New tools could catch disease outbreaks earlier - or predict them

A team at the Catalan Institution for Research and Advanced Studies is utilizing drones and weather stations to collect data on how mosquito breeding patterns are changing in response to climate shifts.
Every year, the villages which lie in the so-called ‘Nipah belt’— which stretches along the western border between Bangladesh and India, brace themselves for the latest outbreak. For since 1998, when Nipah virus—a form of hemorrhagic fever most common in Bangladesh—first spilled over into humans, it has been a grim annual visitor to the people of this region.
With a 70 percent fatality rate, no vaccine, and no known treatments, Nipah virus has been dubbed in the Western world as ‘the worst disease no one has ever heard of.’ Currently, outbreaks tend to be relatively contained because it is not very transmissible. The virus circulates throughout Asia in fruit eating bats, and only tends to be passed on to people who consume contaminated date palm sap, a sweet drink which is harvested across Bangladesh.
But as SARS-CoV-2 has shown the world, this can quickly change.
“Nipah virus is among what virologists call ‘the Big 10,’ along with things like Lassa fever and Crimean Congo hemorrhagic fever,” says Noam Ross, a disease ecologist at New York-based non-profit EcoHealth Alliance. “These are pretty dangerous viruses from a lethality perspective, which don’t currently have the capacity to spread into broader human populations. But that can evolve, and you could very well see a variant emerge that has human-human transmission capability.”
That’s not an overstatement. Surveys suggest that mammals harbour about 40,000 viruses, with roughly a quarter capable of infecting humans. The vast majority never get a chance to do so because we don’t encounter them, but climate change can alter that. Recent studies have found that as animals relocate to new habitats due to shifting environmental conditions, the coming decades will bring around 300,000 first encounters between species which normally don’t interact, especially in tropical Africa and southeast Asia. All these interactions will make it far more likely for hitherto unknown viruses to cross paths with humans.
That’s why for the last 16 years, EcoHealth Alliance has been conducting ongoing viral surveillance projects across Bangladesh. The goal is to understand why Nipah is so much more prevalent in the western part of the country, compared to the east, and keep a watchful eye out for new Nipah strains as well as other dangerous pathogens like Ebola.
"There are a lot of different infectious agents that are sensitive to climate change that don't have these sorts of software tools being developed for them," says Cat Lippi, medical geography researcher at the University of Florida.
Until very recently this kind of work has been hampered by the limitations of viral surveillance technology. The PREDICT project, a $200 million initiative funded by the United States Agency for International Development, which conducted surveillance across the Amazon Basin, Congo Basin and extensive parts of South and Southeast Asia, relied upon so-called nucleic acid assays which enabled scientists to search for the genetic material of viruses in animal samples.
However, the project came under criticism for being highly inefficient. “That approach requires a big sampling effort, because of the rarity of individual infections,” says Ross. “Any particular animal may be infected for a couple of weeks, maybe once or twice in its lifetime. So if you sample thousands and thousands of animals, you'll eventually get one that has an Ebola virus infection right now.”
Ross explains that there is now far more interest in serological sampling—the scientific term for the process of drawing blood for antibody testing. By searching for the presence of antibodies in the blood of humans and animals, scientists have a greater chance of detecting viruses which started circulating recently.
Despite the controversy surrounding EcoHealth Alliance’s involvement in so-called gain of function research—experiments that study whether viruses might mutate into deadlier strains—the organization’s separate efforts to stay one step ahead of pathogen evolution are key to stopping the next pandemic.
“Having really cheap and fast surveillance is really important,” says Ross. “Particularly in a place where there's persistent, low level, moderate infections that potentially have the ability to develop into more epidemic or pandemic situations. It means there’s a pathway that something more dangerous can come through."
