New tools could catch disease outbreaks earlier - or predict them

A team at the Catalan Institution for Research and Advanced Studies is utilizing drones and weather stations to collect data on how mosquito breeding patterns are changing in response to climate shifts.
Every year, the villages which lie in the so-called ‘Nipah belt’— which stretches along the western border between Bangladesh and India, brace themselves for the latest outbreak. For since 1998, when Nipah virus—a form of hemorrhagic fever most common in Bangladesh—first spilled over into humans, it has been a grim annual visitor to the people of this region.
With a 70 percent fatality rate, no vaccine, and no known treatments, Nipah virus has been dubbed in the Western world as ‘the worst disease no one has ever heard of.’ Currently, outbreaks tend to be relatively contained because it is not very transmissible. The virus circulates throughout Asia in fruit eating bats, and only tends to be passed on to people who consume contaminated date palm sap, a sweet drink which is harvested across Bangladesh.
But as SARS-CoV-2 has shown the world, this can quickly change.
“Nipah virus is among what virologists call ‘the Big 10,’ along with things like Lassa fever and Crimean Congo hemorrhagic fever,” says Noam Ross, a disease ecologist at New York-based non-profit EcoHealth Alliance. “These are pretty dangerous viruses from a lethality perspective, which don’t currently have the capacity to spread into broader human populations. But that can evolve, and you could very well see a variant emerge that has human-human transmission capability.”
That’s not an overstatement. Surveys suggest that mammals harbour about 40,000 viruses, with roughly a quarter capable of infecting humans. The vast majority never get a chance to do so because we don’t encounter them, but climate change can alter that. Recent studies have found that as animals relocate to new habitats due to shifting environmental conditions, the coming decades will bring around 300,000 first encounters between species which normally don’t interact, especially in tropical Africa and southeast Asia. All these interactions will make it far more likely for hitherto unknown viruses to cross paths with humans.
That’s why for the last 16 years, EcoHealth Alliance has been conducting ongoing viral surveillance projects across Bangladesh. The goal is to understand why Nipah is so much more prevalent in the western part of the country, compared to the east, and keep a watchful eye out for new Nipah strains as well as other dangerous pathogens like Ebola.
"There are a lot of different infectious agents that are sensitive to climate change that don't have these sorts of software tools being developed for them," says Cat Lippi, medical geography researcher at the University of Florida.
Until very recently this kind of work has been hampered by the limitations of viral surveillance technology. The PREDICT project, a $200 million initiative funded by the United States Agency for International Development, which conducted surveillance across the Amazon Basin, Congo Basin and extensive parts of South and Southeast Asia, relied upon so-called nucleic acid assays which enabled scientists to search for the genetic material of viruses in animal samples.
However, the project came under criticism for being highly inefficient. “That approach requires a big sampling effort, because of the rarity of individual infections,” says Ross. “Any particular animal may be infected for a couple of weeks, maybe once or twice in its lifetime. So if you sample thousands and thousands of animals, you'll eventually get one that has an Ebola virus infection right now.”
Ross explains that there is now far more interest in serological sampling—the scientific term for the process of drawing blood for antibody testing. By searching for the presence of antibodies in the blood of humans and animals, scientists have a greater chance of detecting viruses which started circulating recently.
Despite the controversy surrounding EcoHealth Alliance’s involvement in so-called gain of function research—experiments that study whether viruses might mutate into deadlier strains—the organization’s separate efforts to stay one step ahead of pathogen evolution are key to stopping the next pandemic.
“Having really cheap and fast surveillance is really important,” says Ross. “Particularly in a place where there's persistent, low level, moderate infections that potentially have the ability to develop into more epidemic or pandemic situations. It means there’s a pathway that something more dangerous can come through."
