New tools could catch disease outbreaks earlier - or predict them

A team at the Catalan Institution for Research and Advanced Studies is utilizing drones and weather stations to collect data on how mosquito breeding patterns are changing in response to climate shifts.
Every year, the villages which lie in the so-called ‘Nipah belt’— which stretches along the western border between Bangladesh and India, brace themselves for the latest outbreak. For since 1998, when Nipah virus—a form of hemorrhagic fever most common in Bangladesh—first spilled over into humans, it has been a grim annual visitor to the people of this region.
With a 70 percent fatality rate, no vaccine, and no known treatments, Nipah virus has been dubbed in the Western world as ‘the worst disease no one has ever heard of.’ Currently, outbreaks tend to be relatively contained because it is not very transmissible. The virus circulates throughout Asia in fruit eating bats, and only tends to be passed on to people who consume contaminated date palm sap, a sweet drink which is harvested across Bangladesh.
But as SARS-CoV-2 has shown the world, this can quickly change.
“Nipah virus is among what virologists call ‘the Big 10,’ along with things like Lassa fever and Crimean Congo hemorrhagic fever,” says Noam Ross, a disease ecologist at New York-based non-profit EcoHealth Alliance. “These are pretty dangerous viruses from a lethality perspective, which don’t currently have the capacity to spread into broader human populations. But that can evolve, and you could very well see a variant emerge that has human-human transmission capability.”
That’s not an overstatement. Surveys suggest that mammals harbour about 40,000 viruses, with roughly a quarter capable of infecting humans. The vast majority never get a chance to do so because we don’t encounter them, but climate change can alter that. Recent studies have found that as animals relocate to new habitats due to shifting environmental conditions, the coming decades will bring around 300,000 first encounters between species which normally don’t interact, especially in tropical Africa and southeast Asia. All these interactions will make it far more likely for hitherto unknown viruses to cross paths with humans.
That’s why for the last 16 years, EcoHealth Alliance has been conducting ongoing viral surveillance projects across Bangladesh. The goal is to understand why Nipah is so much more prevalent in the western part of the country, compared to the east, and keep a watchful eye out for new Nipah strains as well as other dangerous pathogens like Ebola.
"There are a lot of different infectious agents that are sensitive to climate change that don't have these sorts of software tools being developed for them," says Cat Lippi, medical geography researcher at the University of Florida.
Until very recently this kind of work has been hampered by the limitations of viral surveillance technology. The PREDICT project, a $200 million initiative funded by the United States Agency for International Development, which conducted surveillance across the Amazon Basin, Congo Basin and extensive parts of South and Southeast Asia, relied upon so-called nucleic acid assays which enabled scientists to search for the genetic material of viruses in animal samples.
However, the project came under criticism for being highly inefficient. “That approach requires a big sampling effort, because of the rarity of individual infections,” says Ross. “Any particular animal may be infected for a couple of weeks, maybe once or twice in its lifetime. So if you sample thousands and thousands of animals, you'll eventually get one that has an Ebola virus infection right now.”
Ross explains that there is now far more interest in serological sampling—the scientific term for the process of drawing blood for antibody testing. By searching for the presence of antibodies in the blood of humans and animals, scientists have a greater chance of detecting viruses which started circulating recently.
Despite the controversy surrounding EcoHealth Alliance’s involvement in so-called gain of function research—experiments that study whether viruses might mutate into deadlier strains—the organization’s separate efforts to stay one step ahead of pathogen evolution are key to stopping the next pandemic.
“Having really cheap and fast surveillance is really important,” says Ross. “Particularly in a place where there's persistent, low level, moderate infections that potentially have the ability to develop into more epidemic or pandemic situations. It means there’s a pathway that something more dangerous can come through."