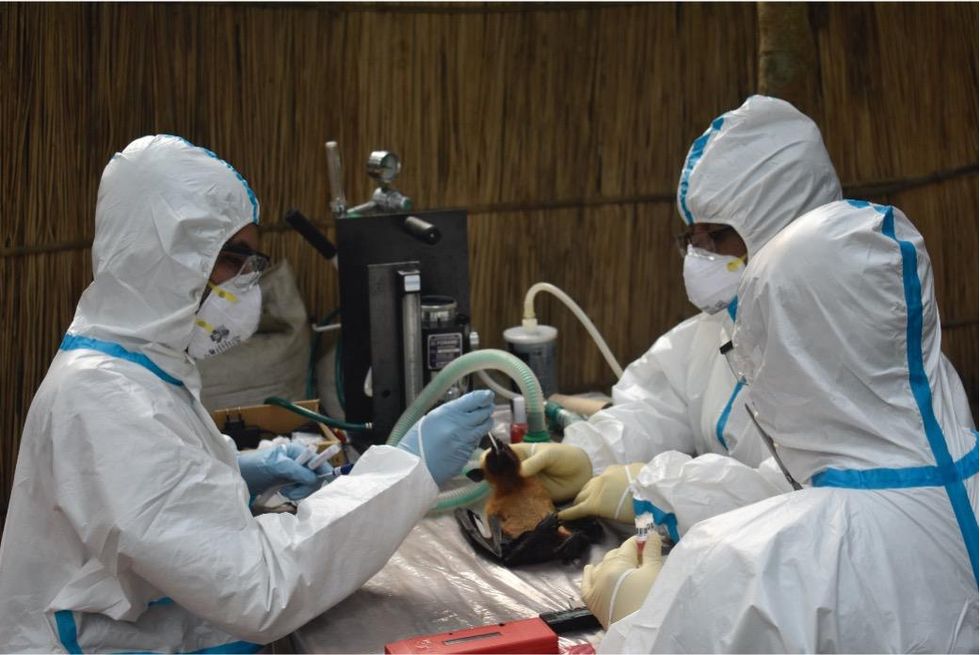
Scientists are searching for the presence of antibodies in the blood of humans and animals in hopes to detect viruses that recently started circulating.
EcoHealth Alliance
In Bangladesh, EcoHealth Alliance is attempting to do this using a newer serological technology known as a multiplex Luminex assay, which tests samples against a panel of known antibodies against many different viruses. It collects what Ross describes as a ‘footprint of information,’ which allows scientists to tell whether the sample contains the presence of a known pathogen or something completely different and needs to be investigated further.
By using this technology to sample human and animal populations across the country, they hope to gain an idea of whether there are any novel Nipah virus variants or strains from the same family, as well as other deadly viral families like Ebola.
This is just one of several novel tools being used for viral discovery in surveillance projects around the globe. Multiple research groups are taking PREDICT’s approach of looking for novel viruses in animals in various hotspots. They collect environmental DNA—mucus, faeces or shed skin left behind in soil, sediment or water—which can then be genetically sequenced.
Five years ago, this would have been a painstaking work requiring bringing collected samples back to labs. Today, thanks to the vast amounts of money spent on new technologies during COVID-19, researchers now have portable sequencing tools they can take out into the field.
Christopher Jerde, a researcher at the UC Santa Barbara Marine Science Institute, points to the Oxford Nanopore MinION sequencer as one example. “I tried one of the early versions of it four years ago, and it was miserable,” he says. “But they’ve really improved, and what we’re going to be able to do in the next five to ten years will be amazing. Instead of having to carefully transport samples back to the lab, we're going to have cigar box-shaped sequencers that we take into the field, plug into a laptop, and do the whole sequencing of an organism.”
In the past, viral surveillance has had to be very targeted and focused on known families of viruses, potentially missing new, previously unknown zoonotic pathogens. Jerde says that the rise of portable sequencers will lead to what he describes as “true surveillance.”
“Before, this was just too complex,” he says. “It had to be very focused, for example, looking for SARS-type viruses. Now we’re able to say, ‘Tell us all the viruses that are here?’ And this will give us true surveillance – we’ll be able to see the diversity of all the pathogens which are in these spots and have an understanding of which ones are coming into the population and causing damage.”