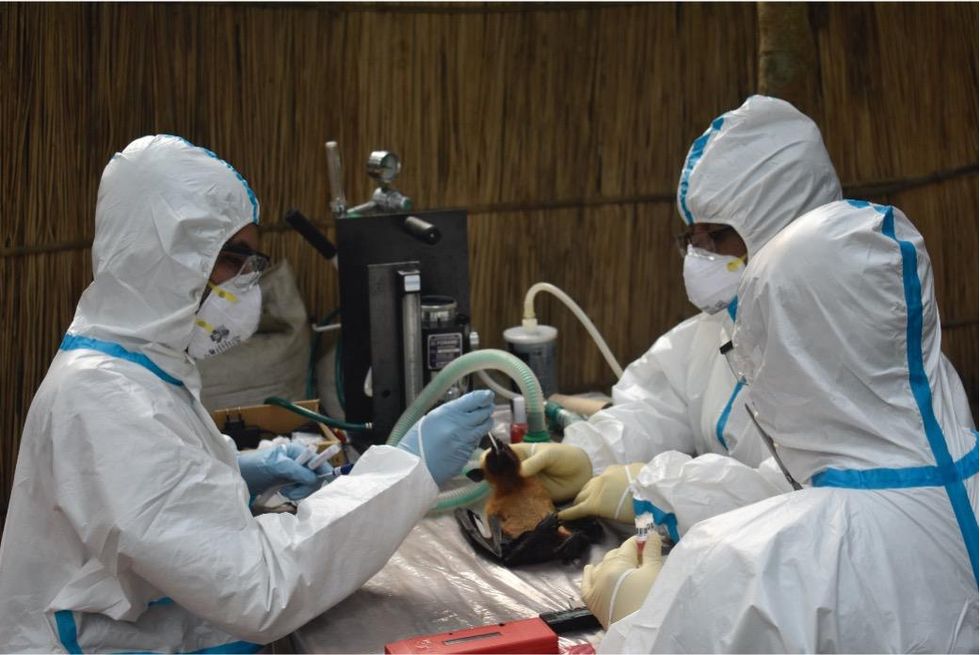
Scientists are searching for the presence of antibodies in the blood of humans and animals in hopes to detect viruses that recently started circulating.
EcoHealth Alliance
In Bangladesh, EcoHealth Alliance is attempting to do this using a newer serological technology known as a multiplex Luminex assay, which tests samples against a panel of known antibodies against many different viruses. It collects what Ross describes as a ‘footprint of information,’ which allows scientists to tell whether the sample contains the presence of a known pathogen or something completely different and needs to be investigated further.
By using this technology to sample human and animal populations across the country, they hope to gain an idea of whether there are any novel Nipah virus variants or strains from the same family, as well as other deadly viral families like Ebola.
This is just one of several novel tools being used for viral discovery in surveillance projects around the globe. Multiple research groups are taking PREDICT’s approach of looking for novel viruses in animals in various hotspots. They collect environmental DNA—mucus, faeces or shed skin left behind in soil, sediment or water—which can then be genetically sequenced.
Five years ago, this would have been a painstaking work requiring bringing collected samples back to labs. Today, thanks to the vast amounts of money spent on new technologies during COVID-19, researchers now have portable sequencing tools they can take out into the field.
Christopher Jerde, a researcher at the UC Santa Barbara Marine Science Institute, points to the Oxford Nanopore MinION sequencer as one example. “I tried one of the early versions of it four years ago, and it was miserable,” he says. “But they’ve really improved, and what we’re going to be able to do in the next five to ten years will be amazing. Instead of having to carefully transport samples back to the lab, we're going to have cigar box-shaped sequencers that we take into the field, plug into a laptop, and do the whole sequencing of an organism.”
In the past, viral surveillance has had to be very targeted and focused on known families of viruses, potentially missing new, previously unknown zoonotic pathogens. Jerde says that the rise of portable sequencers will lead to what he describes as “true surveillance.”
“Before, this was just too complex,” he says. “It had to be very focused, for example, looking for SARS-type viruses. Now we’re able to say, ‘Tell us all the viruses that are here?’ And this will give us true surveillance – we’ll be able to see the diversity of all the pathogens which are in these spots and have an understanding of which ones are coming into the population and causing damage.”
But being able to discover more viruses also comes with certain challenges. Some scientists fear that the speed of viral discovery will soon outpace the human capacity to analyze them all and assess the threat that they pose to us.
“I think we're already there,” says Jason Ladner, assistant professor at Northern Arizona University’s Pathogen and Microbiome Institute. “If you look at all the papers on the expanding RNA virus sphere, there are all of these deposited partial or complete viral sequences in groups that we just don't know anything really about yet.” Bats, for example, carry a myriad of viruses, whose ability to infect human cells we understand very poorly.