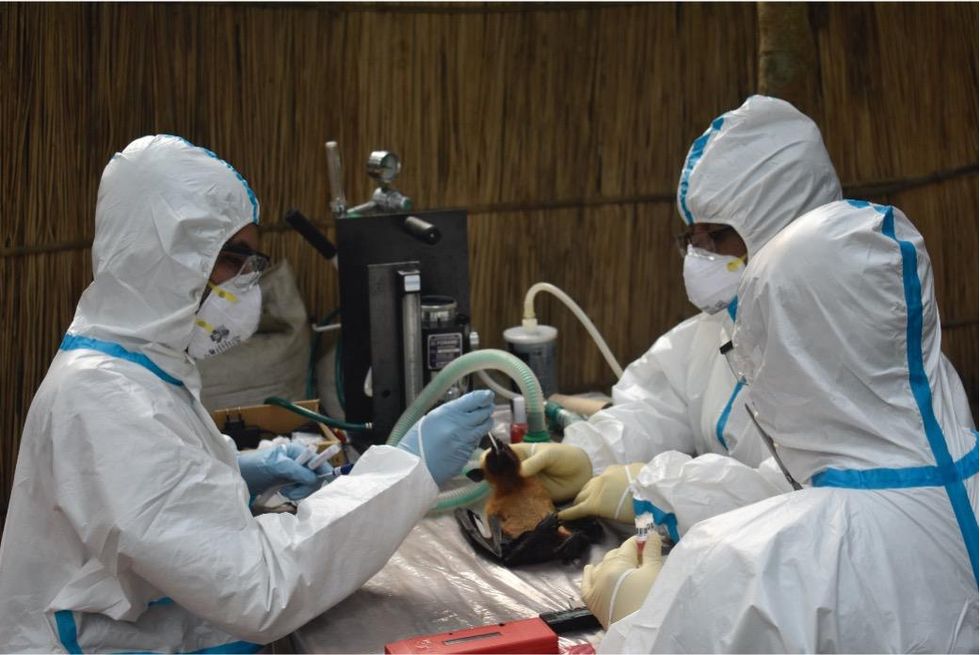
Scientists are searching for the presence of antibodies in the blood of humans and animals in hopes to detect viruses that recently started circulating.
EcoHealth Alliance
In Bangladesh, EcoHealth Alliance is attempting to do this using a newer serological technology known as a multiplex Luminex assay, which tests samples against a panel of known antibodies against many different viruses. It collects what Ross describes as a ‘footprint of information,’ which allows scientists to tell whether the sample contains the presence of a known pathogen or something completely different and needs to be investigated further.
By using this technology to sample human and animal populations across the country, they hope to gain an idea of whether there are any novel Nipah virus variants or strains from the same family, as well as other deadly viral families like Ebola.
This is just one of several novel tools being used for viral discovery in surveillance projects around the globe. Multiple research groups are taking PREDICT’s approach of looking for novel viruses in animals in various hotspots. They collect environmental DNA—mucus, faeces or shed skin left behind in soil, sediment or water—which can then be genetically sequenced.
Five years ago, this would have been a painstaking work requiring bringing collected samples back to labs. Today, thanks to the vast amounts of money spent on new technologies during COVID-19, researchers now have portable sequencing tools they can take out into the field.
Christopher Jerde, a researcher at the UC Santa Barbara Marine Science Institute, points to the Oxford Nanopore MinION sequencer as one example. “I tried one of the early versions of it four years ago, and it was miserable,” he says. “But they’ve really improved, and what we’re going to be able to do in the next five to ten years will be amazing. Instead of having to carefully transport samples back to the lab, we're going to have cigar box-shaped sequencers that we take into the field, plug into a laptop, and do the whole sequencing of an organism.”
In the past, viral surveillance has had to be very targeted and focused on known families of viruses, potentially missing new, previously unknown zoonotic pathogens. Jerde says that the rise of portable sequencers will lead to what he describes as “true surveillance.”
“Before, this was just too complex,” he says. “It had to be very focused, for example, looking for SARS-type viruses. Now we’re able to say, ‘Tell us all the viruses that are here?’ And this will give us true surveillance – we’ll be able to see the diversity of all the pathogens which are in these spots and have an understanding of which ones are coming into the population and causing damage.”