But being able to discover more viruses also comes with certain challenges. Some scientists fear that the speed of viral discovery will soon outpace the human capacity to analyze them all and assess the threat that they pose to us.
“I think we're already there,” says Jason Ladner, assistant professor at Northern Arizona University’s Pathogen and Microbiome Institute. “If you look at all the papers on the expanding RNA virus sphere, there are all of these deposited partial or complete viral sequences in groups that we just don't know anything really about yet.” Bats, for example, carry a myriad of viruses, whose ability to infect human cells we understand very poorly.
Cultivating these viruses under laboratory conditions and testing them on organoids— miniature, simplified versions of organs created from stem cells—can help with these assessments, but it is a slow and painstaking work. One hope is that in the future, machine learning could help automate this process. The new SpillOver Viral Risk Ranking platform aims to assess the risk level of a given virus based on 31 different metrics, while other computer models have tried to do the same based on the similarity of a virus’s genomic sequence to known zoonotic threats.
However, Ladner says that these types of comparisons are still overly simplistic. For one thing, scientists are still only aware of a few hundred zoonotic viruses, which is a very limited data sample for accurately assessing a novel pathogen. Instead, he says that there is a need for virologists to develop models which can determine viral compatibility with human cells, based on genomic data.
“One thing which is really useful, but can be challenging to do, is understand the cell surface receptors that a given virus might use,” he says. “Understanding whether a virus is likely to be able to use proteins on the surface of human cells to gain entry can be very informative.”
As the Earth’s climate heats up, scientists also need to better model the so-called vector borne diseases such as dengue, Zika, chikungunya and yellow fever. Transmitted by the Aedes mosquito residing in humid climates, these blights currently disproportionally affect people in low-income nations. But predictions suggest that as the planet warms and the pests find new homes, an estimated one billion people who currently don’t encounter them might be threatened by their bites by 2080. “When it comes to mosquito-borne diseases we have to worry about shifts in suitable habitat,” says Cat Lippi, a medical geography researcher at the University of Florida. “As climate patterns change on these big scales, we expect to see shifts in where people will be at risk for contracting these diseases.”
Public health practitioners and government decision-makers need tools to make climate-informed decisions about the evolving threat of different infectious diseases. Some projects are already underway. An ongoing collaboration between the Catalan Institution for Research and Advanced Studies and researchers in Brazil and Peru is utilizing drones and weather stations to collect data on how mosquitoes change their breeding patterns in response to climate shifts. This information will then be fed into computer algorithms to predict the impact of mosquito-borne illnesses on different regions.

The team at the Catalan Institution for Research and Advanced Studies is using drones and weather stations to collect data on how mosquito breeding patterns change due to climate shifts.
Gabriel Carrasco
Lippi says that similar models are urgently needed to predict how changing climate patterns affect respiratory, foodborne, waterborne and soilborne illnesses. The UK-based Wellcome Trust has allocated significant assets to fund such projects, which should allow scientists to monitor the impact of climate on a much broader range of infections. “There are a lot of different infectious agents that are sensitive to climate change that don't have these sorts of software tools being developed for them,” she says.
COVID-19’s havoc boosted funding for infectious disease research, but as its threats begin to fade from policymakers’ focus, the money may dry up. Meanwhile, scientists warn that another major infectious disease outbreak is inevitable, potentially within the next decade, so combing the planet for pathogens is vital. “Surveillance is ultimately a really boring thing that a lot of people don't want to put money into, until we have a wide scale pandemic,” Jerde says, but that vigilance is key to thwarting the next deadly horror. “It takes a lot of patience and perseverance to keep looking.”
This article originally appeared in One Health/One Planet, a single-issue magazine that explores how climate change and other environmental shifts are increasing vulnerabilities to infectious diseases by land and by sea. The magazine probes how scientists are making progress with leaders in other fields toward solutions that embrace diverse perspectives and the interconnectedness of all lifeforms and the planet.