Cultivating these viruses under laboratory conditions and testing them on organoids— miniature, simplified versions of organs created from stem cells—can help with these assessments, but it is a slow and painstaking work. One hope is that in the future, machine learning could help automate this process. The new SpillOver Viral Risk Ranking platform aims to assess the risk level of a given virus based on 31 different metrics, while other computer models have tried to do the same based on the similarity of a virus’s genomic sequence to known zoonotic threats.
However, Ladner says that these types of comparisons are still overly simplistic. For one thing, scientists are still only aware of a few hundred zoonotic viruses, which is a very limited data sample for accurately assessing a novel pathogen. Instead, he says that there is a need for virologists to develop models which can determine viral compatibility with human cells, based on genomic data.
“One thing which is really useful, but can be challenging to do, is understand the cell surface receptors that a given virus might use,” he says. “Understanding whether a virus is likely to be able to use proteins on the surface of human cells to gain entry can be very informative.”
As the Earth’s climate heats up, scientists also need to better model the so-called vector borne diseases such as dengue, Zika, chikungunya and yellow fever. Transmitted by the Aedes mosquito residing in humid climates, these blights currently disproportionally affect people in low-income nations. But predictions suggest that as the planet warms and the pests find new homes, an estimated one billion people who currently don’t encounter them might be threatened by their bites by 2080. “When it comes to mosquito-borne diseases we have to worry about shifts in suitable habitat,” says Cat Lippi, a medical geography researcher at the University of Florida. “As climate patterns change on these big scales, we expect to see shifts in where people will be at risk for contracting these diseases.”
Public health practitioners and government decision-makers need tools to make climate-informed decisions about the evolving threat of different infectious diseases. Some projects are already underway. An ongoing collaboration between the Catalan Institution for Research and Advanced Studies and researchers in Brazil and Peru is utilizing drones and weather stations to collect data on how mosquitoes change their breeding patterns in response to climate shifts. This information will then be fed into computer algorithms to predict the impact of mosquito-borne illnesses on different regions.

The team at the Catalan Institution for Research and Advanced Studies is using drones and weather stations to collect data on how mosquito breeding patterns change due to climate shifts.
Gabriel Carrasco
Lippi says that similar models are urgently needed to predict how changing climate patterns affect respiratory, foodborne, waterborne and soilborne illnesses. The UK-based Wellcome Trust has allocated significant assets to fund such projects, which should allow scientists to monitor the impact of climate on a much broader range of infections. “There are a lot of different infectious agents that are sensitive to climate change that don't have these sorts of software tools being developed for them,” she says.
COVID-19’s havoc boosted funding for infectious disease research, but as its threats begin to fade from policymakers’ focus, the money may dry up. Meanwhile, scientists warn that another major infectious disease outbreak is inevitable, potentially within the next decade, so combing the planet for pathogens is vital. “Surveillance is ultimately a really boring thing that a lot of people don't want to put money into, until we have a wide scale pandemic,” Jerde says, but that vigilance is key to thwarting the next deadly horror. “It takes a lot of patience and perseverance to keep looking.”
This article originally appeared in One Health/One Planet, a single-issue magazine that explores how climate change and other environmental shifts are increasing vulnerabilities to infectious diseases by land and by sea. The magazine probes how scientists are making progress with leaders in other fields toward solutions that embrace diverse perspectives and the interconnectedness of all lifeforms and the planet.
The Stunning Comeback of a Top Transplant Surgeon Who Got a New Heart at His Own Hospital
Dr. Robert Montgomery, almost one year post-transplant, at a vineyard in Casablanca, Chile, August 2019.
Having spent my working life as a transplant surgeon, it is the ultimate irony that I have now become a heart transplant patient. I knew this was a possibility since 1987, when I was 27 years old and I received a phone call from my sister-in-law telling me that my 35-year-old brother, Rich, had just died suddenly while water skiing.
Living from one heartbeat to the next I knew I had to get it right and nail my life—and in that regard my disease was a blessing.
After his autopsy, dots were connected and it was clear that the mysterious heart disease my father had died from when I was 15 years old was genetic. I was evaluated and it was clear that I too had inherited cardiomyopathy, a progressive weakening condition of the heart muscle that often leads to dangerous rhythm disturbances and sudden death. My doctors urged me to have a newly developed device called an implantable cardioverter-defibrillator (ICD) surgically placed in my abdomen and chest to monitor and shock my heart back into normal rhythm should I have a sudden cardiac arrest.