But being able to discover more viruses also comes with certain challenges. Some scientists fear that the speed of viral discovery will soon outpace the human capacity to analyze them all and assess the threat that they pose to us.
“I think we're already there,” says Jason Ladner, assistant professor at Northern Arizona University’s Pathogen and Microbiome Institute. “If you look at all the papers on the expanding RNA virus sphere, there are all of these deposited partial or complete viral sequences in groups that we just don't know anything really about yet.” Bats, for example, carry a myriad of viruses, whose ability to infect human cells we understand very poorly.
Cultivating these viruses under laboratory conditions and testing them on organoids— miniature, simplified versions of organs created from stem cells—can help with these assessments, but it is a slow and painstaking work. One hope is that in the future, machine learning could help automate this process. The new SpillOver Viral Risk Ranking platform aims to assess the risk level of a given virus based on 31 different metrics, while other computer models have tried to do the same based on the similarity of a virus’s genomic sequence to known zoonotic threats.
However, Ladner says that these types of comparisons are still overly simplistic. For one thing, scientists are still only aware of a few hundred zoonotic viruses, which is a very limited data sample for accurately assessing a novel pathogen. Instead, he says that there is a need for virologists to develop models which can determine viral compatibility with human cells, based on genomic data.
“One thing which is really useful, but can be challenging to do, is understand the cell surface receptors that a given virus might use,” he says. “Understanding whether a virus is likely to be able to use proteins on the surface of human cells to gain entry can be very informative.”
As the Earth’s climate heats up, scientists also need to better model the so-called vector borne diseases such as dengue, Zika, chikungunya and yellow fever. Transmitted by the Aedes mosquito residing in humid climates, these blights currently disproportionally affect people in low-income nations. But predictions suggest that as the planet warms and the pests find new homes, an estimated one billion people who currently don’t encounter them might be threatened by their bites by 2080. “When it comes to mosquito-borne diseases we have to worry about shifts in suitable habitat,” says Cat Lippi, a medical geography researcher at the University of Florida. “As climate patterns change on these big scales, we expect to see shifts in where people will be at risk for contracting these diseases.”
Public health practitioners and government decision-makers need tools to make climate-informed decisions about the evolving threat of different infectious diseases. Some projects are already underway. An ongoing collaboration between the Catalan Institution for Research and Advanced Studies and researchers in Brazil and Peru is utilizing drones and weather stations to collect data on how mosquitoes change their breeding patterns in response to climate shifts. This information will then be fed into computer algorithms to predict the impact of mosquito-borne illnesses on different regions.

The team at the Catalan Institution for Research and Advanced Studies is using drones and weather stations to collect data on how mosquito breeding patterns change due to climate shifts.
Gabriel Carrasco
Lippi says that similar models are urgently needed to predict how changing climate patterns affect respiratory, foodborne, waterborne and soilborne illnesses. The UK-based Wellcome Trust has allocated significant assets to fund such projects, which should allow scientists to monitor the impact of climate on a much broader range of infections. “There are a lot of different infectious agents that are sensitive to climate change that don't have these sorts of software tools being developed for them,” she says.
COVID-19’s havoc boosted funding for infectious disease research, but as its threats begin to fade from policymakers’ focus, the money may dry up. Meanwhile, scientists warn that another major infectious disease outbreak is inevitable, potentially within the next decade, so combing the planet for pathogens is vital. “Surveillance is ultimately a really boring thing that a lot of people don't want to put money into, until we have a wide scale pandemic,” Jerde says, but that vigilance is key to thwarting the next deadly horror. “It takes a lot of patience and perseverance to keep looking.”
This article originally appeared in One Health/One Planet, a single-issue magazine that explores how climate change and other environmental shifts are increasing vulnerabilities to infectious diseases by land and by sea. The magazine probes how scientists are making progress with leaders in other fields toward solutions that embrace diverse perspectives and the interconnectedness of all lifeforms and the planet.