In the future, a paper strip reminiscent of a pregnancy test could be used to quickly diagnose the flu and other infectious diseases.
Trying to get a handle on CRISPR news in 2019 can be daunting if you haven't been avidly reading up on it for the last five years.
CRISPR as a diagnostic tool would be a major game changer for medicine and agriculture.
On top of trying to grasp how the science works, and keeping track of its ever expanding applications, you may also have seen coverage of an ongoing legal battle about who owns the intellectual property behind the gene-editing technology CRISPR-Cas9. And then there's the infamous controversy surrounding a scientist who claimed to have used the tool to edit the genomes of two babies in China last year.
But gene editing is not the only application of CRISPR-based biotechnologies. In the future, it may also be used as a tool to diagnose infectious diseases, which could be a major game changer for medicine and agriculture.
How It Works
CRISPR is an acronym for a naturally occurring DNA sequence that normally protects microbes from viruses. It's been compared to a Swiss army knife that can recognize an invader's DNA and precisely destroy it. Repurposed for humans, CRISPR can be paired with a protein called Cas9 that can detect a person's own DNA sequence (usually a problematic one), cut it out, and replace it with a different sequence. Used this way, CRISPR-Cas9 has become a valuable gene-editing tool that is currently being tested to treat numerous genetic diseases, from cancer to blood disorders to blindness.
CRISPR can also be paired with other proteins, like Cas13, which target RNA, the single-stranded twin of DNA that viruses rely on to infect their hosts and cause disease. In a future clinical setting, CRISPR-Cas13 might be used to diagnose whether you have the flu by cutting a target RNA sequence from the virus. That spliced sequence could stick to a paper test strip, causing a band to show up, like on a pregnancy test strip. If the influenza virus and its RNA are not present, no band would show up.
To understand how close to reality this diagnostic scenario is right now, leapsmag chatted with CRISPR pioneer Dr. Feng Zhang, a molecular biologist at the Broad Institute of MIT and Harvard.
What do you think might be the first point of contact that a regular person or patient would have with a CRISPR diagnostic tool?
FZ: I think in the long run it will be great to see this for, say, at-home disease testing, for influenza and other sorts of important public health [concerns]. To be able to get a readout at home, people can potentially quarantine themselves rather than traveling to a hospital and then carrying the risk of spreading that disease to other people as they get to the clinic.
"You could conceivably get a readout during the same office visit, and then the doctor will be able to prescribe the right treatment right away."
Is this just something that people will use at home, or do you also foresee clinical labs at hospitals applying CRISPR-Cas13 to samples that come through?
FZ: I think we'll see applications in both settings, and I think there are advantages to both. One of the nice things about SHERLOCK [a playful acronym for CRISPR-Cas13's longer name, Specific High-sensitivity Enzymatic Reporter unLOCKing] is that it's rapid; you can get a readout fairly quickly. So, right now, what people do in hospitals is they will collect your sample and then they'll send it out to a clinical testing lab, so you wouldn't get a result back until many hours if not several days later. With SHERLOCK, you could conceivably get a readout during the same office visit, and then the doctor will be able to prescribe the right treatment right away.
I just want to clarify that when you say a doctor would take a sample, that's referring to urine, blood, or saliva, correct?
FZ: Right. Yeah, exactly.
Thinking more long term, are there any Holy Grail applications that you hope CRISPR reaches as a diagnostic tool?
FZ: I think in the developed world we'll hopefully see this being used for influenza testing, and many other viral and pathogen-based diseases—both at home and also in the hospital—but I think the even more exciting direction is that this could be used and deployed in parts of the developing world where there isn't a fancy laboratory with elaborate instrumentation. SHERLOCK is relatively inexpensive to develop, and you can turn it into a paper strip test.
Can you quantify what you mean by relatively inexpensive? What range of prices are we talking about here?
FZ: So without accounting for economies of scale, we estimate that it can cost less than a dollar per test. With economy of scale that cost can go even lower.