They also told me I was the first surgeon in the world to undergo an ICD implant and that having one of these devices would not be compatible with the life of a surgeon and I should change careers to something less rigorous. With the support of a mentor and armed with what the British refer to as my "bloody-mindedness," I refused to give up this dream of becoming a transplant surgeon. I completed my surgical training and embarked on my career.
What followed were periods of stability punctuated by near-death experiences. I had a family, was productive in my work, and got on with life, knowing that this was a fragile situation that could turn on its head in a moment. In a way, it made my decisions about how to spend my time and focus my efforts more deliberate and purposeful. Living from one heartbeat to the next I knew I had to get it right and nail my life—and in that regard my disease was a blessing.
In 2017 while pursuing my passion for the outdoors in a remote part of Patagonia, I collapsed from bacterial pneumonia and sepsis. Unknowingly, I had brought in my lungs one of those super-bugs that you read about from the hospital where I worked. Several days into the trip, the bacteria entered my blood stream and brought me as close to death as a human can get.
I lay for nearly 3 weeks in a coma on a stretcher in a tiny hospital in Argentina, septic and in cardiogenic shock before stabilizing enough to be evaced to NYU Langone Hospital, where I was on staff. I awoke helpless, unable to walk, talk, or swallow food or drink. It was a long shot but I managed to recover completely from this episode; after 3 months, I returned to work and the operating room. My heart rebounded, but never back to where it had been.
Then, on the eve of my mother's funeral, I arrested while watching a Broadway show, and this time my ICD failed to revive me. There was prolonged CPR that broke my ribs and spine and a final shock that recaptured my heart. It was literally a show stopper and I awoke to a standing ovation from the New York theatre audience who were stunned by my modern recreation of the biblical story of Lazarus, or for the more hip among them, my real-life rendition of the resurrection of Jon Snow at the end of season 5 of Game of Thrones.
Against the advice of my doctors, I attended my mom's funeral and again tried to regain some sense of normalcy. We discussed a transplant at this point but, believe it or not, there is such a scarcity of organs I was not yet "sick enough" to get enough priority to receive a heart. I had more surgery to supercharge my ICD so it would be more likely to save my life the next time -- and there would be a next time, I knew.
As a transplant surgeon, I have been involved in some important innovations to expand the number of organs available for transplantation.
Months later in Matera, Italy, where I was attending a medical meeting, I developed what is referred to as ventricular tachycardia storm. I had 4 cardiac arrests over a 3-hour period. With the first one, I fell on to a stone floor and split my forehead open. When I arrived at the small hospital it seemed like Patagonia all over again. One of the first people I met was a Catholic priest who gave me the Last Rights.
I knew now was the moment and so with the help of one of my colleagues who was at the meeting with me and the compassion of the Italian doctors who supplied my friend with resuscitation medications and left my IV in place, I signed out of the hospital against medical advice and boarded a commercial flight back to New York. I was admitted to the NYU intensive care unit and received a heart transplant 3 weeks later.
Now, what I haven't said is that as a transplant surgeon, I have been involved in some important innovations to expand the number of organs available for transplantation. I came to NYU in 2016 to start a new Transplant Institute which included inaugurating a heart transplant program. We hired heart transplant surgeons, cardiologists, and put together a team that unbeknownst to me at the time, would save my life a year later.
It gets even more interesting. One of the innovations that I had been involved in from its inception in the 1990s was using organs from donors at risk for transmitting viruses like HIV and Hepatitis C (Hep C). We popularized new ways to detect these viruses in donors and ensure that the risk was minimized as much as possible so patients in need of a life-saving transplant could utilize these organs.
When the opioid crisis hit hard about four years ago, there were suddenly a lot of potential donors who were IV drug users and 25 percent of them were known to be infected with Hep C (which is spread by needles). In 2018, 49,000 people died in the U.S. from drug overdoses. There were many more donors with Hep C than potential recipients who had previously been exposed to Hep C, and so more than half of these otherwise perfectly good organs were being discarded. At the same time, a new class of drugs was being tested that could cure Hep C.