Genes that protect health with Dr. Nir Barzilai
Centenarians essentially won the genetic lottery, says Nir Barzilai of Albert Einstein College of Medicine. He is studying their genes to see how the rest of us can benefit from understanding how they work.
In today’s podcast episode, I talk with Nir Barzilai, a geroscientist, which means he studies the biology of aging. Barzilai directs the Institute for Aging Research at the Albert Einstein College of Medicine.
My first question for Dr. Barzilai was: why do we age? And is there anything to be done about it? His answers were encouraging. We can’t live forever, but we have some control over the process, as he argues in his book, Age Later.
Dr. Barzilai told me that centenarians differ from the rest of us because they have unique gene mutations that help them stay healthy longer. For most of us, the words “gene mutations” spell trouble - we associate these words with cancer or neurodegenerative diseases, but apparently not all mutations are bad.
Listen on Apple | Listen on Spotify | Listen on Stitcher | Listen on Amazon | Listen on Google
Centenarians may have essentially won the genetic lottery, but that doesn’t mean the rest of us are predestined to have a specific lifespan and health span, or the amount of time spent living productively and enjoyably. “Aging is a mother of all diseases,” Dr. Barzilai told me. And as a disease, it can be targeted by therapeutics. Dr. Barzilai’s team is already running clinical trials on such therapeutics — and the results are promising.
More about Dr. Barzilai: He is scientific director of AFAR, American Federation for Aging Research. As part of his work, Dr. Barzilai studies families of centenarians and their genetics to learn how the rest of us can learn and benefit from their super-aging. He also organizing a clinical trial to test a specific drug that may slow aging.
Show Links
Age Later: Health Span, Life Span, and the New Science of Longevity https://www.amazon.com/Age-Later-Healthiest-Sharpest-Centenarians/dp/1250230853
American Federation for Aging Research https://www.afar.org
https://www.afar.org/nir-barzilai
https://www.einsteinmed.edu/faculty/484/nir-barzilai/
Metformin as a Tool to Target Aging
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5943638/
Benefits of Metformin in Attenuating the Hallmarks of Aging https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7347426/
The Longevity Genes Project https://www.einsteinmed.edu/centers/aging/longevity-genes-project/
Lina Zeldovich has written about science, medicine and technology for Popular Science, Smithsonian, National Geographic, Scientific American, Reader’s Digest, the New York Times and other major national and international publications. A Columbia J-School alumna, she has won several awards for her stories, including the ASJA Crisis Coverage Award for Covid reporting, and has been a contributing editor at Nautilus Magazine. In 2021, Zeldovich released her first book, The Other Dark Matter, published by the University of Chicago Press, about the science and business of turning waste into wealth and health. You can find her on http://linazeldovich.com/ and @linazeldovich.
Often called the window to the soul, the eyes are more sacred than other body parts, at least for some.
Awash in a fluid finely calibrated to keep it alive, a human eye rests inside a transparent cubic device. This ECaBox, or Eyes in a Care Box, is a one-of-a-kind system built by scientists at Barcelona’s Centre for Genomic Regulation (CRG). Their goal is to preserve human eyes for transplantation and related research.
In recent years, scientists have learned to transplant delicate organs such as the liver, lungs or pancreas, but eyes are another story. Even when preserved at the average transplant temperature of 4 Centigrade, they last for 48 hours max. That's one explanation for why transplanting the whole eye isn’t possible—only the cornea, the dome-shaped, outer layer of the eye, can withstand the procedure. The retina, the layer at the back of the eyeball that turns light into electrical signals, which the brain converts into images, is extremely difficult to transplant because it's packed with nerve tissue and blood vessels.
These challenges also make it tough to research transplantation. “This greatly limits their use for experiments, particularly when it comes to the effectiveness of new drugs and treatments,” said Maria Pia Cosma, a biologist at Barcelona’s Centre for Genomic Regulation (CRG), whose team is working on the ECaBox.