Is there value in developing what is actually quite an innovative tool in a way that visually doesn't seem innovative because it's reminiscent of a pregnancy test? And I don't mean that as an insult.
FZ: [Laughs] Ultimately, we want the technology to be as accessible as possible, and pregnancy test strips have such a convenient and easy-to-use form. I think modeling after something that people are already familiar with and just changing what's under the hood makes a lot of sense.

Feng Zhang
(Photo credit: Justin Knight, McGovern Institute)
It's probably one of the most accessible at-home diagnostic tools at this point that people are familiar with.
FZ: Yeah, so if people know how to use that, then using something that's very similar to it should make the option very easy.
You've been quite vocal in calling for some pauses in CRISPR-Cas9 research to make sure it doesn't outpace the ethics of establishing pregnancies with that version of the tool. Do you have any concerns about using CRISPR-Cas13 as a diagnostic tool?
I think overall, the reception for CRISPR-based diagnostics has been overwhelmingly positive. People are very excited about the prospect of using this—for human health and also in agriculture [for] detection of plant infections and plant pathogens, so that farmers will be able to react quickly to infection in the field. If we're looking at contamination of foods by certain bacteria, [food safety] would also be a really exciting application.
Do you feel like the controversies surrounding using CRISPR as a gene-editing tool have overshadowed its potential as a diagnostics tool?
FZ: I don't think so. I think the potential for using CRISPR-Cas9 or CRISPR-Cas12 for gene therapy, and treating disease, has captured people's imaginations, but at the same time, every time I talk with someone about the ability to use CRISPR-Cas13 as a diagnostic tool, people are equally excited. Especially when people see the very simple paper strip that we developed for detecting diseases.
Are CRISPR as a gene-editing tool and CRISPR as a diagnostics tool on different timelines, as far as when the general public might encounter them in their real lives?
FZ: I think they are all moving forward quite quickly. CRISPR as a gene-editing tool is already being deployed in human health and agriculture. We've already seen the approval for the development of growing genome-edited mushrooms, soybeans, and other crop species. So I think people will encounter those in their daily lives in that manner.
Then, of course, for disease treatment, that's progressing rapidly as well. For patients who are affected by sickle cell disease, and also by a degenerative eye disease, clinical trials are already starting in those two areas. Diagnostic tests are also developing quickly, and I think in the coming couple of years, we'll begin to see some of these reaching into the public realm.
"There are probably 7,000 genetic diseases identified today, and most of them don't have any way of being treated."
As far its limits, will it be hard to use CRISPR as a diagnostic tool in situations where we don't necessarily understand the biological underpinnings of a disease?
FZ: CRISPR-Cas13, as a diagnostic tool, at least in the current way that it's implemented, is a detection tool—it's not a discovery tool. So if we don't know what we're looking for, then it's going to be hard to develop Cas13 to detect it. But even in the case of a new infectious disease, if DNA sequencing or RNA sequencing information is available for that new virus, then we can very rapidly program a Cas13-based system to detect it, based on that sequence.
What's something you think the public misunderstands about CRISPR, either in general, or specifically as a diagnostic tool, that you wish were better understood?
FZ: That's a good question. CRISPR-Cas9 and CRISPR-Cas12 as gene editing tools, and also CRISPR-Cas13 as a diagnostic tool, are able to do some things, but there are still a lot of capabilities that need to be further developed. So I think the potential for the technology will unfold over the next decade or so, but it will take some time for the full impact of the technology to really get realized in real life.
What do you think that full impact is?
FZ: There are probably 7,000 genetic diseases identified today, and most of them don't have any way of being treated. It will take some time for CRISPR-Cas9 and Cas12 to be really developed for addressing a larger number of those diseases. And then for CRISPR-based diagnostics, I think you'll see the technology being applied in a couple of initial cases, and it will take some time to develop that more broadly for many other applications.
Researchers Are Experimenting With Magic Mushrooms' Fascinating Ability to Improve Mental Health Disorders
Magic mushrooms, in conjunction with a psychotherapist's treatment, may be helpful in treating addiction, depression, anxiety, and other mental health ailments.