I was at Johns Hopkins at the time and our team developed a protocol for using these Hep C positive organs for Hep C negative recipients who were willing to take them, even knowing that they were likely to become infected with the virus. We would then treat them after the transplant with this new class of drugs and in all likelihood, cure them. I brought this protocol with me to NYU.
When my own time came, I accepted a Hep C heart from a donor who overdosed on heroin. I became infected with Hep C and it was then eliminated from my body with 2 months of anti-viral therapy. All along this unlikely journey, I was seemingly making decisions that would converge upon that moment in time when I would arise to catch the heart that was meant for me.

Dr. Montgomery with his wife Denyce Graves, September 2019.
(Courtesy Montgomery)
Today, I am almost exactly one year post-transplant, back to work, operating, traveling, enjoying the outdoors, and giving lectures. My heart disease is gone; gone when my heart was removed. Gone also is my ICD. I am no longer at risk for a sudden cardiac death. I traded all that for the life of a transplant patient, which has its own set of challenges, but I clearly traded up. It is cliché, I know, but I enjoy every moment of every day. It is a miracle I am still here.
Scientists Just Created Liquid Solar Power That Can Be Stored for 18 Years
A team of Swedish scientists has gathered solar power so pure, that until recently, capturing it was an impossibility.
Look no further than this week's climate strikes for evidence that millions of people are passionate about curbing global warming.
Unlike relatively limited solar panel energy storage, norbornadiene can potentially maintain its potency for years.
But even potential solutions, like alternative meats, have their own challenges. Some scientists are putting their focus on the sun to help balance out our energy consumption.
In fact, they are gathering solar power so pure that, until recently, capturing it was an impossibility.
The Lowdown
A group of Swedish scientists has created a liquid called norbornadiene. This liquid sunshine can capture up to 30 percent of raw solar power. To put it in perspective, the best publicly available solar panels can harness 21 percent. Norbornadiene would bring in about 50 percent more power – a significant difference in energy efficiency.
Most notably, unlike relatively limited solar panel energy storage, norbornadiene can potentially maintain its potency for years. We could have the ability to collect and store premium solar power, making it easier for current and future generations to use fossil and nuclear fuel alternatives.
"The norbornadiene molecules that we have made have very good properties, in terms of solar energy capture efficiency, storage time and energy density," says team lead Dr. Kasper Moth-Poulson of the Chamlers University of Technology. "They can store energy without the need for insulation materials for 18 or more years."
Next Up
Swedish scientist Moth-Poulsen and his team have been testing the norbornadiene on the physics building roof at the Chalmers University of Technology. Once activated, it heats up to just below boiling and provides enough power to be useful.
The energy density is 250 watt-hours per kilogram, twice the strength of Tesla's popular Powerall battery.
It requires potentially toxic solvents, like a cobalt-based activator, to transform into its full potential. The team is currently trying to find less-hazardous catalysts to help transform the norbornadiene to its active form, quadricyclane. Exposing it to sunlight is the main way to reactivate the norbornadiene's power. Over time, scientists will likely make it more efficient with less toxic agents.
The energy density is 250 watt-hours per kilogram, twice the strength of Tesla's popular Powerall battery.
Open Questions
The biggest question is safety, perceived or otherwise: Are you ready to drive around with 250 kWh of pure solar in your Hyundai? Norbornadiene may be stable in a hermetically sealed lab, but sculpting it for everyday use requires another level of security.
The half-life of the sunshine power is also an estimate, too. The challenge with new scientific substances is you don't know how the matter will evolve over time. It is easy to be overly optimistic about this one discovery being the key to our energy needs. For the time being, it is wiser to look at norbornadiene as a progressive step rather than a revolutionary one.
Even at its least effective, norbornadiene and its related material is a step toward us utilizing the one natural resource that won't run out for generations. In the short-term, a stable form of it could offset our fossil and nuclear fuel use and even help lower the carbon footprint made by long-distance transportation. It will be fascinating to see what future aircraft builders, home designers and even car manufacturers do as the solar technology conversation heats up.
Moth-Poulsen wants norbornadiene to be a definitive part of the climate change puzzle.
"I hope that in five years, we will see the first products based on our molecules and could help mitigate the daily variations in temperature," he says. "This will lead to increased thermal comfort and reduced energy consumption for heating and cooling."