Eye transplants are desperately needed, but they're nowhere in sight. About 12.7 million people worldwide need a corneal transplant, which means that only one in 70 people who require them, get them. The gaps are international. Eye banks in the United Kingdom are around 20 percent below the level needed to supply hospitals, while Indian eye banks, which need at least 250,000 corneas per year, collect only around 45 to 50 thousand donor corneas (and of those 60 to 70 percent are successfully transplanted).
As for retinas, it's impossible currently to put one into the eye of another person. Artificial devices can be implanted to restore the sight of patients suffering from severe retinal diseases, but the number of people around the world with such “bionic eyes” is less than 600, while in America alone 11 million people have some type of retinal disease leading to severe vision loss. Add to this an increasingly aging population, commonly facing various vision impairments, and you have a recipe for heavy burdens on individuals, the economy and society. In the U.S. alone, the total annual economic impact of vision problems was $51.4 billion in 2017.
Even if you try growing tissues in the petri dish route into organoids mimicking the function of the human eye, you will not get the physiological complexity of the structure and metabolism of the real thing, according to Cosma. She is a member of a scientific consortium that includes researchers from major institutions from Spain, the U.K., Portugal, Italy and Israel. The consortium has received about $3.8 million from the European Union to pursue innovative eye research. Her team’s goal is to give hope to at least 2.2 billion people across the world afflicted with a vision impairment and 33 million who go through life with avoidable blindness.
Their method? Resuscitating cadaveric eyes for at least a month.
If we succeed, it will be the first intact human model of the eye capable of exploring and analyzing regenerative processes ex vivo. -- Maria Pia Cosma.
“We proposed to resuscitate eyes, that is to restore the global physiology and function of human explanted tissues,” Cosma said, referring to living tissues extracted from the eye and placed in a medium for culture. Their ECaBox is an ex vivo biological system, in which eyes taken from dead donors are placed in an artificial environment, designed to preserve the eye’s temperature and pH levels, deter blood clots, and remove the metabolic waste and toxins that would otherwise spell their demise.

Scientists work on resuscitating eyes in the lab of Maria Pia Cosma.
Courtesy of Maria Pia Cosma.
“One of the great challenges is the passage of the blood in the capillary branches of the eye, what we call long-term perfusion,” Cosma said. Capillaries are an intricate network of very thin blood vessels that transport blood, nutrients and oxygen to cells in the body’s organs and systems. To maintain the garland-shaped structure of this network, sufficient amounts of oxygen and nutrients must be provided through the eye circulation and microcirculation. “Our ambition is to combine perfusion of the vessels with artificial blood," along with using a synthetic form of vitreous, or the gel-like fluid that lets in light and supports the the eye's round shape, Cosma said.
The scientists use this novel setup with the eye submersed in its medium to keep the organ viable, so they can test retinal function. “If we succeed, we will ensure full functionality of a human organ ex vivo. It will be the first intact human model of the eye capable of exploring and analyzing regenerative processes ex vivo,” Cosma added.
A rapidly developing field of regenerative medicine aims to stimulate the body's natural healing processes and restore or replace damaged tissues and organs. But for people with retinal diseases, regenerative medicine progress has been painfully slow. “Experiments on rodents show progress, but the risks for humans are unacceptable,” Cosma said.
The ECaBox could boost progress with regenerative medicine for people with retinal diseases, which has been painfully slow because human experiments involving their eyes are too risky. “We will test emerging treatments while reducing animal research, and greatly accelerate the discovery and preclinical research phase of new possible treatments for vision loss at significantly reduced costs,” Cosma explained. Much less time and money would be wasted during the drug discovery process. Their work may even make it possible to transplant the entire eyeball for those who need it.
“It is a very exciting project,” said Sanjay Sharma, a professor of ophthalmology and epidemiology at Queen's University, in Kingston, Canada. “The ability to explore and monitor regenerative interventions will increasingly be of importance as we develop therapies that can regenerate ocular tissues, including the retina.”
Seemingly, there's no sacred religious text or a holy book prohibiting the practice of eye donation.