Mental illness is a dark undercurrent in the lives of tens of millions of Americans. According to the World Health Organization, about 450 million people worldwide have a mental health disorder, which cut across all demographics, cultures, and socioeconomic classes.
One area of research seems to herald the first major breakthrough in decades — hallucinogen-assisted psychotherapy.
The U.S. National Institute on Mental Health estimates that severely debilitating mental health disorders cost the U.S. more than $300 billion per year, and that's not even counting the human toll of broken lives, devastated families, and a health care system stretched to the limit.
However, one area of research seems to herald the first major breakthrough in decades — hallucinogen-assisted psychotherapy. Drugs like psilocybin (obtained from "magic mushrooms"), LSD, and MDMA (known as the club drug, ecstasy) are being tested in combination with talk therapy for a variety of mental illnesses. These drugs, administered by a psychotherapist in a safe and controlled environment, are showing extraordinary results that other conventional treatments would take years to accomplish.
But the therapy will likely continue to face an uphill legal battle before it achieves FDA approval. It is up against not only current drug laws (all psychedelics remain illegal on the federal level) and strict FDA regulations, but a powerful status quo that has institutionalized fear of any drug used for recreational purposes.
How We Got Here
According to researchers Sean Belouin and Jack Henningfield, the use of psychedelic drugs has a long and winding history. It's believed that hallucinogenic substances have been used in healing ceremonies and religious rituals for thousands of years. Indigenous people in the U.S., Mexico, and Central and South America still use distillations from the peyote cactus and other hallucinogens in their religious ceremonies. And psilocybin mushrooms, also capable of causing hallucinations, grow throughout the world and are thought to have been used for millennia.
But psychedelic drugs didn't receive much research until 1943, when LSD's psychoactive effects were discovered by chemist Albert Hoffman. Hoffman tested the compound he had discovered years earlier on himself and found that the drug had profound mind-altering effects. He made the drug available to psychiatrists who were interested in testing it out as an adjunct to talk therapy. There were no truly effective drugs at the time for mental illnesses, and psychiatrists early on saw the possibility of psychedelics providing a kind of emotional catharsis that might represent therapeutic breakthroughs for many mental conditions.
During the 1950s and early 1960s, psychedelic drugs saw an increase in use within psychology, according to a 2018 article in Neuropharmacology. During this time, research on LSD and other hallucinogens was the subject of over 1,000 scientific papers, six international conferences, and several dozen books. LSD was widely prescribed to psychiatric patients, and by 1958, Hoffman had identified psilocybin as the hallucinogenic in "magic mushrooms," which was also administered. By 1965 some type of hallucinogenic had been given to more than 40,000 patients.
Then came a sea change. Psychedelic drugs caught the public's attention and there was widespread experimentation. The association with Hippie counterculture alarmed many and led to a legal and cultural backlash that stigmatized psychedelics for decades to come. In the mid-1960s, psychedelics were designated Schedule 1 drugs in the U.S., meaning they were seen as having "no accepted medical use and a high potential of abuse." Schedule 1 also implied that the drugs were more dangerous than cocaine, methamphetamine, Vicodin, and oxycodone, a perception that was far from proven but became an institutionalized part of drug enforcement. Medical use ceased and research dwindled down to close to zero.
For years, research into hallucinogenic-assisted therapy was basically dormant, until the 1990s when interest started to revive. In the 2000s, the first modern clinical trials of psilocybin were done by Francisco Moreno at the University of Arizona and Matthew Johnson at Johns Hopkins. Scientists in the 2010s, including Robin Carhart-Harris, started studying the use of psychedelics in the treatment of major depressive disorder (MDD).
In small trials with these patients, results showed significant and long-term improvement (for at least six months) after only two episodes of psilocybin-assisted therapy. In several studies, the guided experience of administering one of the psychedelic drugs along with psychotherapy seemed to result in marked improvement in a variety of disorders, including depression, anxiety, PTSD, and addiction.
