an autism gene) emitted bursts of synchronized firing, separated by 20-second silences. At nine months, the organoids were producing up to 300,000 spikes per minute, across a range of frequencies.
He shared his vision for "brain farms," which would grow organoids en masse for drug development or tissue transplants.
When the team used an artificial intelligence system to compare these patterns with EEGs of gestating fetuses, the program found them to be nearly identical at each stage of development. As many scientists noted when the news broke, that didn't mean the organoids were conscious. (Their chaotic bursts bore little resemblance to the orderly rhythms of waking adult brains.) But to some observers, it suggested that they might be approaching the borderline.
***
Shortly after Muotri's team published their findings, I attended a conference at UCSD on the ethical questions they raised. The scientist, in jeans and a sky-blue shirt, spoke rhapsodically of brain organoids' potential to solve scientific mysteries and lead to new medical treatments. He showed video of a spider-like robot connected to an organoid through a computer interface. The machine responded to different brainwave patterns by walking or stopping—the first stage, Muotri hoped, in teaching organoids to communicate with the outside world. He described his plans to develop organoids with multiple brain regions, and to hook them up to retinal organoids so they could "see." He shared his vision for "brain farms," which would grow organoids en masse for drug development or tissue transplants.

Muotri holds a spider-like robot that can connect to an organoid through a computer interface.
(Credit: ROLAND LIZARONDO/KPBS)
Yet Muotri also stressed the current limitations of the technology. His organoids contain approximately 2 million neurons, compared to about 200 million in a rat's brain and 86 billion in an adult human's. They consist only of a cerebral cortex, and lack many of a real brain's cell types. Because researchers haven't yet found a way to give organoids blood vessels, moreover, nutrients can't penetrate their inner recesses—a severe constraint on their growth.
Another panelist strongly downplayed the imminence of any Rubicon. Patricia Churchland, an eminent philosopher of neuroscience, cited research suggesting that in mammals, networked connections between the cortex and the thalamus are a minimum requirement for consciousness. "It may be a blessing that you don't have the enabling conditions," she said, "because then you don't have the ethical issues."
Christof Koch, for his part, sounded much less apprehensive than the Times had made him seem. He noted that science lacks a definition of consciousness, beyond an organism's sense of its own existence—"the fact that it feels like something to be you or me." As to the competing notions of how the phenomenon arises, he explained, he prefers one known as Integrated Information Theory, developed by neuroscientist Giulio Tononi. IIT considers consciousness to be a quality intrinsic to systems that reach a certain level of complexity, integration, and causal power (the ability for present actions to determine future states). By that standard, Koch doubted that brain organoids had stepped over the threshold.
One way to tell, he said, might be to use the "zap and zip" test invented by Tononi and his colleague Marcello Massimini in the early 2000s to determine whether patients are conscious in the medical sense. This technique zaps the brain with a pulse of magnetic energy, using a coil held to the scalp. As loops of neural impulses cascade through the cerebral circuitry, an EEG records the firing patterns. In a waking brain, the feedback is highly complex—neither totally predictable nor totally random. In other states, such as sleep, coma, or anesthesia, the rhythms are simpler. Applying an algorithm commonly used for computer "zip" files, the researchers devised a scale that allowed them to correctly diagnose most patients who were minimally conscious or in a vegetative state.
If scientists could find a way to apply "zap and zip" to brain organoids, Koch ventured, it should be possible to rank their degree of awareness on a similar scale. And if it turned out that an organoid was conscious, he added, our ethical calculations should strive to minimize suffering, and avoid it where possible—just as we now do, or ought to, with animal subjects. (Muotri, I later learned, was already contemplating sensors that would signal when organoids were likely in distress.)
During the question-and-answer period, an audience member pressed Churchland about how her views might change if the "enabling conditions" for consciousness in brain organoids were to arise. "My feeling is, we'll answer that when we get there," she said. "That's an unsatisfying answer, but it's because I don't know. Maybe they're totally happy hanging out in a dish! Maybe that's the way to be."
***
Muotri himself admits to no qualms about his creations attaining consciousness, whether sooner or later. "I think we should try to replicate the model as close as possible to the human brain," he told me after the conference. "And if that involves having a human consciousness, we should go in that direction." Still, he said, if strong evidence of sentience does arise, "we should pause and discuss among ourselves what to do."
"The field is moving so rapidly, you blink your eyes and another advance has occurred."
Churchland figures it will be at least a decade before anyone reaches the crossroads. "That's partly because the thalamus has a very complex architecture," she said. It might be possible to mimic that architecture in the lab, she added, "but I tend to think it's not going to be a piece of cake."
If anything worries Churchland about brain organoids, in fact, it's that Muotri's visionary claims for their potential could set off a backlash among those who find them unacceptably spooky. "Alysson has done brilliant work, and he's wonderfully charismatic and charming," she said. "But then there's that guy back there who doesn't think it's exciting; he thinks you're the Devil incarnate. You're playing into the hands of people who are going to shut you down."
Koch, however, is more willing to indulge Muotri's dreams. "Ten years ago," he said, "nobody would have believed you can take a stem cell and get an entire retina out of it. It's absolutely frigging amazing. So who am I to say the same thing can't be true for the thalamus or the cortex? The field is moving so rapidly, you blink your eyes and another advance has occurred."
The point, he went on, is not to build a Cartesian thought experiment—or a Matrix-style dystopia—but to vanquish some of humankind's most terrifying foes. "You know, my dad passed away of Parkinson's. I had a twin daughter; she passed away of sudden death syndrome. One of my best friends killed herself; she was schizophrenic. We want to eliminate all these terrible things, and that requires experimentation. We just have to go into it with open eyes."
Harvard Scientist’s Breakthrough Could Make Humans Resistant to All Viruses
DNA recoding has already made E. coli bacteria immune to 90 percent of viruses that can infect it.
[Ed. Note: We're thrilled to present the first episode in our new Moonshot series, which will explore four cutting-edge scientific developments that stand to fundamentally transform our world.]
Kira Peikoff was the editor-in-chief of Leaps.org from 2017 to 2021. As a journalist, her work has appeared in The New York Times, Newsweek, Nautilus, Popular Mechanics, The New York Academy of Sciences, and other outlets. She is also the author of four suspense novels that explore controversial issues arising from scientific innovation: Living Proof, No Time to Die, Die Again Tomorrow, and Mother Knows Best. Peikoff holds a B.A. in Journalism from New York University and an M.S. in Bioethics from Columbia University. She lives in New Jersey with her husband and two young sons. Follow her on Twitter @KiraPeikoff.