But is the world ready for eye transplants? “People are a bit weird or very emotional about donating their eyes as compared to other organs,” Cosma said. And much can be said about the problem of eye donor shortage. Concerns include disfigurement and healthcare professionals’ fear that the conversation about eye donation will upset the departed person’s relatives because of cultural or religious considerations. As just one example, Sharma noted the paucity of eye donations in his home country, Canada.
Yet, experts like Sharma stress the importance of these donations for both the recipients and their family members. “It allows them some psychological benefit in a very difficult time,” he said. So why are global eye banks suffering? Is it because the eyes are the windows to the soul?
Seemingly, there's no sacred religious text or a holy book prohibiting the practice of eye donation. In fact, most major religions of the world permit and support organ transplantation and donation, and by extension eye donation, because they unequivocally see it as an “act of neighborly love and charity.” In Hinduism, the concept of eye donation aligns with the Hindu principle of daan or selfless giving, where individuals donate their organs or body after death to benefit others and contribute to society. In Islam, eye donation is a form of sadaqah jariyah, a perpetual charity, as it can continue to benefit others even after the donor's death.
Meanwhile, Buddhist masters teach that donating an organ gives another person the chance to live longer and practice dharma, the universal law and order, more meaningfully; they also dismiss misunderstandings of the type “if you donate an eye, you’ll be born without an eye in the next birth.” And Christian teachings emphasize the values of love, compassion, and selflessness, all compatible with organ donation, eye donation notwithstanding; besides, those that will have a house in heaven, will get a whole new body without imperfections and limitations.
The explanation for people’s resistance may lie in what Deepak Sarma, a professor of Indian religions and philosophy at Case Western Reserve University in Cleveland, calls “street interpretation” of religious or spiritual dogmas. Consider the mechanism of karma, which is about the causal relation between previous and current actions. “Maybe some Hindus believe there is karma in the eyes and, if the eye gets transplanted into another person, they will have to have that karmic card from now on,” Sarma said. “Even if there is peculiar karma due to an untimely death–which might be interpreted by some as bad karma–then you have the karma of the recipient, which is tremendously good karma, because they have access to these body parts, a tremendous gift,” Sarma said. The overall accumulation is that of good karma: “It’s a beautiful kind of balance,” Sarma said.
For the Jews, Christians, and Muslims who believe in the physical resurrection of the body that will be made new in an afterlife, the already existing body is sacred since it will be the basis of a new refashioned body in an afterlife.---Omar Sultan Haque.
With that said, Sarma believes it is a fallacy to personify or anthropomorphize the eye, which doesn’t have a soul, and stresses that the karma attaches itself to the soul and not the body parts. But for scholars like Omar Sultan Haque—a psychiatrist and social scientist at Harvard Medical School, investigating questions across global health, anthropology, social psychology, and bioethics—the hierarchy of sacredness of body parts is entrenched in human psychology. You cannot equate the pinky toe with the face, he explained.
“The eyes are the window to the soul,” Haque said. “People have a hierarchy of body parts that are considered more sacred or essential to the self or soul, such as the eyes, face, and brain.” In his view, the techno-utopian transhumanist communities (especially those in Silicon Valley) have reduced the totality of a person to a mere material object, a “wet robot” that knows no sacredness or hierarchy of human body parts. “But for the Jews, Christians, and Muslims who believe in the physical resurrection of the body that will be made new in an afterlife, the [already existing] body is sacred since it will be the basis of a new refashioned body in an afterlife,” Haque said. “You cannot treat the body like any old material artifact, or old chair or ragged cloth, just because materialistic, secular ideologies want so,” he continued.
For Cosma and her peers, however, the very definition of what is alive or not is a bit semantic. “As soon as we die, the electrophysiological activity in the eye stops,” she said. “The goal of the project is to restore this activity as soon as possible before the highly complex tissue of the eye starts degrading.” Cosma’s group doesn’t yet know when they will be able to keep the eyes alive and well in the ECaBox, but the consensus is that the sooner the better. Hopefully, the taboos and fears around the eye donations will dissipate around the same time.