The drugs allowed patients to experience a radical reframing of reality, helping them to become "unstuck" from the anxious and negative tape loops that played in their heads. According to Michael Pollan, an American author and professor of journalism who wrote the book, "How to Change Your Mind: What the New Science of Psychedelics Teaches Us About Consciousness, Dying, Addiction, Depression and Transcendence," psychedelics allow patients to see their lives through a kind of wide angle, where boundaries vanish and they're able to experience "consciousness without self." This perspective is usually accompanied by profound feelings of oneness with the universe.
Pollan likens the effect to a fresh blanketing of snow over the deep ruts of unproductive thinking, which characterize depression and other mental disorders. Once the new snow has fallen, the ruts disappear and a new path can be chosen. Relief from symptoms comes immediately, and in numerous studies, is sustained for months.
In spite of growing evidence for the safety and efficacy of psychedelic-assisted psychotherapy, the practice has major hurdles to cross on its quest for FDA approval.
Some of the most influential studies have focused on testing the use of psilocybin to treat end-of-life anxiety in patients diagnosed with a terminal illness. In 2016, Stephen Ross and colleagues tested a single dose of psilocybin on 29 subjects with end-of-life anxiety due to a terminal cancer diagnosis. A control group received a niacin pill. The researchers reported that of the 29 receiving psilocybin, all of the patients had "immediate, substantial, and sustained clinical benefits," even after six months.
In spite of growing evidence for the safety and efficacy of psychedelic-assisted psychotherapy, the practice has major hurdles to cross on its quest for FDA approval. The National Institutes of Health is not currently supporting any clinical trials and the research relies on private sources of funding, often with small research organizations that cannot afford the high cost of clinical trials.
Given the controversial nature of the drugs, researchers in psychedelic-assisted therapies may be cautious about publicity. Leapsmag reached out to several leaders in the field but none agreed to an interview.
Looking Ahead
Still, interest is building in the combination of psychedelic drugs and psychotherapy for treatment-resistant mental illnesses. Two months ago, Johns Hopkins University launched a new psychedelic research center with an infusion of $17 million from private investors. The center will focus on psychedelic-assisted therapies for opioid addiction, Alzheimer's disease, PTSD and major depression, to name just a few. Currently, of 51 cancer patients enrolled in a Hopkins study, more than half reported a decrease in depression and anxiety after receiving therapy with psilocybin. Two thirds even claimed that the experience was one of the most meaningful of their lives.
It is not unheard of for Schedule 1 drugs to make their way into medical use if they're shown to provide a bonafide improvement in a medical condition through well-designed clinical trials. MDMA, for example, has been designated a Breakthrough Therapy by the FDA as part of an Investigational New Drug Application. The FDA has agreed to a special protocol assessment that could speed up phase three clinical trials. The next step is for the data to be submitted to the FDA for an in-depth regulatory review. If the FDA agrees, MDMA-assisted therapy could be legalized.
Will the positive buzz around psychedelics persuade the NIH to provide the millions of dollars needed to push the field forward?
Robin Carhart-Harris believes the first drug that will receive FDA clearance is psilocybin, which he speculates could become legal in the next five to ten years. However, the field of psychedelic-assisted therapy needs more and larger clinical trials, preferably with the support of the NIH.
As Rucker and colleagues noted, the scientific literature bends toward the theme that the drugs are not necessarily therapeutic in and of themselves. It's the use of hallucinogens within a "psychologically supportive context" with a trained expert that's helpful. It's currently unknown how many users of recreational drugs are self-medicating for depression, anxiety, or other mental illnesses. But without the guidance of a knowledgeable psychotherapist, those who are self-medicating may not be helping themselves at all.
Will the positive buzz around psychedelics persuade the NIH to provide the millions of dollars needed to push the field forward? Given the changing climate in public opinion around these drugs and the need for breakthroughs in mental health therapies, it's possible that in the foreseeable future, this bold new therapy will become part of the mental health arsenal.